What is the Melt Electrowriting Technology Market Size?
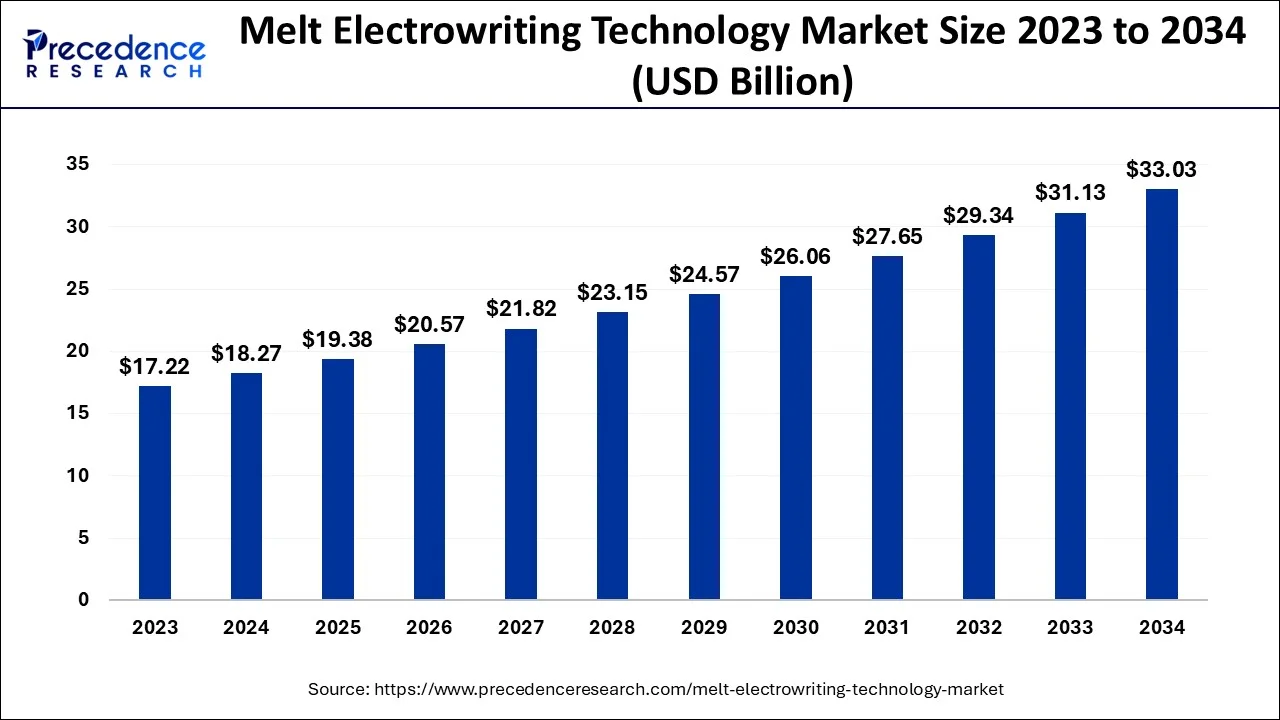
The global melt electrowriting technology market size is calculated at USD 19.38 billion in 2025 and is predicted to increase from USD 20.57 billion in 2026 to approximately USD 34.86 billion by 2035, expanding at a CAGR of 6.05% from 2026 to 2035.
Melt Electrowriting Technology Market Key Takeways
- North America dominated melt electrowriting technology market in 2025.
- By application, the diarthrodial joint melt electro writing segment led the market in 2025.
Market Overview
Melt electrowriting (MEW) is a cutting-edge additive manufacturing process that deposits preset micrometric strands. However, due to the necessity to eliminate polymer jet instabilities, it has thus far been naturally constrained to basic planar and tubular scaffold designs.
The fabrication of fibrous scaffolds with customized micrometric characteristics and anatomically appropriate three-dimensional (3D) geometries for soft tissue engineering applications continue to be a significant problem. Melt electrowriting (MEW) is a cutting-edge additive manufacturing process that deposits preset micrometric strands. However, due to the necessity to eliminate polymer jet instabilities, it has thus far been naturally constrained to basic planar and tubular scaffold designs. In this paper, they push the technological limitations of MEW to create complex fibrous scaffolds with regulated micrometric and patient-specific anatomic characteristics.
Melt electrowriting (MEW) is a new high-resolution additive manufacturing technology based on electrohydrodynamic polymer processing. MEW is most commonly employed to make scaffolds for biomedical purposes, where the microscale fibre location has a significant impact on the macroscopic mechanical characteristics. This paper provides an update on the growing number of polymers processed using MEW as well as several commercial sources of the gold standard poly(-caprolactone) (PCL). A description of MEW-processed polymers other than PCL, such as blends and coated fibres, is provided to give special benefits in biomedical applications. Furthermore, a viewpoint on printer designs and improvements is presented in order to continue extending the range of processable polymers for MEW.
Melt Electrowriting Technology Market Growth Factors
Because of the possibility of patient-specific designs, additive manufacturing (AM) is becoming more popular in biomaterials, tissue engineering, and biofabrication. Every fabrication process has an optimal printing resolution range, which for many techniques is 50-500 m. It is important to be able to print increasingly smaller structural elements at the low micrometre or nanometer scale for biological applications that aim to replicate tissue microarchitecture. Electrohydrodynamic (EHD) methods such as solution electrospinning (SES) melt electrospinning (MES), and melt electrowriting can help achieve such tiny fibre diameters (MEW). This study focuses on the later AM technology, emphasizing the various materials employed and providing a future outlook on the material alternatives for this approach.
The recent invention of electrostatic writing (electro writing) using molten jets gives a possibility to address several key tissue engineering difficulties. An applied voltage is used to create a steady fluid jet with a predictable course that is continually deposited onto a collector. The fibre diameter varies during the procedure, and it is suitable for clinically used polymers. Melt electro writing offers the potential for clinical translation if the implant's biological effectiveness can be enhanced above existing gold standards. It offers scientists a one-of-a-kind chance to conduct low-cost, high-resolution additive manufacturing research that is ideally positioned for clinical translation while utilising current regulatory frameworks.
The creation of biological grafts that resemble the composition, structure, and mechanical behaviour of natural tissue is required for successful cartilage engineering. Melt-electro writing (MEW) was utilised to create arrays of polymeric structures whose role was to direct the formation of spontaneously created cellular aggregates inside these structures, as well as to give tensile reinforcement to the emerging tissues. Inkjet was used to deposit specific quantities of cells into MEW structures, which self-assembled into an orderly array of spheroids within hours, resulting in a hyaline-like hybrid tissue.
The manufactured cartilage was structurally similar to the historical arrangement seen in skeletally immature synovial joints. In less than 15 minutes, this biofabrication framework was utilised to create scaled-up (50mm) cartilage implants comprising over 3,500 cellular aggregates. After 8 weeks in culture, the compressive characteristics of these MEW-reinforced tissues increased 50-fold. Helium ion microscopy revealed the formation of an arcing collagen network within the created tissue. This hybrid bioprinting technology enables the development of cartilage biomimetic grafts for biological joint resurfacing in a varied and scalable manner.
The Melt electro writing (MEW) employs voltage-stabilized jets to precisely position low-micrometre-scale fibres in 3D space. The fibre diameter ranges from 820 nm to 140 m, whereas it is often greater than 200 m with FDM, restricting this additive manufacturing technology's ability to make fully biomimetic implants. MEW, on the other hand, allows for the creation of extremely porous (80-98 vol per cent pore volume), complex, and biomimetic scaffolds. MEW has been utilised, for example, to simulate the anisotropy of the collagen network in cartilage, mechanically reinforcing hydrogels in a way that mimics the behaviour of collagens in cartilage. MEW scaffolds have also been employed as a platform for pre-formed multicellular spheroids assembly.
Market Outlook
- Industry Outlook: The industry is transitioning from basic digital writing tools to intelligent, AI-enabled writing platforms. Future developments will focus on handwriting recognition accuracy, multilingual support, and seamless integration with productivity software.
- Global Expansion Strategy: Global expansion will be driven by localized manufacturing, region-specific pricing models, and partnerships with educational institutions and governments.
- Startup Ecosystem: The startup ecosystem around Melto electronic writing technology is vibrant, particularly in software integration, AI handwriting analytics, and niche educational applications.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 19.38 Billion |
| Market Size in 2026 | USD 20.57 Billion |
| Market Size by 2035 | USD 34.86 Billion |
| Growth Rate from 2026 to 2035 | CAGR of 6.05% |
| Largest Market | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| By Type |
Scaffolds, Poly |
| By Application |
Diarthrodial Joint Melt Electro writing, Functionalized Titanate Nanofiller Melt Electro writing, Coronary Artery Stents Melt Electro writing, Controlled Drug Release Melt Electro writing |
| Regions Covered |
North America, Europe, Asia-Pacific, Latin America, Middle East & Africa |
Segment Insights
Type Insights
The electrohydrodynamic stability of direct-written fluid jets is being investigated for the purpose of designing and manufacturing tissue engineering scaffolds with the necessary fibre diameters. Melt electro writing is shown to be capable of producing a wide range of fibres with distinct diameters (2-50 m) using a single nozzle. This change in fibre diameter is controlled digitally by combining the mass flow rate to the nozzle with variations in collector speed without affecting the applied voltage. The broadest range of fibre diameters was obtained by changing those parameters simultaneously during printing. The maximum placement accuracy might be obtained by keeping the collector speed slightly faster than the essential translation speed.
This enables the construction of sophisticated multimodal and multiphase scaffolds from medical-grade poly (-caprolactone) using a single nozzle in a single print. Controlling fibre diameter during printing brings up new design possibilities for precise scaffold creation in biological applications.
The concepts of additive manufacturing (AM) have been used in the construction of 3D scaffolds for tissue engineering (TE), including the regeneration of bone, cartilage and skin. AM is very useful for providing tailored therapy, and one facet of this is the creation of multiphasic scaffolds to mimic regional changes in tissue ultrastructure. In this context, multiphasic scaffolds are described as having diverse regional morphologies from one another. Multiphasic scaffolds have a wide range of applications in TE when combined with a method that closely mimics the native tissue ultrastructure.
Furthermore, bimodal/multimodal fibrous scaffolds provide a variety of size characteristics within each zone and often comprise tiny diameter fibres to enhance cell adherence while bigger fibres preserve the overall construct's structural integrity. The small diameter fraction is typically created using solution electrospinning, whilst the larger diameter fibres are created using a variety of techniques such as melt spinning, fused deposition modelling (FDM), or a different electrospinning configuration. Due to a more straightforward regulatory route to clinical translation, melt processing was chosen in this work.
Application Insights
The heart valves are highly flexible but durable, with complicated deformation properties including anisotropy, nonlinearity, and viscoelasticity that are only partially replicated in scaffolds for heart valve tissue engineering (HVTE). The structural characteristics and microarchitecture of the primary tissue constituents, particularly collagen fibres, define these biomechanical traits.
Melt electrowetting (MEW) is used to build functional scaffolds with highly regulated fibrous microarchitecture that imitate the wavy character of collagen fibres and their load-dependent recruitment. As established by quasistatic and dynamic mechanical characterization, scaffolds with well-specified serpentine designs recreate the J-shaped strain stiffening, anisotropic, and viscoelastic behaviour of natural heart valve leaflets. They also increase the development of human vascular smooth muscle cells sown directly or in fibrin encapsulation, as well as the deposition of valvular extracellular matrix components.
Three-dimensional printed hydrogel constructions with well-organized melt electrowritten (MEW) fibre-reinforcing scaffolds have been shown to be a potential restorative technique for treating minor cartilage lesions. Scientists are investigating ways to convert the manufacturing of tiny fibre-reinforced structures on flat surfaces into anatomically meaningful structures. Their work demonstrates that therapeutically important materials with low conductivities may be resurfaced with ordered MEW fibres. Importantly, precise patterning on non-flat surfaces was demonstrated.
Regenerative techniques based on biofabrication technology may be a viable option for repairing damaged articular cartilage tissue. Recent advancements in (micro) fibre production and deposition methods, such as melt electro writing (MEW) and extrusion-based bioink deposition, have enabled the fabrication of mechanically stable, fibre-reinforced cartilage implants.
Regional Insights
What is the U.S. Melt Electrowriting Technology Market Size?
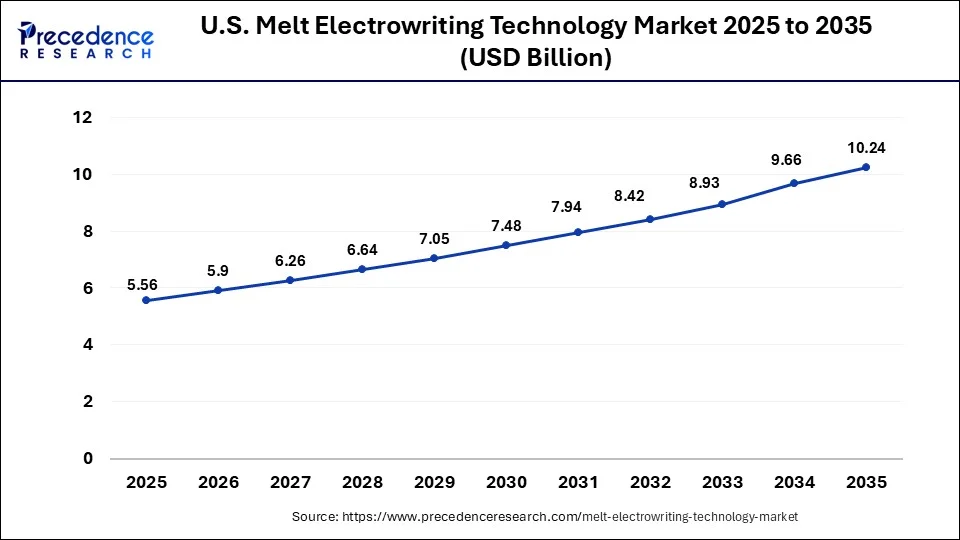
The U.S. melt electrowriting technology market size is estimated at USD 5.56 billion in 2025 and is expected to be worth around USD 10.24 billion by 2035, rising at a CAGR of 6.30% from 2026 to 2035.
Until recently, the area lagged behind North America and Europe in the use of Melt Electro writing Technology. However, increased interest from APAC manufacturing businesses, as well as the implementation of several government-led plans and policies, have assisted several nations in the area in developing a sustainable AM ecosystem.
Because of the massive government backing for the sector, China is likely the most powerful engine driving Melt Electro writing Technology development in Asia. In 2018, the Melt Electro writing Technology market in China was expected to be worth $1.8 billion. It is now the third-largest Melt Electro writing Technology market after the United States and Western Europe.
The medical industry has taken a leadership role in adopting and deploying Melt Electro writing Technology in Korea, owing largely to substantial government assistance.
Healthcare is the most important of the government's ten priority areas, as defined in the Melt Electro writing Technology plan. Furthermore, the government's pilot programs to "stimulate fresh demand" are mostly aimed at the healthcare business. This means that, from the government's perspective, healthcare Melt Electro writing Technology applications provide the quickest victories at the outset. By the end of 2016, the Ministry of Food and Drug Safety had formally registered 19 3D-printed medical items, principally orthopaedic implants, thanks to government funding.
Singapore has prioritized the development of the country's Melt Electro writing Technology business. Singapore's government launched the Research, Innovation, and Enterprise (RIE) 2020 strategy, which highlighted AM as a vital enabler to promote the growth and competitiveness of the national manufacturing industry. The creation of the National Additive Manufacturing Innovation Cluster (NAMIC) in 2015 was possibly the most significant government move to boost AM. NAMIC finds and develops AM start-ups and innovations, as well as kick-starts public-private partnerships.
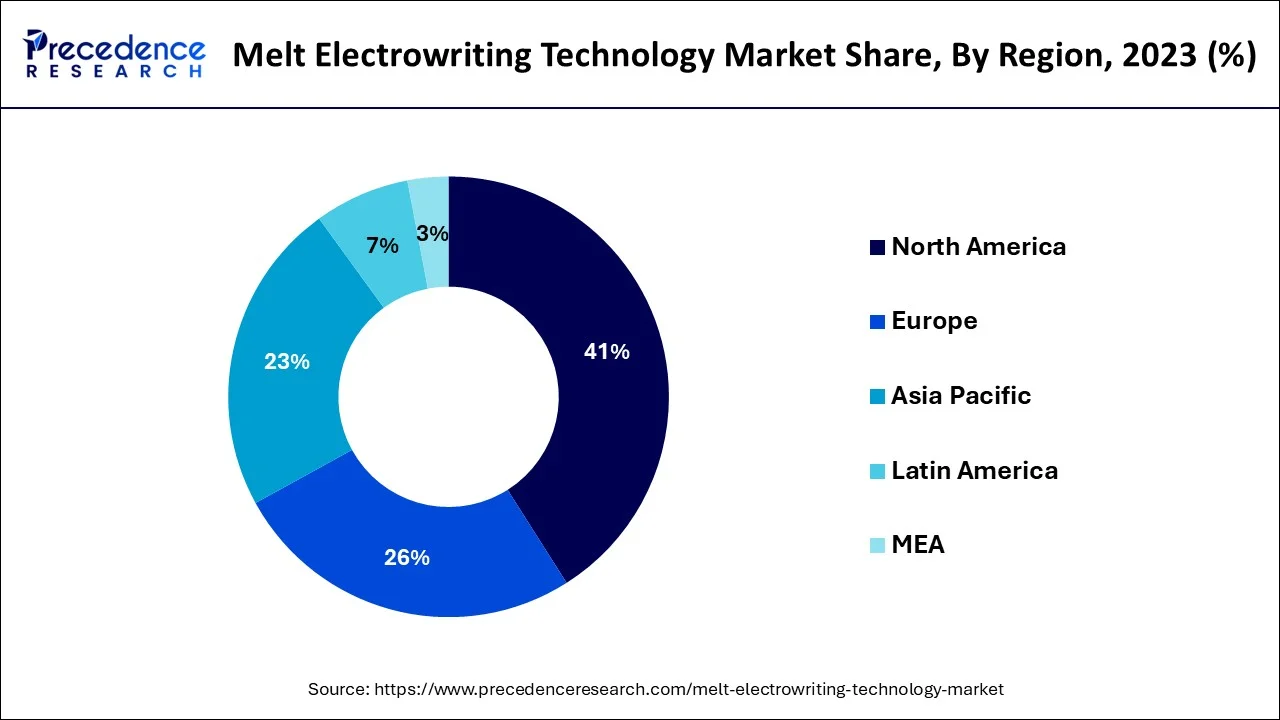
Asia Pacific
Asia Pacific is the region that is experiencing the highest rate of electronic writing technology Melton electronic writing technology growth due to the high rate of digitalization in the education sector, digitization initiatives by governments, and the increased use of hybrid work places. There is a high student population, price sensitive markets, and rising ed-tech investments and smart classrooms infrastructure in the region. The increased need of paperless records, interactive learning tools has increased its adoption both in the public and the privately operated sectors. The benefits of manufacturing and close access to electronics supply chains also reinforce regional competitiveness.
China is the leader of the region because of effective domestic production, intensive smart education programs, and a high access to digital devices in schools and businesses. India is a growing high-potential market, driven by a massive educational reform agenda, the growing startup scenes, and increasing adoption of the low-cost digital writing tools.
Latin America
The Latin America is an emerging yet highly prospects market of Melto electronic writing technology, which is backed by slow digitalization of the education systems and the government. There is still a problem with budget constraints, but the need to have more affordable digital substitutes to conventional writing instruments is on the rise.
Brazil has a huge education sector, is corporate-digitizing and is increasing ed-tech investments, making it dominate the regional market. Mexico comes in second with the advantage of cross-border technology transfer, increasing enterprise adoption, and digitization efforts by the government.
Middle East & Africa
The Middle East & Africa region has diverse but a changing demand of Melto electronic writing technology. The main drivers of adoption include government-led digital education reforms, city programs, as well as public service modernization. Gulf nations with a high-income level present high demand to the high-end electronic writing solutions, whereas portions of Africa are hindered by infrastructure constraints.
The Middle East market has been led by the United Arab Emirates and Saudi Arabia with the support of smart investment in education, digital government, and the use of high-quality technology. Israel has a hand to play in terms of innovation and niche application in design, engineering, and enterprise collaboration tools.
Market Value Chain Analysis
- Raw Material Sourcing: Raw material sourcing primarily involves electronic components such as sensors, display panels, microcontrollers, and stylus materials. Asia Pacific dominates sourcing due to established semiconductor and electronics ecosystems.
Key players: Stratasys Ltd - Branding and Packaging: Branding focuses on positioning Melto electronic writing technology as a productivity and sustainability enabler rather than just a digital accessory. Packaging trends emphasize minimalism, eco-friendly materials, and clear communication of use cases.
Key Players: Protolabs - Customer Support and Feedback: Customer support is a critical differentiator, particularly in emerging markets where digital adoption maturity varies.
Key Players: Velo3D Inc.
Melt Electrowriting Technology Market Companies
- Stratasys Ltd. (NASDAQ: SSYS): Polymer 3D printing leader, offering systems (FDM, PolyJet), software (GrabCAD), and solutions for prototyping to production in aerospace, auto, healthcare; crucial for broad industry adoption.
- Protolabs (NASDAQ: PRLB): Digital manufacturing service bureau, providing rapid prototyping (3D printing, CNC, injection molding) for designers and engineers; key for on-demand parts.
- Velo3D Inc. (NYSE: VLD): Specializes in metal additive manufacturing, known for high-quality, complex metal parts (especially for aerospace/energy) with unique printing technology.
- Desktop Metal Inc. (NYSE: DM): Focuses on metal 3D printing (binder jetting, DLP) for mass production, aiming to disrupt traditional manufacturing, bridging prototyping to industrial scale.
Other Major Key Players
- 3D Systems (NASDAQ: DDD)
- Xometry Inc. (NASDAQ: XMTR)
- Materialise N.V. ADR (NASDAQ: MTLS)
- Nano Dimension (NASDAQ: NNDM)
- Markforged Holding Corp. (NYSE: MKFG)
- Shapeways Holdings Inc.: (NYSE: SHPW)
- MeaTech 3D Ltd. (NASDAQ: MITC)
- Organovo Holding Inc. (NASDAQ: ONVO)
- Voxeljet AG ADR (NASDAQ: VJET)
- Final Thoughts
Segments Covered in the Report
By Type
- Scaffolds
- Poly
By Application
- Diarthrodial Joint Melt Electro writing
- Functionalized Titanate Nanofiller Melt Electro writing
- Coronary Artery Stents Melt Electro writing
- Controlled Drug Release Melt Electro writing
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa (MEA)
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
