What is the Deep Brain Stimulation Devices Market Size?
The global deep brain stimulation devices market size is valued at USD 1.61 billion in 2025 and is predicted to increase from USD 1.78 billion in 2026 to approximately USD 4.26 billion by 2035, expanding at a CAGR of 10.22% from 2026 to 2035.
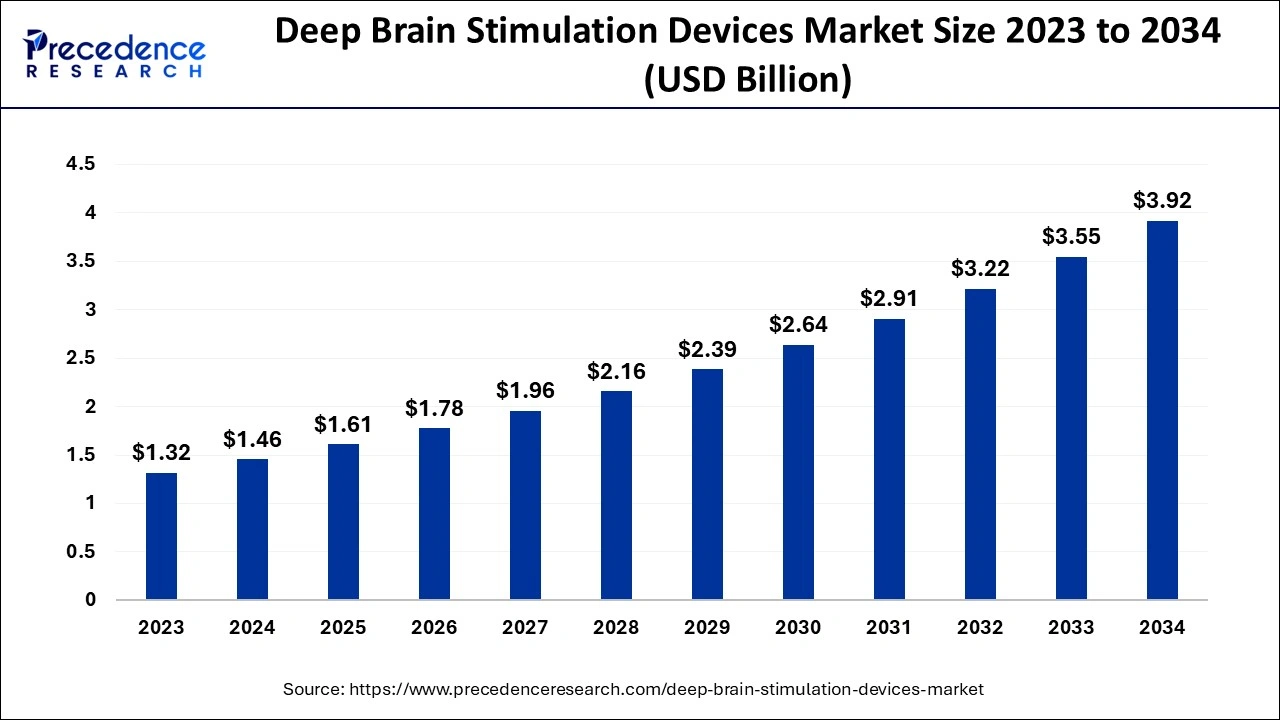
Deep Brain Stimulation Devices Market Key Takeaways
- The North America region has captured a market share of around 52% in 2025.
- By product, the dual-channel segment held a market share of around 58.2% in 2025.
- By application, the Parkinson's disease segment has dominated the market in 2025 with a revenue share of around 65.4%.
- By end-use, the hospital's segment accounted for 52.8% of the total market share in 2025.
Market Overview
A surgical method called deep brain stimulation involves implanting a device that sends electrical signals to the parts of the brain in charge of controlling movement. A DBS system is made up of three components that are implanted within the body of a patient: a lead, an extension, and a neurostimulator. The lead links the pacemaker device to the brain to synchronize electrical pulses. DBS devises aid in lowering the stiffness and tremor symptoms brought on by a number of neurological conditions, including Parkinson's disease, dystonia, or essential tremor. People can reduce their drug dosage after deep brain stimulation surgery, perhaps removing any negative effects.
Due to the rising occurrence of neurological illnesses, the deep brain stimulation systems market is anticipated to develop profitably throughout the forecasted period. One of the key reasons driving market expansion is the rising prevalence of Parkinson's disease across a number of continents. For example, the Parkinson's Foundation estimates that over 10 million individuals worldwide suffer from Parkinson's disease. Demand for DBS devices will also increase due to the increased prevalence of essential tremors and dystonia. As a result, the aforementioned factor will considerably boost the growth of the deep brain stimulation devices market during the course of the forecast year.
Additionally, neurological problems are more common in adults over the age of 65. Therefore, the incidence of neurological illnesses would be significantly stimulated by the rising rate of the senior population, expanding the market. Another important element driving the use of DBS procedures and consequently boosting market growth is the population's increased awareness of a variety of neurological conditions.
Deep Brain Stimulation Devices Market Growth Factors
During the ongoing coronavirus epidemic, telehealth and eHealth applications are becoming more and more popular for frequently monitoring PD patients. The global market for deep brain stimulation devices for Parkinson's disease is growing as a result of favorable reimbursement policies in healthcare institutions. However, implanted pulse generators for DBS devices require technical breakthroughs. Therefore, businesses in the Parkinson's disease market for deep brain stimulation devices should invest more in R&D to maximize battery life, along with patient-related factors like patient compliance with nightly device cut-off or IPG recharging dependability. For medical technology businesses to acquire a competitive edge in the worldwide deep brain stimulation devices market for Parkinson's disease, device-specific aspects including parameter delivery, lead configuration choice, implantation site, and cautious electrode material selection are critical.
Companies that make deep brain stimulation devices for Parkinson's disease are working harder to sell their products internationally. Other businesses involved in the DBS (deep brain stimulation) sector have products, but they are still in the clinical development stage and are not yet commercially accessible. Businesses are eager to release novel goods that are simple for patients and doctors to use. These businesses seek to increase the gadgets' portability, affordability, efficiency, and effectiveness.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.61 Billion |
| Market Size by 2035 | USD 4.26 Billion |
| Growth Rate from 2026 to 2035 | CAGR of 10.22% |
| Largest Market | North America |
| Fastest Growing Market | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered |
|
| Regions Covered |
|
Market Dynamics
Drivers
The rising desire for less invasive operations among the general public
- The rising desire for less invasive operations among the general public is one of the most important factors propelling the market for deep brain stimulators. DBS is a minimally invasive targeted procedure primarily used to treat dystonia, Parkinson's disease, and essential tremor movement disorders. Compared to conventional surgical methods, minimally invasive treatments provide a number of advantages, including shorter recovery times, smaller incisions, less discomfort, and decreased scarring. Therefore, these operations are highly sought-after due to a variety of patient advantages.
- In addition, there are some instances when less intrusive surgery shows a greater accuracy rate compared to conventional surgical techniques. Additionally, these techniques only need microscopic incisions big enough to introduce cameras and thin tubing. Due to the aforementioned advantages, there is a rising demand for such surgical treatments, which is driving market expansion.
Growth in the market will be affected by the unprecedented growth in the geriatric population
- One of the key factors boosting the market growth for deep brain stimulation devices is the rising senior population. At this age, people are more susceptible to chronic illnesses. Some of the most prevalent illnesses that can affect elderly persons are neurological disorders. Neuropathy, Parkinson's disease, Alzheimer's disease, dystonia, and other neurological conditions that commonly afflict elderly individuals include these.
- According to the National Institutes of Health (NIH), 617 million individuals, or 8.5% of the world's population, are 65 and older. Therefore, the market for DBS systems would see significant development during the period of 2018 to 2026 due to the expanding elderly population and increased frequency of neurological illnesses.
Key Market Challenges
- Concerns regarding side effects - Market expansion is anticipated to be hindered by worries about side effects including allergic response and tingling or prickling of the skin. Also anticipated to provide a barrier to the market for deep brain stimulation devices is the availability of alternative treatment alternatives such as pharmacological therapy and surgery.
Key Market Opportunities
The increasing geriatric population is propelling the market growth
- The main drivers of the market's expansion are the rapidly aging population, which is raising the risk and prevalence of neurological disorders, the rising demand for the devices due to the growing acceptance of minimally invasive procedures, and the availability of advanced deep brain stimulation devices. As a result of advancements in technology, deep brain stimulation technologies are predicted to become more widespread.
Deep Brain Stimulation Devices Market Segment Insights
[[segment_insights]]
Deep Brain Stimulation Devices Market Value Chain Analysis
- Device Processing
DBS devices are created through processes such as electrode fabrication, neurostimulator design, pulse generator programming, device miniaturization, and integration with wireless and rechargeable systems to treat neurological disorders.
Key players: Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, PINS Medical Co. Ltd - Quality Testing and Certification
DBS devices need certifications for medical safety, electromagnetic compatibility, biocompatibility, and regulatory approval. These include ISO 13485, FDA premarket approval (PMA), CE marking for medical devices, and IEC 60601 compliance.
Key players: FDA (U.S. Food and Drug Administration), ISO (International Organization for Standardization), CE Marking, UL Solutions. - Distribution to Industrial Users
DBS devices are supplied to hospitals, neurology clinics, specialized surgical centers, and medical device distributors that serve patients with Parkinson's disease, epilepsy, dystonia, and other movement disorders.
Key players: Medtronic plc, Abbott Laboratories, Boston Scientific Corporation.
Top Companies in the Deep Brain Stimulation Devices Market & Their Offerings:
- Medtronic plc
Medtronic is a global leader in deep brain stimulation (DBS) devices, providing implantable systems for movement disorders such as Parkinson's disease, essential tremor, and dystonia. Its devices include both rechargeable and non-rechargeable neurostimulators with advanced programming and MRI-compatible features. - Abbott Laboratories
Abbott offers DBS solutions through its Infinity™ DBS platform, focusing on precise therapy for neurological disorders. Its devices support patient-specific programming, wireless monitoring, and integration with digital health platforms to optimize treatment results. - Boston Scientific Corporation
Boston Scientific provides DBS systems with directional leads and advanced neurostimulators. Its solutions aim for better symptom control, energy efficiency, and flexibility in treating movement disorders, supported by comprehensive clinician training and programming software. - NeuroPace, Inc.
NeuroPace develops implantable neurostimulation devices targeting neurological disorders, with applications in managing movement disorders. The company emphasizes responsive and closed-loop stimulation technologies to enhance effectiveness and improve patient outcomes. - SceneRay Co., Ltd.
SceneRay specializes in neurostimulation devices, including DBS systems for Parkinson's disease and dystonia. The company creates affordable, locally produced DBS solutions aimed at expanding in the Asian markets with competitive prices and adherence to regulations.
Deep Brain Stimulation Devices Market Companies
- Abbott (St. Jude Medical)
- Medtronic
- Boston Scientific Corporation
- Aleva Neurotherapeutics S.A.
- Nexstim
- LivaNova PLC
- Neuropace Inc.
Recent Developments
-
In March 2025, Newronika is preparing for the commercial launch of its AlphaDBS™ system in select European markets, following CE Mark approval in March 2025. The company has not yet released the product for commercial use.
https://parkinsonsnewstoday.com -
In March 2025, the Medtronic BrainSense™ Adaptive DBS system was approved by the FDA as the first DBS technology that automatically adjusts stimulation therapy based on real-time brain signals. This innovation aims to personalize treatment for patients with Parkinson's disease.
https://www.news-medical.net
Deep Brain Stimulation DevicesMarket Segment Covered in Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
