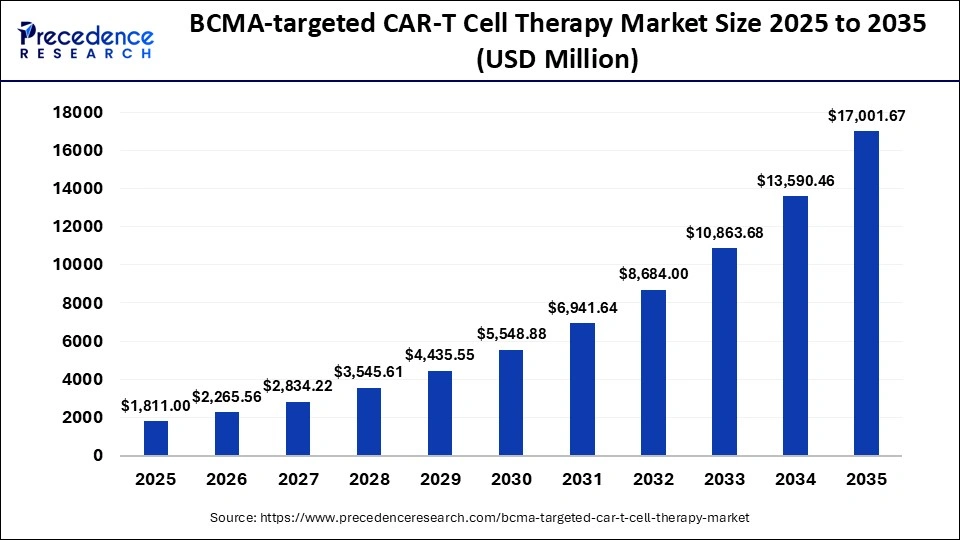
The BCMA-targeted CAR-T cell therapy market size was estimated at USD 1,811.00 million in 2025 and is predicted to increase from USD 2,265.56 million in 2026 to approximately USD 17,001.67 million by 2035, expanding at A CAGR of 25.1% from 2026 to 2035. The B-cell maturation antigen (BCMA) protein is targeted in this advanced immunotherapy, which includes idecabtagene vicleucel (Ide-cel) and ciltacabtagene autoleucel (Cilta-cel) approved therapies that show deep and durable responses.
BCMA-targeted CAR-T Cell Therapy Market Statical Scope
| Reports Attributes | Statistics |
| Market Size in 2025 | USD 1,811 Million |
| Market Size in 2026 | USD 2,265.56 Million |
| Market Size by 2035 | USD 17,001.67 Million |
| CAGR 2026 to 2035 | 25.1% |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
The BCMA-targeted CAR-T cell therapy is applied for patients with relapsed or refractory multiple myeloma and central nervous system involvement. The major growth drivers for the market's growth are clinical trial advancements, an increased incidence of multiple myeloma, and high response rates to CAR-T cell therapy .
The improved manufacturing has led to promising products like Abecma, the first approved BCMA CAR-T, and Carvykti, which shows better efficacy. North America led the market due to major clinical trials that advance BCMA CAR-T therapies. Hospitals are dominating with the highest revenue to meet complex inpatient care needs, including oncology clinics handling follow-ups.
The leading market players, like Bristol Myers Squibb and bluebird bio, have developed the first FDA-approved BCMA CAR-T named Abecma to treat patients who have dealt with at least four prior therapies. Similarly, Johnson & Johnson has manufactured the FDA-approved Carvykti as a fifth-line and then earlier-line treatment. These products have undergone manufacturing optimization to improve their access and received expanded approvals in Europe and other regions. A rapid manufacturing scale-up and enhanced efficacy have driven the huge adoption of these products.
The potential challenges faced by this market are manufacturing complexity, high costs, safety risks, and market access barriers. Researchers and medical professionals find variability in T-cell quality from pretreated patients, which reduces efficacy. The scalability issues also hinder goals to meet the growing consumer demand for medicinal products.
In some cases, the side effects of treatments arise in the form of cytokine release syndrome, neurotoxicity, and infections that occur in up to 90% of patients. These conditions require intensive monitoring and specialized centers. On the other hand, the long-term treatment concerns include secondary malignancies and lifelong surveillance.
BCMA-targeted CAR-T Cell Therapy Market Share, By Region, 2025 (%)
| Regions | Shares (%) |
| North America | 62% |
| Europe | 20% |
| Asia Pacific | 12% |
| Latin America | 4% |
| Middle East & Africa (MEA) | 2% |
-
North America - Advanced healthcare ecosystem with strong CAR-T adoption and regulatory support. Dominates with 62% share due to high treatment accessibility and presence of key market players.
-
Europe - Established healthcare systems with growing adoption of advanced therapies. Does not dominate with 20% share due to pricing pressures and slower approval processes.
-
Asia Pacific - Emerging market with increasing investment in advanced cancer therapies. Gaining momentum with 12% share as healthcare infrastructure and clinical research expand.
-
Latin America - Developing healthcare systems with limited CAR-T penetration. Does not dominate with 4% share due to high therapy costs and access constraints.
-
Middle East & Africa (MEA) - Nascent market with gradual adoption of advanced oncology treatments. Does not dominate with 2% share due to limited infrastructure and affordability challenges.
BCMA-targeted CAR-T Cell Therapy Market Share, By Type, 2025 (%)
| Segments | Shares (%) |
| Abecma | 48% |
| Carvykti | 42% |
| Others | 10% |
-
Abecma - A BCMA-targeted CAR-T therapy widely used for relapsed/refractory multiple myeloma. Dominates with 48% share due to earlier market entry and strong clinical adoption.
-
Carvykti - A next-generation BCMA CAR-T therapy with high efficacy outcomes. Gaining momentum with 42% share as superior response rates drive increasing physician preference.
-
Others - Includes emerging and pipeline BCMA-targeted CAR-T therapies. Does not dominate with 10% share due to limited approvals and early-stage commercialization.
BCMA-targeted CAR-T Cell Therapy Market Share, By Application, 2025 (%)
| Segments | Shares (%) |
| Hospital | 82% |
| Oncology Clinic | 18% |
-
Hospital - Major healthcare facilities providing advanced CAR-T administration and patient monitoring. Dominates with 82% share due to the need for specialized infrastructure and intensive care support.
-
Oncology Clinic - Specialized cancer treatment centers offering targeted therapies. Does not dominate with 18% share due to limited capability for complex CAR-T procedures.
| Type | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 | 2035 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Abecma | 869.28 | 1064.81 | 1303.74 | 1595.52 | 1906.85 | 2329.15 | 2773.81 | 3381.33 | 4118.16 | 4876.35 | 5925.96 |
| Carvykti | 760.62 | 974.19 | 1247.05 | 1595.52 | 2084.23 | 2661.89 | 3467.26 | 4421.74 | 5635.38 | 7179.07 | 8465.66 |
| Others | 181.10 | 226.56 | 283.42 | 354.56 | 443.45 | 554.56 | 693.45 | 867.01 | 1083.73 | 1490.00 | 2539.70 |
| Application | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 | 2035 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Hospital | 1485.02 | 1835.10 | 2267.37 | 2801.02 | 3458.94 | 4214.66 | 5200.89 | 6415.86 | 7911.21 | 9752.70 | 11851.92 |
| Oncology Clinic | 325.98 | 430.46 | 566.84 | 744.57 | 975.60 | 1330.94 | 1733.63 | 2254.22 | 2926.06 | 3792.72 | 5079.40 |
| Region | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 | 2035 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| North America | 1122.82 | 1359.34 | 1643.84 | 1985.53 | 2394.65 | 2883.71 | 3467.26 | 4161.64 | 5093.52 | 6230.89 | 7619.09 |
| Europe | 362.20 | 453.11 | 566.84 | 673.66 | 842.56 | 1053.66 | 1248.21 | 1560.61 | 1950.71 | 2438.18 | 3047.64 |
| Asia Pacific | 217.32 | 294.52 | 396.79 | 567.29 | 798.22 | 1109.12 | 1525.59 | 1994.12 | 2600.94 | 3386.36 | 4232.83 |
| Latin America | 72.44 | 90.62 | 113.37 | 141.82 | 221.73 | 277.28 | 346.73 | 520.20 | 650.24 | 812.73 | 1015.88 |
| MEA | 36.22 | 67.97 | 113.37 | 177.28 | 177.38 | 221.82 | 346.73 | 433.50 | 541.86 | 677.27 | 1015.88 |
Research Methodology
Related Databooks
March 2026
March 2026
March 2026
March 2026





 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Download Databook
Download Databook
 Schedule a Meeting
Schedule a Meeting