Cardiac Resynchronization Therapy Market is Representing 7% Growth by 2030
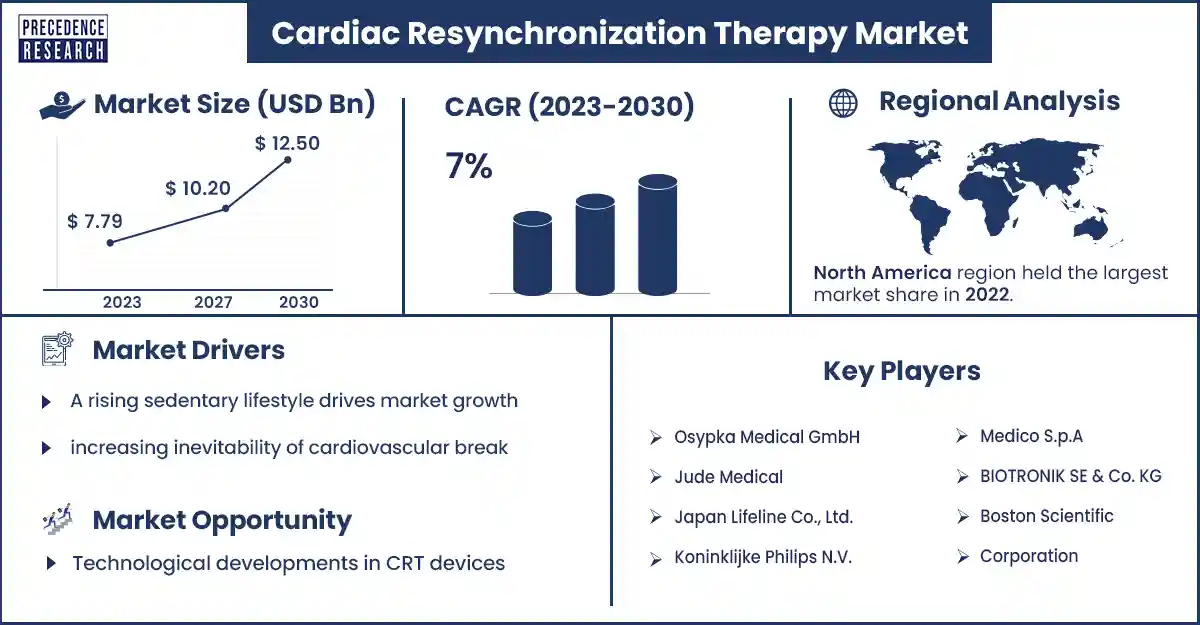
The global cardiac resynchronization therapy market size was exhibited at USD 7.79 billion in 2023 and is anticipated to touch around USD 12.50 billion by 2030, expanding at a CAGR of 7% from 2023 to 2030. The cardiac resynchronization market is divided into region, end-use and products.
Market Overview
Cardiac resynchronization therapy is a treatment for people who have heart failure and whose lower heart chambers don't contract in a coordinated fashion. The combination of cardiac resynchronization therapy devices with remote monitoring capacities allows for improved follow-up care and continuous patient monitoring. This feature drives patient care and contributes to the CRT market demand. The cardiac resynchronization therapy market is anticipated to grow significantly due to development activities and rising research for product development. The market growth is attributed to the growing advancements in the aging population and cardiac resynchronization therapy technologies. The increasing sedentary lifestyles and technological developments are the main factors enhancing CRT market growth.
A rising sedentary lifestyle drives market growth
Heart failure is a complex clinical syndrome that is rapidly increasing due to the rising sedentary lifestyle. Physical inactivity is a primary public concern as it is directly connected to the rising number of deaths. A sedentary lifestyle can lead to morbid obesity and so many other chronic diseases, such as diabetes and cardiac disorders. Extended durations of inactivity in otherwise physically active individuals have been generally found to increase symptoms of cardiovascular disease in atherosclerotic and diabetic patients.
Major outcomes of this sedentary lifestyle are cardiac disorders, such as angina, myocardial infarction, and heart stroke. Sedentary lifestyles are spreading all over the world due to the lack of increased penetration of video and television devices, as well as the lack of available space for exercise. Sedentary behaviors have negative impacts on the human body, including cardiovascular disease mortality, diabetes, cancer risks, hypertension, and many more. These factors of rising sedentary lifestyle are driving the growth of the cardiac resynchronization therapy market.
However, the lack of data format interoperability controls the growth of cardiac resynchronization therapy. High costs for cardiac resynchronization therapy and stringent regulations for cardiac resynchronization therapy devices are responsible for restraining the growth of the CRT market during the forecast period. Also, the increase in surgical site injection risk and unwanted healthcare reforms are among the major factors that restrain the growth of the CRT market. During the forecast period, these factors may further impact the CRT market growth. In addition, the cardiac resynchronization devices sector has been severely affected. These are the restraining factors that are negatively affecting the cardiac resynchronization therapy market.
Cardiac Resynchronization Therapy Market Report Scope
| Report Coverage | Details |
| Market Revenue in 2023 | USD 7.79 Billion |
| Projected Forecast Revenue by 2030 | USD 12.50 Billion |
| Growth Rate from 2023 to 2030 | CAGR of 7% |
| Largest Market | North America |
| Base Year | 2022 |
| Forecast Period | 2023 to 2030 |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Top Companies in the Cardiac Resynchronization Therapy Market
- Boston Scientific Corporation
- Abbott
- BIOTRONIK SE Co. KG
- Biotronik
- Medico S.p.A
- LivaNova PLC
- Koninklijke Philips N.V.
- Medtronic
- Japan Medical
- MicroPort Scientific Corporation
- Jude Medical
- Osypka Medical Gmbh
Recent Development by BIOTRONIK SE & Co.KG
- In July 2023, Biotronik SE & Co. KG announced Food and Drug administration approval in United States. This approval was related to its portfolio of Cardiac resynchronization Therapy and Amvia Edge pacemakers.
Regional Insights
The Asia Pacific region is estimated to grow fastest in the forecast period. This is caused by favorable financial circumstances, expanding medical infrastructure, a geriatric population, an increase in heart problems, and strategic moves made by huge industries. The adoption of innovative technological developments in these devices is accelerating in developed countries.
In China, cardiac resynchronization therapy is one of the best treatments for chronic heart failure. The progress of resynchronization technology, the rising abundance of evidence-based information, and indications of cardiac resynchronization therapy are continuously developing.
India is known for its advanced medical facilities and encouraging technology for cardiac resynchronization therapy. Most of the best hospitals for CRT are found in India, and the cost of CRT is very low compared to other countries.
North America dominated the cardiac resynchronization therapy market in 2023. Today, systolic heart failure is a main problem in America; it is the top cause of death in the U.S., according to the American Heart Association. While pharmacologic treatment has rapidly developed results in patients with systolic heart failure, hospitalizations from systolic heart failure continue to rise and remain a major cost load.
Market Potential and Growth Opportunity
Technological developments in CRT devices
Cardiac resynchronization therapies have been updated with various new features, such as smaller size and longer battery life. Manufacturers have been focusing more on the improvement and development of innovative cardiac resynchronization therapies. Several technological developments have contributed to achieving some of the objectives of advanced cardiac resynchronization therapy. They included novel lead design or the possibility of going beyond conventional delivery of cardiac resynchronization therapy.
Modern CRT technology is used to detect heart failure events instantly. The trial to measure the intra-cardiac barrier for the early detection of heart failure events is now ongoing, and the best result is expected. In addition, embedded devices like cardioMems can directly calculate the pressure inside the pulmonary artery, which they already developed and, shortly, will be available for clinical use. These new heart failure monitors will also have a significant role in the management of heart failure patients with cardiac resynchronization therapy devices in collaboration with the remote monitoring system. These opportunities may boost the growth and demand of the cardiac resynchronization therapy market in the coming years.
Cardiac Resynchronization Therapy Market News
- In July 2023, the U.S. Food and Drug Administration manufactured a Class 1 recall on the cardiac resynchronization therapy devices and Medtronic ICDs. With a glassed feedthrough, they might deliver low energy output when required for high-voltage therapy. To deliver cardiac resynchronization therapy, the problem is more likely to occur for CRT devices with glassed feedthrough configured.
- In May 2023, a pivotal clinic trial was introduced as a late-breaker at Heart Rhythm 2023. It indicated that advanced pacemaker technology could deliver cardiac resynchronization therapy between patients who were not to be medicated with epicardial ends and conventional cardiac resynchronization therapy.
- In July 2023, Abbott announced that the Drug and U.S. Food Administration had launched the AVEIR dual chamber pacemaker system. This was the first dual-chamber pacing system that treated patients who needed two chambers of the heart. Across the U.S., the approval significantly increases access to pacing for millions of people.
- In September 2023, in Pilot RCT, cardiac resynchronization therapy succeeded in pacing technology with the Purkinje conduction system. This therapy is for heart failure. Using this Purkinje conduction system pacing is feasible and safe and supplies a bigger boost to LV function.
Market Segmentation
By Type
- CRT-Pacemaker
- CRT-Defibrillator
By End User
- Cardiac Center
- Hospital
- Other End-users
Buy this Research Report@ https://www.precedenceresearch.com/checkout/1918
You can place an order or ask any questions, please feel free to contact at [email protected] | +1 650 460 3308