What is the Biobetters Market Size?
The global biobetters market size is calculated at USD 71.51 billion in 2025 and is predicted to increase from USD 77.67 billion in 2026 to approximately USD 159.78 billion by 2035, expanding at a CAGR of 8.37% from 2026 to 2035.The biobetters market is driven by the longer half-life of the product.
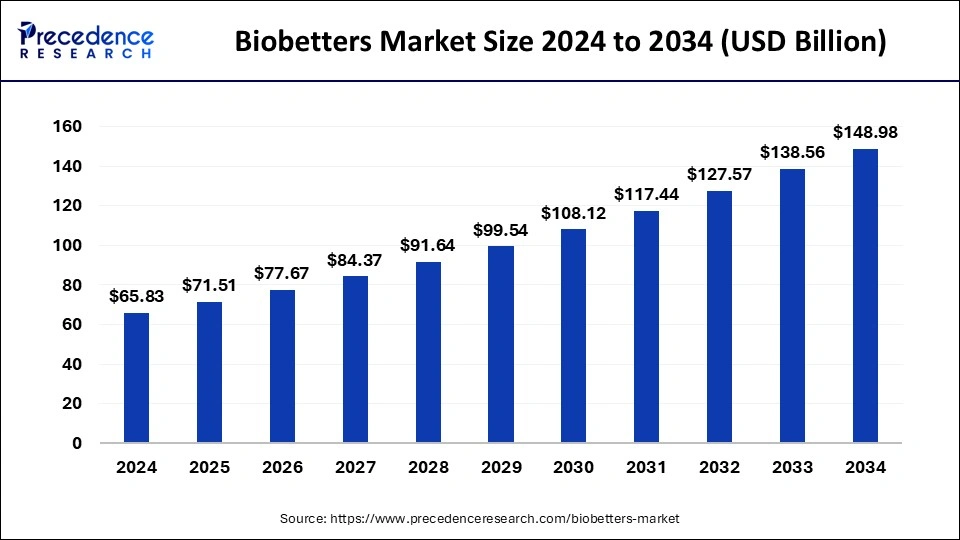
Biobetters Market Key Takeaways
- In terms of revenue, the global biobetters market was valued at USD 65.83 billion in 2025.
- It is projected to reach USD 148.98 billion by 2035.
- The market is expected to grow at a CAGR of 8.51% from 2026 to 2035.
- North America dominated the biobetters market in 2025.
- Asia- Pacific shows a significant growth in the biobetters market during the forecast period.
- By molecule type, the monoclonal antibodies biobetters segment dominated the market in 2025.
- By molecule type, the insulin biobetters segment is observed to be the fastest growing in the biobetters market during the forecast period.
- By disease indication, the cancer segment dominated the market in 2025.
- By disease indication, the neurological disorders segment shows a significant growth in the biobetters market during the forecast period.
- By distribution channel, the hospital pharmacy segment dominated the market.
- By distribution channel, the online pharmacy segment shows a notable growth in the biobetters market during the forecast period.
What is a Biobetters?
The biobetters market revolves around the biopharmaceutical industry segment that focuses on creating, manufacturing, and distributing biobetters. Biologic medications created from current biologics but altered to enhance their efficacy, safety, stability, or delivery are referred to as bio betters or biosuperiors. The creation of biobetters encourages innovation in the biopharmaceutical sector.
Businesses spend money on research and development to discover novel approaches to improving biologic medications currently on the market, advancing science and technology. By developing modified versions of already-existing biologics, businesses can prolong the lifecycle of their products beyond the original patent expiration. This allows them to retain market exclusivity and revenue streams for longer periods of time.
Key AI Integration in the Biobetters Market
AI and machine learning (ML) algorithms focus on the identification and optimization of molecular structures. Generative AI models can now propose novel protein designs or modifications (such as pegylation or glycoengineering) to existing biologics, dramatically decreasing the traditional trial-and-error approach. AI aggregates and determine data from sources such as electronic health records and wearable devices to offer a more nuanced understanding of a drug's effectiveness and even safety in a broader population, funding regulatory approval processes.
Biobetters Market Data and Statistics
- Celltrion Healthcare presented favorable safety and efficacy results on maintenance therapy at the European Crohn's and Colitis Organization Congress. This included information on Remsima SC, the company's infliximab biobetter that can be administered subcutaneously.
- In June 2023, the gene-editing company Jenthera Therapeutics and the biotech company Future Fields established a partnership to produce a novel protein that fights cancer. The Canadian businesses hope to mass-produce therapeutic recombinant proteins more sustainably, scalable, and economically by merging a novel drug delivery mechanism with a bioproduction platform.
What are the Growth Factors in the Biobetters Market?
- Biobetters offer benefits like increased efficacy, fewer adverse effects, and a longer half-life compared to current medications. This increases the need for these improved therapies in conditions such as diabetes, cancer, and hereditary illnesses.
- Advances in cell line generation, protein engineering, and manufacturing techniques enable the production of biobetters with enhanced properties and capabilities.
- Biopharmaceutical businesses perceive an opportunity to create and launch biobetters with possible cost advantages and improved therapeutic characteristics as the patents on current pharmaceuticals expire.
- Biobetters are getting more regulatory body approval, which is driving market expansion. This is due to their established advantages and expedited development procedure, in contrast to wholly novel medications.
- Biobetters fit with the expanding trend of personalized treatment since they may be customized to meet the needs of individual patients. This serves a broader spectrum of patients.
Biobetters Market Outlook
- Industry Growth Overview: It is driven by need for enhanced biologics with better efficacy, safety, and dosing for chronic diseases such as cancer & diabetes, with key regions such as North America and even fast-growing Asia-Pacific leading, as pharma firms invest heavily to gain market share from biosimilars along with original biologics by leveraging new tech such as PEGylation and fusion proteins.
- Major Investors: They are mainly large, established biopharmaceutical firms that conduct substantial in-house research and development, usually supplemented by strategic acquisitions and collaborations. Few companies are Amgen Inc., Merck & Co., Inc., F. Hoffmann-La Roche AG, Sanofi SA, Eli Lilly and Company
Market Scope
| Report Coverage | Details |
| Market Size by 2035 | USD 159.78 Billion |
| Market Size by 2026 | USD 77.67 Billion |
| Market Size in 2025 | USD 71.51 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 8.37% |
| Largest Market | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Molecule Type, Disease Indication, Distribution Channel, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Driver
Growing demand for biopharmaceuticals that offer improved efficacy, safety, and patient convenience
Biobetters are designed to offer higher therapeutic efficacy than original formulations. This enhancement could entail adjustments to lengthen the medication's half-life, improve target specificity, or improve dosage schedules. Biobetters are better than their predecessors at addressing unmet medical needs because they achieve better treatment outcomes. Biosimilars and biobetters are becoming increasingly competitive in the market as many biopharmaceutical patents are about to expire. Thereby, the growing demand for biopharmaceuticals with improved efficacy acts as a driver for the biobetters market. Biopharmaceutical businesses aim to provide a unique product offering and prolong product life cycles by developing biobetters that offer definite advantages over biosimilars in terms of convenience, safety, or efficacy.
Improved protein engineering techniques
With protein engineering, researchers can improve the effectiveness of biologic medications that are already on the market. Researchers can increase tissue penetration, prolong the half-life in circulation, and boost binding affinity to targets by fine-tuning molecular architectures. Because of these changes, biobetters exhibit better treatment effects than their predecessors. Pharmaceutical corporations are able to prolong the patent life of their current biologics by utilizing protein engineering to create biobetters. Because of their exclusivity, they can hold onto market share in the face of competition from biosimilars , which usually become accessible after the patent on the original biologic expires.
Restraint
Stringent regulatory requirements for approval of biobetters
Biobetters need to show that they are safer and more effective than the original reference biologic. A lot of preclinical and clinical data are required for this, frequently including comparison trials with the original medicine. Regulatory bodies require substantial proof to guarantee that alterations or enhancements do not jeopardize patient safety or treatment efficacy. To demonstrate their superiority or noteworthy clinical benefit over current medicines, regulatory agencies frequently mandate that biobetters go through clinical studies.
These trials require a lot of time, resources, and sometimes a huge number of patients. It can also be difficult to plan them in a way that shows incremental advantages over current treatments. This limits the growth of the biobetters market.
Opportunity
Developing a biobetter can provide a strategic advantage in terms of intellectual property (IP) and market exclusivity
A business can frequently acquire or renew new patents when it creates a biobetter. This is significant because, for a set amount of time (usually 20 years from the filing date), patents grant the exclusive right to manufacture and market the product, shielding it from rivalry. Enhancing efficacy, decreasing adverse effects, or improving delivery systems on an already-existing biopharmaceutical will allow the corporation to get extra patent protection. This extension lengthens the exclusive market period, giving the business additional time to recover its costs and make money without rivalry.
Segment Insights
[[segment_insights]]
Regional Insights
[[regional_insights]]
Value Chain Analysis of Biobetters Market
- Formulation and Final Dosage Preparation
In the biobetters market, formulation along with final dosage preparation focus on engineering superior drug products with enhanced clinical performance, stability, and even patient convenience. This process is distinct from biosimilars, which now match the original product's formulation and even dosage form to demonstrate similarity. - Packaging and Serialization
In the biobetters market, packaging as well as serialization follow stringent pharmaceutical regulations thus, similar to those for novel biologics, aiming on maintaining drug stability, ensuring patient safety, and allowing end-to-end supply chain traceability. The major difference from standard pharmaceuticals depends on the specific packaging material requirements because of the sensitivity of biologic products.
Biobetters Market Comppnies

- F. Hoffmann-La Roche AG: F. Hoffmann-La Roche (Roche) leads to the biobetter market primarily through improving its existing biologics (like Avastin) into enhanced versions (like Lucentis for eye conditions) and by supplying vital raw materials, like high-purity enzymes, for API production, targeting on better efficacy, decreased dosing, and addressing unmet needs in areas such as oncology, neuroscience, and autoimmune diseases, positioning itself as a major innovator alongside biosimilar developers.
- Eli Lily and Company: Eli Lilly is a remarkable player in the biobetters market, mainly through its next-generation diabetes and even obesity medications, which are improved versions of earlier biologics.
Other major Key players
- Merck & Co. Inc.
- SERVIER
- Biogen Inc.
- Teva Pharmaceutical Industries Ltd.
- Sanofi SA
- Porton Biopharma Limited
- Novo Nordisk A/S
- CSL Behring GmbH
Recent Developments
- In June 2024, As reported by Syngene International Ltd, a new platform for producing proteins has been launched. The platform combines Syngene's clone selection and development procedures with in-licensed cell line and transposon-based technology from the Swiss biotech services company ExcellGene, which should result in a notable increase in accuracy and efficiency. By speeding up improved protein manufacturing, the new platform shortens the time to market by facilitating faster preclinical, clinical, and commercial launches.
- In November 2023, the most recent addition to 3M's line of chromatographic clarifiers was revealed. Harvest RC Chromatographic Clarifier, model number BT500, is a 500 mL, single-use chromatographic clarifier for monoclonal antibodies, recombinant proteins, and biologics.
- In March 2022, ProteoGenix declared the XtenCHOTM Transient Expression System to be available. The novel patented mammalian cell-based expression host provides up to ten times greater yields with less hands-on time than previous options because of its improved metabolism and enhanced plasmid stability. The novel CHO host seeks to expedite early-phase drug screening and streamline the creation of recombinant proteins.
Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
