Medical Device Outsourcing Market Size and Forecast 2026 to 2035
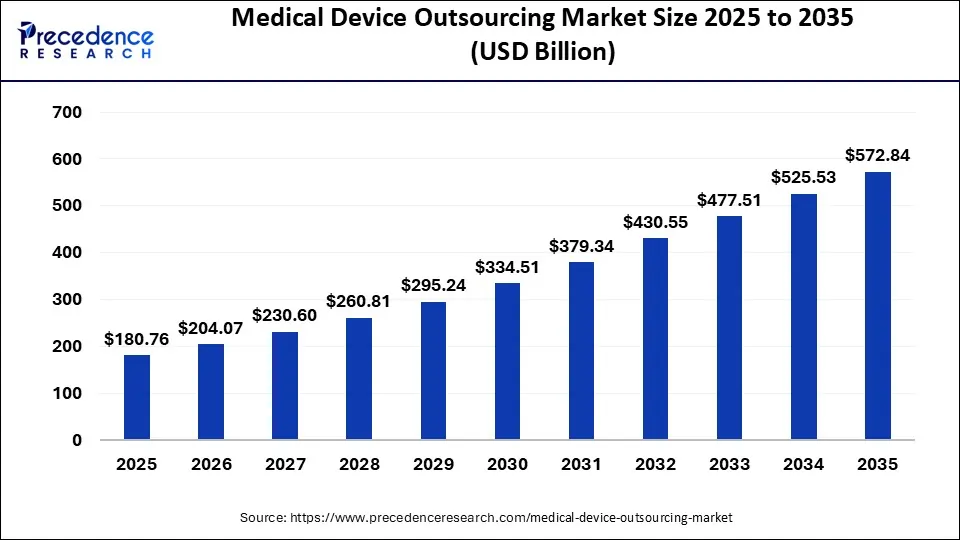
The global medical device outsourcing market size was estimated at USD 180.76 billion in 2025 and is anticipated to reach around USD 572.84 billion by 2035, expanding at a CAGR of 12.23% from 2026 to 2035. The increased demand for non-invasive medical devices is driving the growth of the global medical device outsourcing market. The growing need to reduce product costs and complexity enhances the adoption rate of outsourcing. Additionally, rising demand for affordable devices is fueling the market growth.
Medical Device Outsourcing Market Key Takeaways
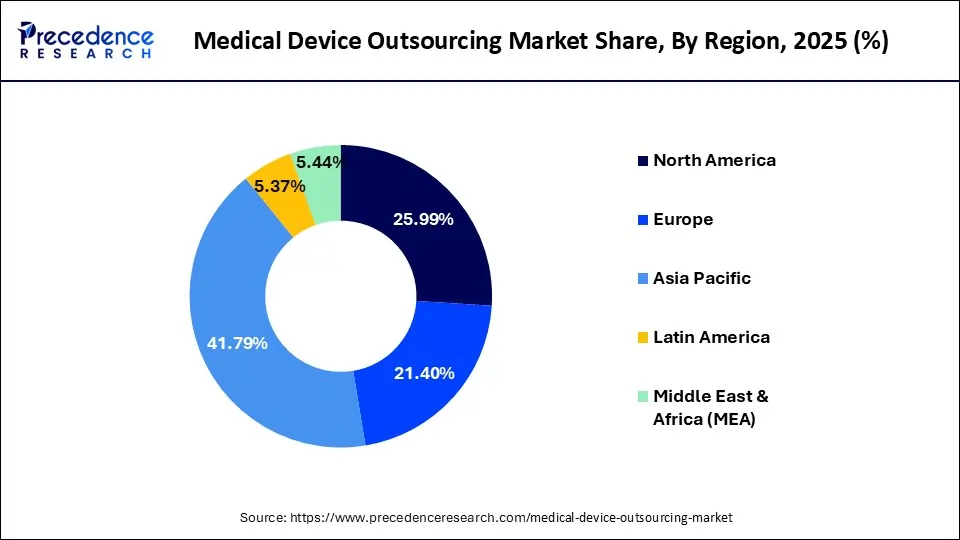
- Asia Pacific dominated the medical device outsourcing market with the largest market share of 41.79% in 2025.
- North America is expected to expand at the fastest CAGR of 12.5% during the forecast period.
- By application, the cardiology segment accounted for the largest share of the market in 2025
- By service, the quality assurance segment generated the biggest market share of 9.56% in 2025.
- By service, the regulatory affairs services is projected to grow at a double digit of CAGR of 13.5% during the forecast period.
Market Overview
The escalating trend toward outsourcing manufacturing operations has become essential as it helps to decrease direct expenses and modernize supply chains while balancing rising operating expenses. At present, a variety of influences are motivating growth in the contract manufacturing segment. Extreme competition has forced device producers to emphasize their core proficiencies of research and development, marketing, clinical education, and sales. Medical device outsourcing bids cost savings and better-quality service delivery. According to a present-day KPMG survey, out of 94 respondents in the medical service and device supplier industries, upgraded service delivery and cost drop are two of the primary influencers for outsourcing. IT outsourcing was recognized by 38% of respondents as the topmost way for cost reduction, whereas 26% of respondents observed it as a good technique to recover service delivery. In contrast, business process outsourcing (BPO) was regarded by 30% of respondents as an uppermost means for cost drop, whereas 19% of respondents thought it was a good method for developing service delivery.
The significance of regulatory compliance is anticipated to lift the growth of consulting services, including quality management systems, remediation, and compliance, thus backing the market growth. However, pricing pressure, budget scrutiny in established countries, and changes in repayment schemes are certain major features expected to upsurge the implementation of cost suppression procedures by the original equipment manufacturers (OEM). These factors are also anticipated to lift medical device outsourcing to emergent nations, including India and China.
“Outsourced Medical Device Production: 2023 Snapshot”
In 2023, the medical device industry increasingly relied on outsourcing to meet global demand and optimize production efficiency. Over 500 million finished devices were produced through contract manufacturing, highlighting the scale and critical role of external partners. Key contributions included 120 million syringes, 90 million catheters, and 60 million glucose meters, showcasing the diversity of devices managed through outsourcing. This approach not only accelerates manufacturing timelines but also allows OEMs to focus on innovation and regulatory compliance. The data underscores the growing dependence on specialized manufacturers to maintain supply, ensure quality, and support the healthcare ecosystem worldwide.

How Outsourcing Innovations in the MedTech Industry are driven by AI
Artificial intelligence (AI) is driving change in the medical device outsourcing marketplace by improving operational efficiencies, speeding up product development cycles, and providing greater data-driven quality assurance. More and more outsourcing partners are using AI-powered tools such as predictive analytics, automated testing, and better supply chain management to assist OEMs in reducing costs and accelerating time-to-market. The dramatic increase in AI usage is leading to a resurgence of strategic partnerships and investments across MedTech as seen by recent industry events like the increased integration of AI into GE HealthCare and the establishment of stronger technology alliances highlighted at RSNA 2025.
These technological advancements aid in the development of complex devices that meet regulatory requirements while requiring outsourcing firms to develop and improve their skillsets and innovative solutions in order to remain competitive in a rapidly changing healthcare environment.
Trends Changing Landscape of Medical Device Outsourcing Market
- Technological advancements- Outsourcing companies utilize automation, robots, additive manufacturing, and smart production systems to increase accuracy, minimize mistakes, decrease time-to-market, and assist with complex designs for next-generation medical devices.
- Demand for high quality medical devices- Medical device manufacturers are turning to industry experts in order to meet the increased demands for regulatory compliance and validated quality systems. Outsourcing companies with sophisticated global compliance capabilities can facilitate quicker regulatory approvals and decreased risk while maintaining ongoing compliance with evolving safety regulations.
- Strategic partnership and acquisitions- There is a growing trend toward long-term strategic partnerships, mergers and acquisitions as companies pursue total outsourcing solutions for the design, engineering, testing and manufacturing phases of the product life cycle. Companies are pursuing these total outsourcing solutions in an effort to take advantage of lower costs, accelerate product innovation and build stronger supply chain resilience.
- Digital transformation and automation- The digital transformation of the medical device outsourcing market relies on implementing digital tools such as artificial intelligence-based design, data analytics, cloud-based platforms and automated quality inspection systems.
Medical Device Outsourcing Market Growth Factors
- Rising demand for non-invasive and minimally invasive medical devices is the key factor enhancing the growth of the market.
- The growing prevalence of the disease requires advanced medical devices, which has enhanced the production of medical devices. The need for reducing production costs and complexity is driving the need for outsourcing.
- The need to comply with regulatory compliance to maintain quality and safety standards is playing a favorable role in market growth.
- Rising numbers of cosmetic surgeries are requiring medical device outsourcing to prevent complexity and improve safety.
- The rising adoption of personalized medicines requires the development of customized medical devices, making outsourcing more essential.
- Government initiatives and investment in R&D healthcare and the pharmaceutical sector encourage manufacturing companies to adopt cutting-edge technologies.
- The growing adoption of outsourcing devices in medical device manufacturing is boosting the market growth.
Market Scope
| Report Highlights | Details |
| Market Size in 2025 | USD 180.76 Billion |
| Market Size in 2026 | USD 204.07 Billion |
| Market Size by 2035 | USD 572.84 Billion |
| Growth Rate from 2026 to 2035 | CAGR of 12.23% |
| Largest Market | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Service Type, Application Type, Region Type |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market dynamics
Driver
Growing complexity and cost
The rising complexity and cost of medical device development and production are the key drivers of the global medical device outsourcing market. The rising prevalence of chronic disease and the number of surgeries is driving demand for advanced, safe, and more effective medical devices; however, the complexity and expensive production of medical devices occurring due to regulatory compliance, clinical trials, quality control, and quality assurance are driving the need for outsourcing. The rising need for specialized expertise to develop and maintain medical devices is fueling the market growth. Additionally, the need for increasing scalability, flexibility, cost-effectiveness, quality, and reliability of medical devices is driving the need for outsourcing, leading to a positive influence on the market.
Restraint
Data security
The medical devices outsourcing market significantly faces challenges in managing access to sensitive information such as patient data and product designs. The critical medical device development process is likely to be attacked by unauthorized aspects due to outsourcing. Additionally, the connection of medical device outsourcing can cause challenges such as cyber threats. The implementation of data security measures and risk assessment of outsourcing partners with data security practices can help to overcome such a restraint.
Opportunity
Rising demand for cost-effective medical devices
The rising prevalence of chronic disease and the number of surgeries requiring cutting-edge medical devices. However, the high costs of medical devices are hampering the healthcare expenditure. The rising demand for affordable medical devices by healthcare professionals and patients is holding market potential. Outsourcing is beneficial for low-cost manufacturing as it helps to reduce cost-related regulatory compliance, clinical trials, waste management, and complexity. Large-scale outsourcing helps to reduce overall medical device production and maintenance costs. Additionally, access to specialized expertise further helps to reduce risk and complexity, making more cost-effective production and manufacturing. The ability of outsourcing to reduce product development downtime is increasing their adoption in medical device manufacturing practice.
Application Insights
The cardiology segment underwent notable growth in the medical device outsourcing market during 2025. In cardiology, medical device outsourcing plays an important role. Cardiac implantable electronic devices including cardiac loop recorders, biventricular pacemakers, implantable cardioverter defibrillator (ICD), and pacemakers are designed to help control or monitor irregular heartbeats in people with specific heart rhythm disorders and heart failure.
- In March 2025, the U.S. launch of Flyrcado (flurpiridaz F 18) injection was announced by GE HealthCare. The company made the announcement as it prepares to showcase new artificial intelligence (AI)-powered innovations at the 2025 American College of Cardiology (ACC) Annual Scientific Session and Expo, reflecting its strategy to leverage AI to help increase efficiency and allow seamless data integration across the cardiology care pathway.
GE HealthCare Introduces Measures to Improve Cardiac Care Pathway - Medical Product Outsourcing
The general and plastic surgery device segment will gain a significant share of the market over the studied period of 2026 to 2035. General and plastic surgery benefits include psychological benefits, restored functionality, better physical health, improved self-confidence, and enhanced physical appearance. Plastic surgeries have been found to enhance the mental health of people. Researchers have found that plastic surgery can increase happiness and boost patient's self-esteem.
- In August 2025, the formation of wholly owned subsidiary in Australia and New Zealand, alongside an exclusive partnership with High Tech Medical Pty Ltd. to launch the ELIXIR MD Device across the region was announced by the ELIXIR MD, Inc., a pioneer in advanced medical technology and proprietary treatment protocols for plastic surgery.
Medical Device Outsourcing Market 2023-2025, By Application ($ Million)
| By Application | 2023 | 2024 | 2025 |
| Drug delivery | 29,617.8 | 33,581.4 | 38,107.9 |
| Dental | 12,725.7 | 14,300.8 | 16,085.0 |
| Diabetes care | 14,686.9 | 16,476.5 | 18,500.1 |
| Cardiology | 24,303.3 | 27,339.5 | 30,782.1 |
| Endoscopy | 13,725.3 | 15,429.1 | 17,359.6 |
| IVD | 3,855.5 | 4,387.9 | 4,997.8 |
| Ophthalmic | 21,736.3 | 24,559.5 | 27,773.8 |
| Diagnostic imaging | 3,183.8 | 3,573.5 | 4,014.4 |
| Orthopedic | 8,759.5 | 9,795.1 | 10,962.0 |
| General and plastic surgery | 7,128.2 | 8,052.4 | 9,104.5 |
| Others | 2,468.6 | 2,753.5 | 3,073.7 |
Services Insights
The quality assurance segment enjoyed a prominent position in the market during 2025 and is predicted to witness significant growth in the market over the forecast period. Medical device quality assurance is important to ensure medical devices are of uniform and high quality. The purpose of QA in the medical device industry is to systematically ensure that devices consistently meet quality specifications and related needs through their lifecycle, from development to post-market.
- In March 2025, OpenQMS, an open access, web-based platform for helping companies navigate the Quality Management process throughout the medical device development lifecycle was launched by Archimedic, a medical device development firm that provides design, regulatory, and go-to-market services to its clients.
Archimedic Launches OpenQMS, an Open-Access Quality Platform for Medical Device Development
Medical Device Outsourcing Market 2023-2025, By Service ($ Million)
| By Service | 2023 | 2024 | 2025 |
| Product Upgrade Services | 5,620.3 | 6,338.9 | 7,155.8 |
| Regulatory Affairs Services | 9,019.1 | 10,204.8 | 11,556.5 |
| Quality Assurance | 13,391.7 | 15,316.5 | 17,529.7 |
| Product Maintenance Services | 7,629.9 | 8,618.4 | 9,743.6 |
| Product Testing & Sterilization Services | 9,625.0 | 10,881.9 | 12,313.8 |
| Product Design and Development Services | 11,369.9 | 12,844.5 | 14,523.2 |
| Product Implementation Services | 7,709.5 | 8,717.6 | 9,866.1 |
| Contract Manufacturing | 77,825.4 | 87,326.3 | 98,072.1 |
Asia Pacific Medical Device Outsourcing Market Size and Growth 2026 to 2035
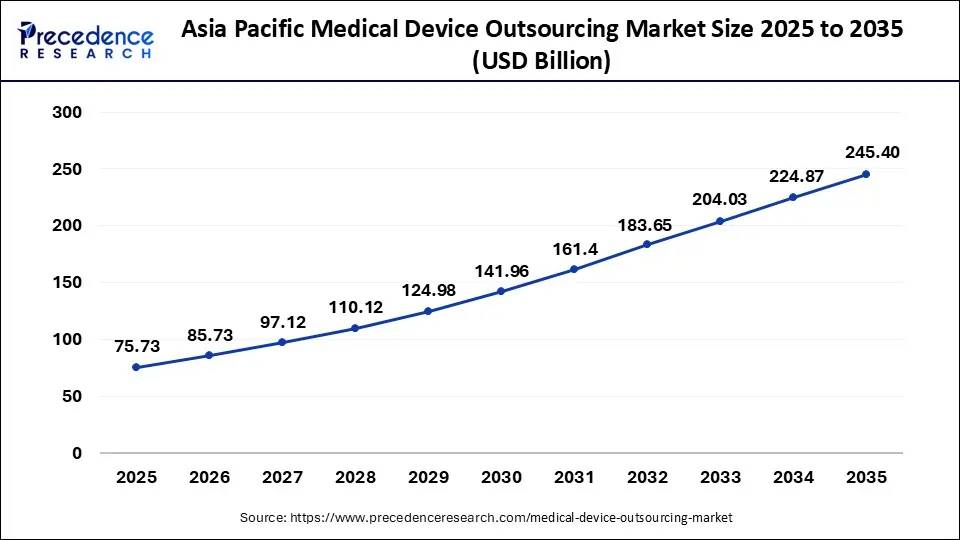
The Asia Pacific medical device outsourcing market size was evaluated at USD 75.73 billion in 2025 and is predicted to be worth around USD 245.40 billion by 2035, rising at a CAGR of 12.48% from 2026 to 2035.
Asia Pacific dominated the medical device outsourcing market with the largest market share of 41.79% in 2025.
For instance, In November 2025, the Indian government invested in a Rs 500 crore scheme aimed at strengthening key areas such as manufacturing, skill development, clinical studies, infrastructure, and industry promotion. The investment is expected to boost India's medical device industry.
North America s expected to expand at the fastest CAGR of 12.5% during the forecast period.
North America is expected to witness significant growth in the medical device outsourcing market due to the availability of advanced healthcare infrastructure, which supports the innovations and developments of advanced outsourcing medical devices. The expanding healthcare infrastructure in North America is contributing to the market growth. Additionally, government and regulatory initiatives and investments in the research & development sector are shaping the market in North America.
Though the total medical device industry in the U.S. is estimated to continue growing at a robust rate, the outsourced manufacturing market is anticipated to perceive even faster growth. As medical device producers attempt to recover margins and time to market, outsourced manufacturing will continue to grow in approval.
Asia Pacific dominated the global medical device outsourcing market in 2025. Rising adoption of connected medical care and smart devices, rising investments to outsource medical device designing and development, rising demand for surgical interventions and medical diagnosis, and rapid innovation in medical device technologies are driving the growth of the market in the Asia Pacific region.
- For instance, in July 2025, the Invisalign System with mandibular advancement featuring occlusal blocks, designed specifically to address Class II skeletal and dental correction by simultaneously advancing the mandible while aligning the teeth was launched by Align Technology Inc. in India.
New Mandibular Advancement for Invisalign System Released in India - Medical Product Outsourcing
North America is projected to host the fastest growing market in the coming years. Increased medical device consumption, increasing demand for cost-effective production solutions, enhanced healthcare infrastructure, and rapidly aging population are contributing the growth of the medical device outsourcing market.
- In April 2025, operations in the U.S. with MeKo MedTech Inc. in Bloomington, Minnesota was launched by MeKo Manufacturing. Sarstedt, Germany-based MeKo specializes in high-precision laser material processing, with more than three decades of experience with laser cutting, laser drilling, and laser welding of metals and other materials.
MeKo Manufacturing launches US medtech operations
Does Europe Have A Stronger Level Of Growth In The Medical Device Outsourcing Market?
Europe is a strong growth region; however, with continued demand for specialized, high-quality medical devices and a strong focus on precision engineering, Europe will continue to grow as we see more and more manufacturers outsourcing their regulatory support, testing, manufacturing of complex components, and other outsourcing services. Europe is also benefiting from advanced healthcare systems, the continued focus on innovative designs and new products, and the continued requirement for compliant manufacturing. With implantable and diagnostic devices being particularly susceptible to variations in quality, companies are going to begin working with outsourcing partners who demonstrate strong controls over the quality and traceability of their goods.
Germany
Germany is the leader of supply chain events within the European region due to their precision manufacturing capabilities, strong engineering base, and proven ability to deliver high-quality medical devices. The majority of outsourcing activities will be focused on complex component, automated production processes, and processes that are compliant with all applicable regulations.
How Latin America Emerging in Medical Device Outsourcing Market?
With manufacturers improving their manufacturing capabilities, labor costs remaining low and increasing demand for healthcare services, Latin American countries are rapidly emerging as viable offshore outsourcing destinations for medical device companies to outsource assembly, packaging and selected manufacturing operations. For example, there has been increasing activity that demonstrates expanding, local-market growth, improved regulatory frameworks and an increase in the amount of foreign direct investment, which are all contributing to an increase in outsourcing to Latin America.
Mexico
Mexico as the leading country in this region and the first choice for offshoring of assembly, packaging and selected manufacturing operations, Mexico is in the best geographic position to take advantage of the costs of manufacturing and the developing medical device manufacturing clusters in Mexico. Furthermore, Mexico's advantages to supporting offshoring in this country include a qualified labor force and rapidly improving infrastructure and multiple advantages of cross-border trade.
How Middle East and Africa Becoming the Notably Growing Region in Medical Device Outsourcing Market?
The Middle East and African area is clearly expanding, as governments sink wealth into building health facilities and producing local medical devices. The increase of order for low and medium complexity devices, in addition to the constant effort to limit dependence on imported goods, has been the major driver for outsourced manufacturing. There is also a strong commitment for regionally under to form regional partnerships with international companies in the manufacturing, assembling, distributing, and packaging functions of an international supply chain. Furthermore, as regulatory improvement occurs in the region and the participation of the private sector increases, there is a strong growth in outsourced manufacturing.
Saudi Arabia
Due to their high levels of investment in healthcare, creating local manufacturing capacity, and providing incentives to foreign medical device manufacturers to locate operations in Saudi Arabia, the country is leading this market. Existing relationships with international medical device manufacturers will enable the development of a local supply chain and the growth in the assembly, localization, and supply chain development for the domestic market.
Key Companies & Market Share Insights
Outsourcing gives OEMs the prospect to take benefit of resources such as labor, machinery or materials. It proves cost-efficient to outsource production particularly if maneuvers and expertise of machinery can be handled from outside than to fetch an expert in the headquarters and then train individuals. Due to outsourcing despite of escalating operating expenses, the medical device manufacturing has exhibited significant growth during the last few years. For example, Medtech company have accomplished to sustain industry productivity by supporting an extremely high level of assurance to product innovation and by implementing a variety of creative cutbacks measures such as progressively popular approach of outsourced manufacturing.
Medical Device Outsourcing Market Companies

- Intertek Group PLC
- TüvSüd AG
- Wuxi Apptec
- SGS SA
- Toxikon, INC.
- Eurofins Scientific
- American Preclinical Services
- Sterigenics International LLC
- Pace Analytical Services LLC.
- North American Science Associates, Inc.
- Charles River Laboratories International, Inc.
Leader's Announcement
- In November 2024, Shri JP Nadda, Union Minister for Chemicals and Fertilizers, stated on the launching of a Rs 500 crore scheme for the medical device industry in India: “The investment may seem small, but the result is huge.”
Recent Developments
- In July 2025, the launch of NYEMED 7312, advanced non-PFAS damping grease specifically for the medical devices was announced by Fusch Lubricants Co. It provides reliability, motion control, and biocompatibility in critical medical applications.
Fuchs Lubricants launches non-PFAS damping grease for devices
- In November 2024, a new AI Innovation Lab to accelerate early-concept artificial intelligence (AI) solutions was opened by GE HealthCare. The projects are a part of GE Healthcare's broader AI and digital strategy, which is focused on integrating AI into medical devices, building AI applications that improve decision making across the care journey and disease states, and using AI to support better outcomes and operational efficiencies system wise.
GE Healthcare Launches New AI Innovation Lab - Medical Product Outsourcing
Segments Covered in the Report
By Service
- Product Upgrade Services
- Regulatory Affairs Services
- Legal representation
- Clinical trials applications
- Regulatory writing and publishing
- Quality Assurance
- Product Maintenance Services
- Product Testing & Sterilization Services
- Product Design and Development Services
- Molding
- Designing & engineering
- Machining
- Packaging
- Product Implementation Services
- Contract Manufacturing
- Accessories manufacturing
- Component manufacturing
- Device manufacturing
- Assembly manufacturing
By Application
- Drug delivery
- Dental
- Diabetes care
- Cardiology
- Endoscopy
- IVD
- Ophthalmic
- Diagnostic imaging
- Orthopedic
- General and plastic surgery
- Others
By Regional
- North America
- U.S.
- Canada
- Europe
- U.K.
- Germany
- France
- Asia Pacific
- China
- India
- Japan
- South Korea
- Middle East & Africa
- Latin America
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content
.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
