What is the Outsourced Small-Molecule API Manufacturing Market Size?
The global outsourced small-molecule API manufacturing market is expanding as pharma companies rely on CDMOs for scalable, cost-efficient, and high-quality API production.The market is largely influenced by the increasing prevalence of chronic diseases, which has resulted in a huge demand for APIs, the growing shift of the pharma industry to outsource API production and commercialization while following strict safety guidelines, and governments' huge investment in R&D across leading sectors, helping the market grow exponentially.
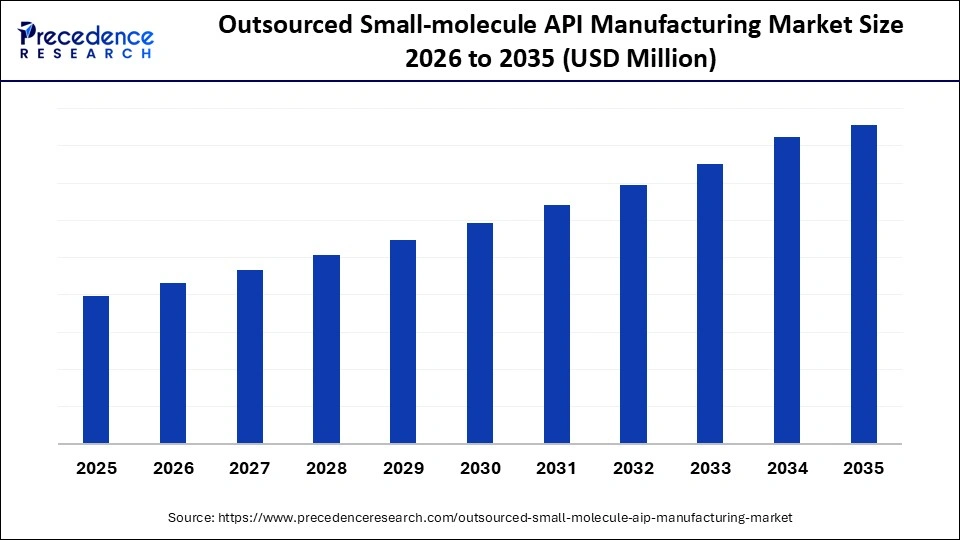
Market Highlights
- North America accounted for the largest market share of 42% in 2025.
- The Asia Pacific is expected to grow at the fastest CAGR of 9% from 2026 to 2035.
- By service type, the commercial scale API manufacturing segment contributed the biggest market share of 34% in 2025.
- By service type, the process development & optimization segment is growing at the fastest CAGR of 8.80% from 2026 to 2035.
- By synthesis type, the synthetic/chemical APIs segment held the highest market share of 68% in 2025.
- By synthesis type, the biocatalysis/Enzyme-mediated synthesis segment is expanding at a strong CAGR of 9% from 2026 to 2035.
- By therapeutic area, the oncology segment accounted for the major market share of 32% in 2025.
- By end user, the pharmaceutical companies segment recoded the highest market share of 56% in 2025.
- By end user, the virtual/emerging pharma companies' segment is expected to expand at the fastest CAGR of 9.20% from 2026 to 2035.
Outsourced Small-molecule API Manufacturing Market: Offerings & its Expansion
The market refers to the sector where pharmaceutical companies deal with third-party specialized companies that offer development and manufacturing of Active Pharmaceutical ingredients for drugs. These companies are popularly known as contract development and manufacturing organizations (CDMOs), provide cost-effective solutions with high-skilled expertise and help achieve scalable production without the need for substantial investment in building their own facilities.
Small and mid-size companies often invest in CDMOs due to a lack of their own facilities for developing APIs and their mass production. They mainly depend on outsourcing this work. In some cases, large firms like Pfizer and Novartis also outsourced their API production to focus on core operations and gain flexibility, which is a major driver of the market.
AI Shifts in Outsourced Small-Molecule API Manufacturing Market
The pharmaceutical industry is transforming due to digital integration and API manufacturing, where drug development can be accelerated using AI/ML technologies to optimize processes and achieve higher yield with quality, resulting in a competitive advantage. AI has evolved from being only a future concept to todays cutting-edge technology readily applicable to various domains.
AI can detect data loopholes that are hindering the manufacturing process, offer possible solutions, and even allow changes to operational processes in real time while optimizing batch scheduling to minimize downtime. It can further affect quality assurance by predicting deviations before they occur, using past data analysis to reduce potential product recalls and avoid human errors.
Outsourced Small-Molecule API Manufacturing Market Trends
- Integration of Advanced Technologies:Advanced technologies such as process analytical technology and automation are significantly influencing the small-molecule APIs manufacturing sector. PAT offers real-time manufacturing, while automation and digitalization streamline critical operations, mitigating errors that lead to higher-quality products.
- Growing Focus on Sustainability:Sustainability is a crucial aspect of many sectors, including outsourced small-molecule API manufacturing. Various enterprises are prioritizing eco-friendly manufacturing processes, energy efficiency, and proper waste disposal methods to avoid harming the environment.
- Quality Compliance:Stringent quality regulation is another trend the market is witnessing, aimed at delivering high-quality products and earning certifications from authorities. CDMOs are following strict regulations set by the US FDA, GMP, and the EMA for drug development and pharmaceutical product launches to reduce the risk of regulatory setbacks.
Market Scope
| Report Coverage | Details |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Service Type, Synthesis Type, Therapeutic Area, End User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Outsourced Small-Molecule API Manufacturing Market Segmental Insights
[[segment_insights]]
Outsourced Small-Molecule API Manufacturing Market Regional Insights
[[regional_insights]]
Outsourced Small-Molecule API Manufacturing Market Value Chain

Top Companies in Outsourced Small-molecule API Manufacturing Market & their Offerings
- Lonza
- Catalent, Inc.
- Siegfried AG
- Piramal Pharma Solutions
- Pfizer Inc
- 6S Pharma
- Merck KGaA
- Novartis
- Cambrex
- Johnson Matthey
- Teva
- Euroapi
- Boehringer Ingelheim GmbH
- Gland Pharma Ltd
Recent Developments
- In November 2025, A CDMO and out-licensing services will be launched by Grindeks Group as the company has found unused capacity at its manufacturing sites that will now be available for development, analytical, and GMP manufacturing as a support for APIs and developed dosage forms.
(Source: https://pharmasource.global ) - In October 2025, the U.S.-based CDMO specialty firm, Wilmington PharmaTech, collaborated with an LA-based private equity firm, Curewell Capital, to expand the manufacturing capacity of Wilmington PharmaTech.
Outsourced Small-Molecule API Manufacturing MarketSegments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content



 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
