What is the Lyme Disease Diagnostic Devices Market Size?
The global lyme disease diagnostic devices market size is accounted at USD 2.82 billion in 2025 and predicted to increase from USD 2.97 billion in 2026 to approximately USD 4.79 billion by 2035, expanding at a CAGR of 5.44% from 2026 to 2035.
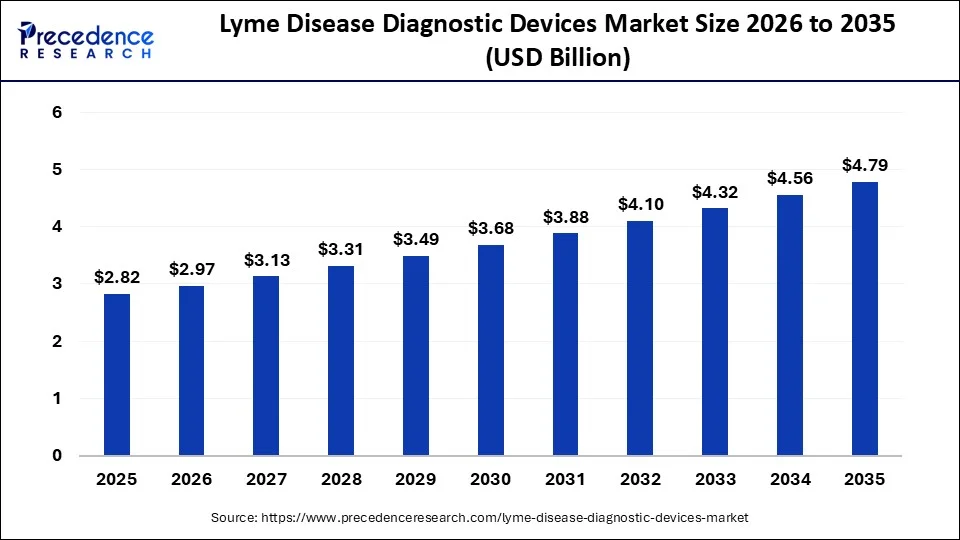
Lyme Disease Diagnostic Devices Market Key Takeaways
- North America led the global market with the largest market share of 40% in 2025.
- By Product, the serological test segment has held the largest revenue share in 2025.
- By Product, the lymphocytic transformation tests segment is anticipated to expand at a significant CAGR during the projected period.
- By End User, the hospitals segment has held the largest revenue share in 2025.
What is the Lyme Disease Diagnostic Devices Market?
Upsurge in incidence of Lyme disease, growing policy attention on improving health care services, and the advent of new diagnostic tests for Lyme disease pushes the market. Nonetheless, lack of knowledge of the disease and ineffectiveness in Lyme disease research reduces the market. Additionally, growth in key players' interests in Lyme disease diagnosis R&D is predicted to provide lucrative business growth opportunities. Moreover, increasing number of cases of tick-borne infectious diseases is boosting Lyme disease diagnostics market. Vector-borne diseases are illnesses caused by pathogens and parasites in human inhabitants. Each year greater than 1 billion individuals are infected and higher than 1 million people die from vector-borne illnesses. They can be infected by bacteria, parasites or viruses. More than 16% of the ailment and disability endured globally is because of vector-borne illnesses, with more than 50% of world's population presently assessed to be at risk of these ailments.
How is AI contributing to the Lyme Disease Diagnostic Devices Market?
Artificial Intelligence is not only able to diagnose Lyme disease better but also faster. The technology behind AI makes the whole thing more efficient. For example, with deep learning, even very subtle signs of the disease can be recognized while using predictive analytics in outbreak monitoring and public health planning. AI is able to support the development of personalized diagnostic practices, and in turn, clinical settings become more efficient, less prone to manual interpretation errors, and very innovative.
Lyme Disease Diagnostic Devices Market Growth Factors
- Growing prevalence of Lyme disease
- Increasing number of cases of tick-borne infectious diseases
- Growing investment in research and development
- Technological advancement
Lyme Disease Diagnostic Devices Market Outlook
[[market_outlook]]
Market Scope
| Report Highlights | Details |
| Market Size in 2025 | USD 2.82 billion |
| Market Size in 2026 | USD 2.97 billion |
| Market Size by 2035 | USD 4.79 billion |
| Growth Rate from 2026 to 2035 | CAGR of 5.44% |
| Largest Market | North America |
| Fastest Growing Market | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, End User, Region |
| Regions Covered | North America, Asia Pacific, Europe, Latin America, Middle East and Africa |
Lyme Disease Diagnostic Devices Market Segment Insights
[[segment_insights]]
Lyme Disease Diagnostic Devices Market Regional Insights
[[regional_insights]]
Lyme Disease Diagnostic Devices Market Value Chain Analysis
[[value_chain]]
Lyme Disease Diagnostic Devices Market Key Players' Offering
[[market_company]]
Other Major Key Players
- Bio-Rad Laboratories, Inc.
- Thermo Fisher Scientific, Inc.
- Boulder Diagnostics
- T2 Biosystems, Inc.
- Abbott
- Oxford Immunotec Global PLC
- Quidel Corporation
- Affymetrix Inc.
- Hoffmann-La Roche Ltd
- Graphene Frontiers
Recent Developments
- In October 2025, Twin brothers Jack and Will Goodreau launched Brothers Tick Kits, designed to safely remove ticks and protect families from tick-borne illnesses, inspired by their sister's Lyme disease fight. (Source: morningagclips.com)
- In March 2025, Antech launched truRapid™ FOUR, an in-house screening test for canine vector-borne diseases, detecting antibodies for Anaplasma, Ehrlichia, Lyme C6, and heartworm using whole blood, serum, or plasma. (Source: businesswire.com)
Market Segments Covered
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
