What is the Medical Devices Reimbursement Market?
The global medical devices reimbursement market size is calculated at USD 621.38 billion in 2025 and is predicted to increase from USD 676.19 billion in 2026 to approximately USD 1,431.59 billion by 2035, expanding at a CAGR of 8.70% from 2026 to 2035.
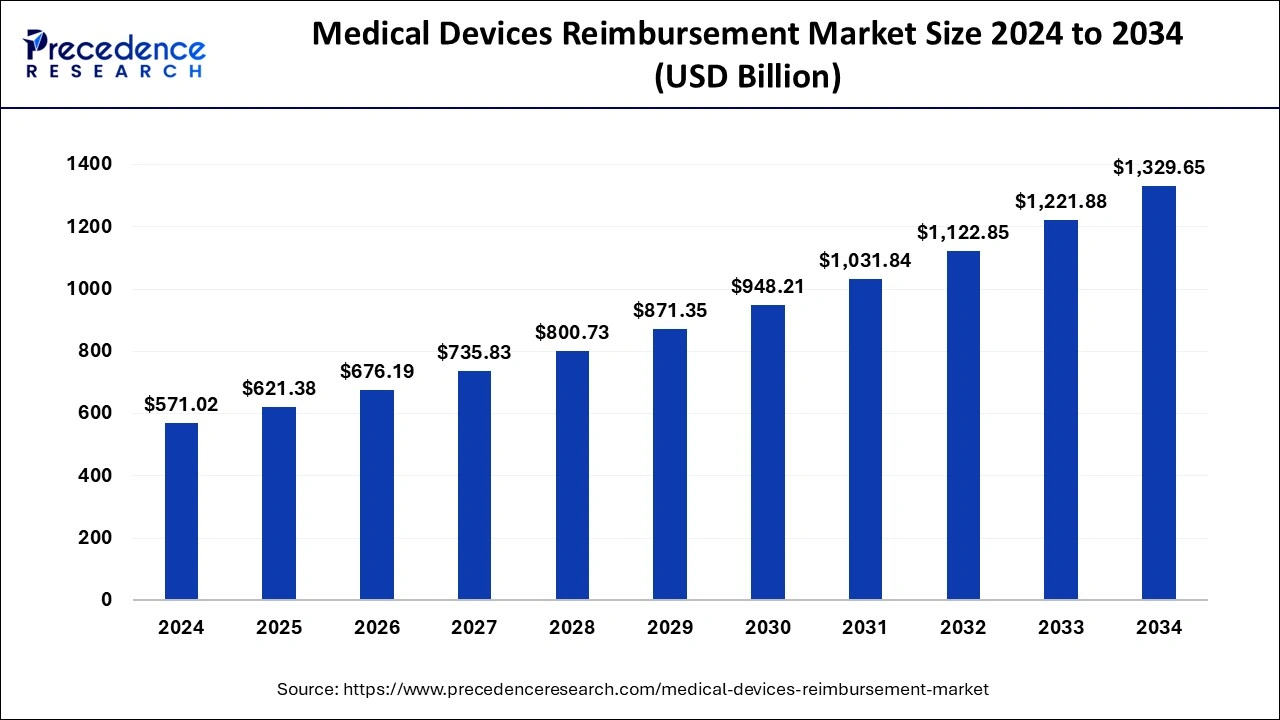
Medical Devices Reimbursement Market Key Takeaways
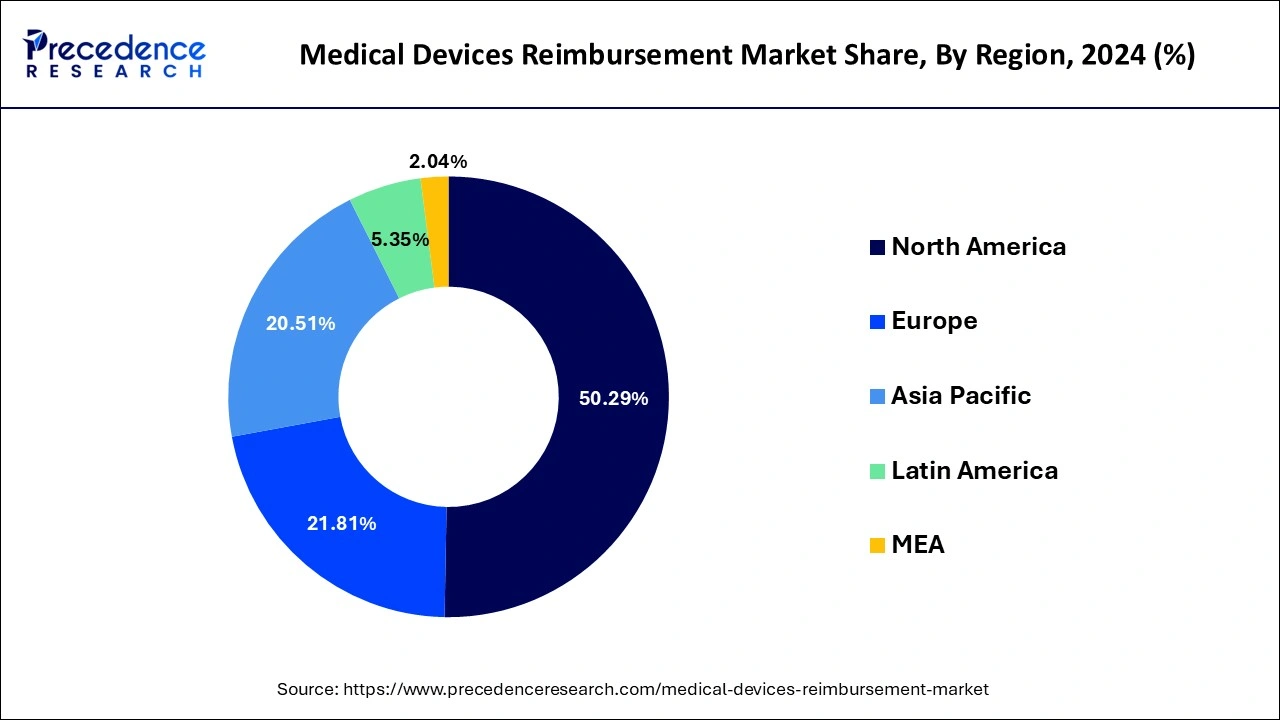
- North America dominated the global medical devices reimbursement market with the largest market share of 50.29% in 2025.
- Asia Pacific is projected to expand at the fastest CAGR during the forecast period.
- By payer, the private medical devices payers segment contributed the highest market share in 2025.
- By payer, the public medical devices payers segment is expected to grow at a significant CAGR from 2026 to 2035.
- By healthcare setting, the hospitals segment captured the biggest market share in 2025.
- By healthcare setting, the outpatient facilities segments is estimated to be the fastest-growing segment during the forecast period.
AI impact
Artificial intelligence holds potential to revolutionize healthcare with AI-enabled clinical services collaborated with patient care, assisting clinicians in diagnosing, treating patients. The strategies of financing and reimbursement for AI-based medical devices change across different countries. They are generally covered by public and private health insurance programs.AI powered medical devices could be reimbursed if they meet the coverage criteria of the insurance providers, fueling the technological facet of medical devices reimbursement market.
Market overview
The regulatory bodies across the globe are recognizing the value of medical devices reimbursement along with favorable healthcare policies. Regulatory bodies and government are collaboratively working to achieve a supportive reimbursement environment and providing clearer guidelines for device reimbursement. Thus, the rise in healthcare expenditure world widely in economically evolving countries is the key driving factor for the market.
However, lack of proper guidance, discontinuation of coverage, strict reimbursement policies along with reimbursement coding and documentation challenges are impeding factors for the market's growth. Though, advancement in healthcare for improved living status and better access for medical care holds potential to combat these Challanges, expecting to increase markets growth in the forecast years.
Medical Devices Reimbursement Market Growth factors
- Increasing incidences of chronic health conditions due to age-related decline in health across the globe.
- Increasing demand for medical devices that help in diagnosis, treatment and management of health conditions.
- Healthcare providers and manufacturers are inclined to develop and offer innovative medical devices that address healthcare requirements.
- The majority of persons' healthcare reimbursement is covered by the governments of their country.
- Increasing technical advancements in electronic medical records.
- Increasing public awareness about insurance policies along with rising ageing population is expected to proliferate the global market.
- In certain Asian countries, hospitals and outpatient facilities get reimbursed for the medical devices by government
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 621.38 Billion |
| Market Size in 2026 | USD 676.19 Billion |
| Market Size by 2035 | USD 1,431.59 Billion |
| Growth Rate from 2026 to 2035 | CAGR of 8.70% |
| Largest Market | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Payer, Healthcare Setting, Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Payer Insights
The private medical devices payers segment contributed the highest market share in 2024. The presence of high number of private businesses in the industry is credited with the segment's rise. Over the forecast period, the segment is expected to maintain its dominance. Furthermore, the rising prevalence of diabetes and cancer, as well as increased need for better treatment, are driving factors for the growth of the segment during the forecast period.
The public medical devices payers segment is expected to grow at a significant CAGR from 2025 to 2034. The rising acceptance of public payers in emerging and developed regions, as well as increased innovation in the production of new medical devices, are important factors driving the public medical device payer's segment growth.
Healthcare Setting Insights
The hospitals segment captured the biggest market share in 2024. During the forecast period, the segment is expected to retain its dominance. Additionally, the segment is being driven by the growing ageing population and the rising prevalence of chronic disorders. In large scale settings such as hospitals, the rising frequency of chronic illnesses is boosting demand for medical devices reimbursement.
The outpatient facilities segments is estimated to be the fastest-growing segment during the forecast period. Due to increased research and development investments and the rising frequency of chronic diseases, the outpatient facilities segment is expected to grow at the fastest rate over the projection period. Furthermore, the segment is expected to be fueled by the growing need for pharmaceuticals and their cost-effective production.
The medical devices reimbursement market is relatively fragmented, with a few large-scale worldwide firms and several small-scale regional competitors. They have long term contracts with medical care organizations around the world and are forming strategic alliances and collaborations with other companies in different countries to expand their business. The significant market innovations will also fuel the global industry's growth. To strengthen their position in the medical devices reimbursement market, major firms use strategies such as collaborations, mergers, acquisitions, and the development of new products. To create technologically improved products, key market players are heavily spending in research and development activities.
Regional Insights
What is the U.S. Medical Devices Reimbursement Market Size?
The U.S. medical devices reimbursement market size is exhibited at USD 234.37 billion in 2025 and is projected to be worth around USD 544.96 billion by 2035, growing at a CAGR of 8.80% from 2026 to 2035.
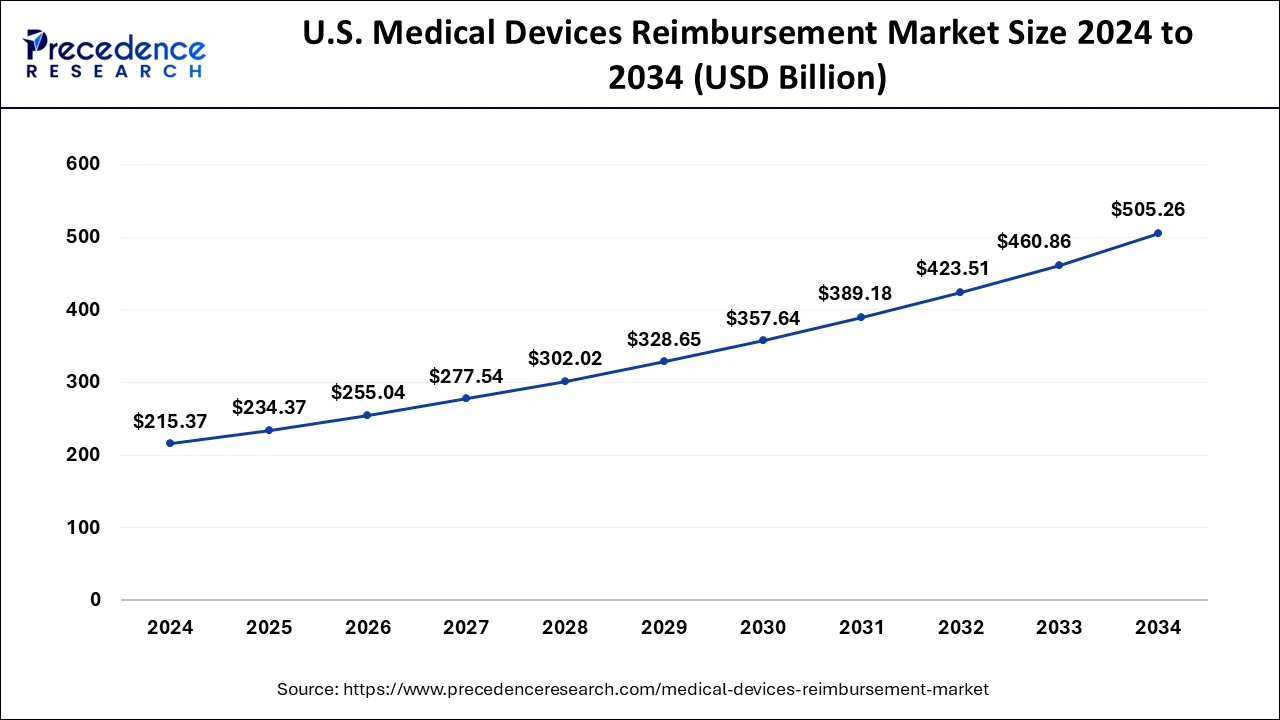
North America dominated the global medical devices reimbursement market with the largest market share of 50.29% in 2025. Over the forecast period, the medical devices reimbursement market is expected to be stimulated by rising cases of numerous severe diseases. Furthermore, in comparison to other regions, the reimbursement structure is the most advanced within the North America region.
U.S. Medical Devices Reimbursement Market
The U.S. dominated the market within North America. The market in the U.S. is growing due to increasing healthcare spending, favorable insurance coverage policies, and a rising focus on advanced and innovative medical technologies. Additionally, the aging population and the growing prevalence of chronic diseases are driving higher demand for reimbursable medical devices.
What Makes Asia Pacific the Fastest-Growing Region in the Market?
Asia Pacific is the fastest-growing region in the medical devices reimbursement market due to rising healthcare expenditure, expanding insurance coverage, and increasing government initiatives to improve access to advanced medical technologies. Rapid economic growth, a growing middle-class population, and rising awareness of innovative medical treatments are further fueling demand for reimbursable devices. Additionally, the prevalence of chronic diseases and an aging population in countries like China, Japan, and India is accelerating market growth across the region.
Japan Market Analysis
Japan is a major contributor to the Asia Pacific medical devices reimbursement market due to its well-established healthcare infrastructure, comprehensive insurance coverage, and strong government support for advanced medical technologies. The country's aging population and high prevalence of chronic diseases also drive significant demand for reimbursable medical devices, boosting market growth.
Why is Europe Considered a Significant Region in the Medical Devices Reimbursement Market?
Europe is considered a significant region in the market due to the region's aging population, which increases the demand for cardiovascular and orthopedic devices under reimbursement programs. Insurers are emphasizing patient access and cost control while maintaining quality standards. Countries like Germany, France, and the UK are leading the region as they benefit from structured policies and supportive government initiatives. These factors help to reflect the region's stability through well-established healthcare funding mechanisms. The region also benefits from harmonized policy frameworks across member states, which promote predictable adoption trends.
What Opportunities Exist in Latin America for the Medical Devices Reimbursement Market?
Latin America offers immense opportunities in the market. These opportunities arise from the region's growing population. Many countries are experiencing rising incomes and an aging population. This demographic shift is leading to an increased demand for healthcare services and advanced medical technologies. Countries like Brazil, Mexico, and Colombia are leading players in the region as they have varying regulatory frameworks, reimbursement systems, and cultural practices, all of which significantly impact the market's growth. Telemedicine and digital health solutions are also gaining traction in the region, driven by the need to improve access to healthcare in remote areas and to reduce healthcare costs.
What Drives the Market in the Middle East & Africa?
The market in the Middle East & Africa (MEA) is driven by the region's increasing healthcare investments, supportive government initiatives aimed at improving healthcare infrastructure, and a rising prevalence of chronic diseases. Countries like South Africa and the UAE are leading players in the market, with significant investments being made to optimize healthcare access and quality. This helps in creating a favorable environment for medical device reimbursement. The region also has diverse healthcare needs, which increases the need for modernization in healthcare. This opens up significant opportunities for growth and innovation.
Medical Devices Reimbursement Market Companies

- UnitedHealth Group
- Allianz
- Nippon Life Insurance
- Aviva
- WellCare Health Plans
- Aetna
- BNP Paribas
- Cigna
- Humana
- CVS Health
Key Developments
- In September 2025, Johnson & Johnson (US) unveiled a new line of surgical instruments designed to improve precision in minimally invasive surgeries. This launch not only reflects the company's commitment to innovation but also positions it to capture a larger share of the surgical device market. The introduction of these advanced instruments could potentially lead to improved surgical outcomes, which may positively influence reimbursement rates as healthcare providers seek to adopt more effective solutions.
(Source: bing.com ) - In July 2025, Abbott Laboratories (US) expanded its cardiovascular device portfolio with the introduction of novel stent technology. This development is significant as it addresses a critical need in the treatment of coronary artery disease. By enhancing its product offerings, Abbott is likely to strengthen its market presence and improve its negotiating power with payers, which is essential in the reimbursement landscape.
(Source: bing.com ) - In September 2024, United healthcare extended their medical policy on cochlear implants to improve hearing services and its access and expand its coverage to younger patients in the U.S. the newly updated policy will covers patients aged six months and older with bilateral sensorineural hearing loss.
- In May 2024, the Indian government launched the national health claim exchange portal, simplifying the insurance claim process for hospitals nationwide. The exchange portal aims to standardize healthcare insurance processing and claims, which will enhance efficiency. NHCX serves as a gateway for exchanging health claim information among insurers, third-party auditors, healthcare providers, and beneficiaries.
Segments Covered in the Report
By Payer
- Public
- Private
By Healthcare Setting
- Hospitals
- Outpatient Facilities
- Others
By Geography
- North America
- U.S.
- Canada
- Mexico
- Europe
- U.K.
- Germany
- France
- Asia-Pacific
- China
- India
- Japan
- South Korea
- Latin America
- Brazil
- Rest of Latin America
- Middle East & Africa (MEA)
- GCC
- North Africa
- South Africa
- Rest of Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
