What is the Cardiac Rhythm Management Devices Market Size?
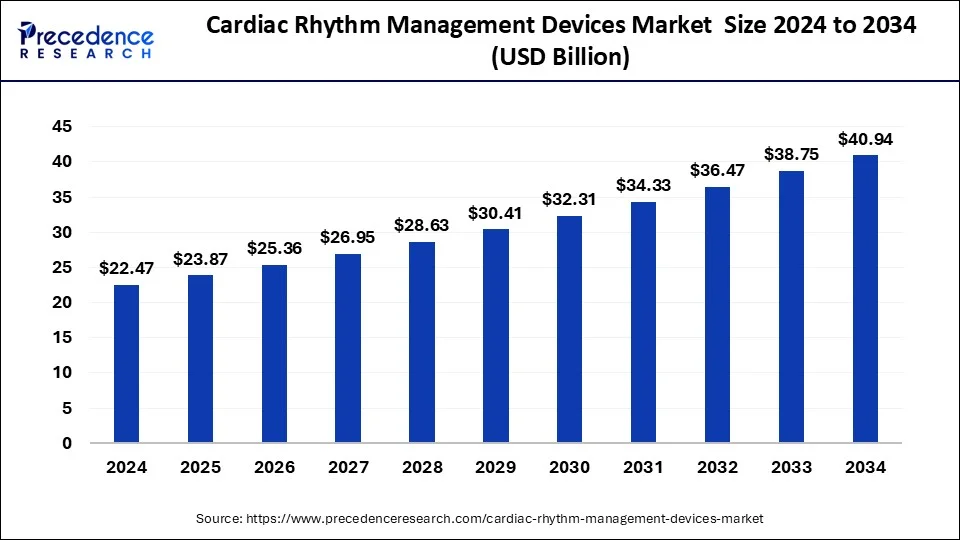
The global cardiac rhythm management devices market size was calculated at USD 23.87 billion in 2025 and is predicted to increase from USD 25.36 billion in 2026 to approximately USD 43.19 billion by 2035, expanding at a CAGR of 6.11%.
Cardiac Rhythm Management Devices Market Key Takeaways
- The global cardiac rhythm management devices market was valued at USD 23.87 billion in 2025.
- It is projected to reach USD 43.19 billion by 2035.
- The cardiac rhythm management devices market is expected to grow at a CAGR of 6.11% from 2026 to 2035.
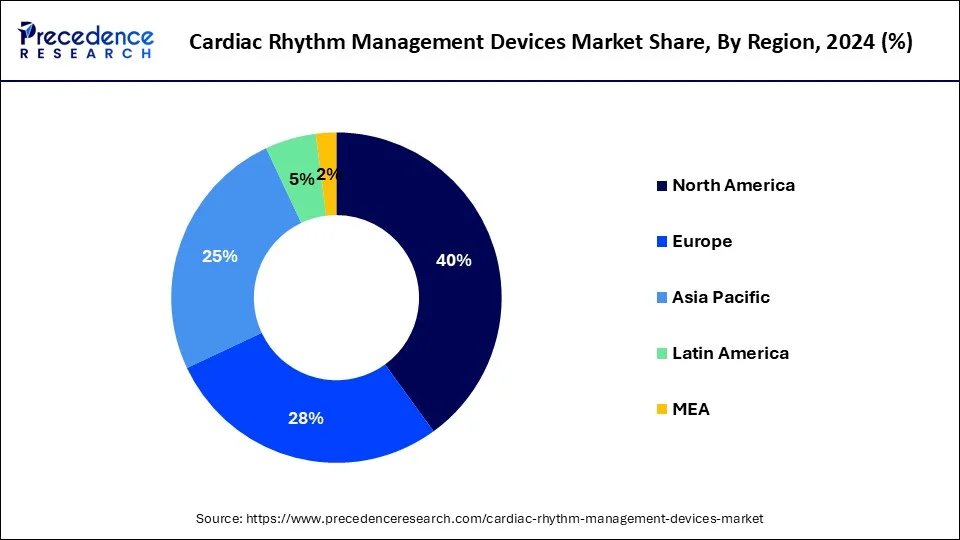
- North America has accounted more than 46% of the market share in 2025.
- By product, the defibrillators segment held the largest share of the market in 2025.
- By application, the arrhythmias segment held the largest share of the market in 2025.
- By end-user, the hospitals segment led the market with the largest market share of 50% in 2025.
Market Overview
The cardiac rhythm management devices market offers products, comprising pacemakers and implantable cardioverter defibrillators (ICDs), constitute integral tools in treating and managing arrhythmia-related conditions. These devices, along with associated leads and accessories, play a crucial role in regulating heart rhythm and improving overall patient health. Pacemakers administer electrical impulses to normalize heartbeats, while ICDs monitor heart activity and intervene when detecting abnormal rhythms, ensuring prompt restoration of a normal rhythm when necessary.
The cardiac rhythm management devices market is witnessing ongoing evolution, encompassing innovations such as cardiac resynchronization therapy and loop recorders. This evolution is facilitating broader usage and effectiveness, with general practitioners increasingly participating in patient follow-up and management. The numerous benefits offered by cardiac rhythm management devices, and concerns regarding potential complications necessitate open communication between patients and implanting physicians.
Post-implantation, patients can anticipate leading normal, active lives, with occasional caution warranted in specific situations, such as proximity to electromagnetic interference from appliances. As these devices continue to advance, they remain pivotal in enhancing patient outcomes and quality of life within the realm of cardiac care.
- In January 2022, Medtronic announced its acquisition of Affera, an innovative developer of cardiac rhythm solutions.
- In July 2023, BIOTRONIK received FDA approval for its next-generation family of pacemakers.
Cardiac Rhythm Management Devices Market Growth Factors
- Cardiac rhythm management devices perform a multitude of functions such as bradycardia pacing, arrhythmia monitoring, cardiac resynchronization for heart failure, defibrillation, and anti-tachycardia pacing for tachyarrhythmias. This broad spectrum of functionalities drives the growth of the cardiac rhythm management devices market as it addresses diverse patient needs and contributes to improved cardiac care outcomes.
- The market encompasses a range of cardiac rhythm management devices including pacemakers, implantable cardioverter defibrillators (ICDs), and loop recorders. Australia's noteworthy history in cardiac pacing adds to market growth, highlighting a culture of innovation and adoption within the country.
- Technologically advanced cardiac rhythm monitoring devices streamline diagnostic processes, benefiting both patients and healthcare providers. These innovations enhance accuracy, efficiency, and accessibility of cardiac care, fostering market expansion by improving diagnostic capabilities and patient outcomes.
- Advanced monitoring devices facilitate early detection and accurate diagnosis of cardiac events, minimizing the risk of incomplete resolutions or incorrect treatment plans. This capability to identify deviations from normal heart activity ensures timely intervention and optimal patient management, thereby driving market growth.
- Cardiac rhythm monitoring devices cater to diverse patient needs, ranging from Holter monitors to remote monitoring solutions. Real-time data analysis prompts healthcare professionals to provide timely medical attention, ensuring efficient patient care across various healthcare settings and specialties, thereby stimulating cardiac rhythm management devices market growth.
- These monitoring devices are complemented by dedicated patient record tools, facilitating seamless integration and accessibility of patient data for healthcare providers. This streamlined data management enhances diagnostic accuracy and treatment planning, contributing to market growth by improving operational efficiency and patient care quality.
- Patients benefit from access to their own cardiac rhythm data, fostering a sense of involvement and responsibility for their health. Real-time access to records promotes patient engagement and adherence to treatment plans, driving market growth by enhancing patient satisfaction and outcomes.
- Cardiac rhythm management devices offer increased efficiency, better survival rates, and economic advantages for both patients and healthcare facilities. The promise of improved outcomes and convenience encourages widespread adoption, fueling market growth and expanding the reach of cardiac care solutions.
Market Scope
| Report Coverage | Details |
| Growth Rate from 2026 to 2035 | CAGR of 6.11% |
| Market Size in 2025 | USD 23.87 Billion |
| Market Size by 2035 | USD 43.19 Billion |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Application, and End-use |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Utilizing clinical magnets for cardiac rhythm management devices
The increasing indications for permanent pacemaker and implantable cardioverter defibrillator (ICD) implantation have led to a rise in patients with cardiac rhythm management devices (CRMDs). However, these devices may occasionally exhibit inappropriate responses to electromagnetic interference or lead noise over-sensing, potentially jeopardizing patient care. Temporary reprogramming of CRMDs using device programmers can mitigate these issues but requires technically qualified personnel and device-specific expertise, leading to delayed care and resource utilization.
By enhancing understanding and promoting the effective use of clinical magnets, healthcare providers can optimize patient care, minimize device-related complications, and contribute to the growth of the cardiac rhythm management devices market. Alternatively, clinical magnets offer a solution by enabling the modification of pacing modes in pacemakers and suspending tachycardia therapies in ICDs when appropriately positioned over the device site. By enhancing understanding and promoting the effective use of clinical magnets, healthcare providers can optimize patient care, minimize device-related complications, and contribute to the growth of the market.
Advancing cardiac rhythm management
Cardiovascular implantable electronic devices (CIEDs) play a vital role in managing patients with brady- and tachyarrhythmias or heart failure, complementing optimal pharmacotherapy. However, despite technological strides, gaps persist in CIED patient management, notably concerning the rising incidence of lead- and pocket-related long-term complications, including cardiac device–related infective endocarditis, demanding meticulous attention. Comprehensive patient care mandates remote monitoring of those with CIEDs, ensuring proactive intervention and holistic management.
Embracing innovation, novel technologies integrated into smartwatches offer a convenient avenue for long-term atrial fibrillation (AF) screening, particularly in high-risk populations. Timely detection of AF holds the potential to mitigate stroke risk and other AF-related complications, underscoring its pivotal role in fostering growth within the cardiac rhythm management devices market.
Restraint
High cost
The development of cardiac rhythm management devices market has been accompanied by increased costs, particularly evident in the median implantation cost of cardiac resynchronization therapy (CRT) devices over time. Device cost stands as a critical input parameter, intricately linked with device longevity. Notably, costs associated with CRT-defibrillator (CRT-D) therapy surpass those of CRT-pacemaker (CRT-P) therapy significantly. This disparity in costs between CRT-D and CRT-P therapies poses a constraint on market growth within the cardiac rhythm sector. As higher costs potentially limit accessibility and adoption rates, addressing this discrepancy becomes paramount in fostering sustained market expansion and ensuring broader patient access to advanced cardiac rhythm management technologies.
Opportunity
Embracing technological advancements
Cardiac rhythm management (CRM) devices are not just confined to monitoring and managing cardiac issues; they are evolving to encompass restoration, repair, and healing functions. As the medical industry and healthcare sector move towards complete digitization, there lies a significant opportunity to capitalize on the demand for efficient, near real-time data reporting technologies offered by cardiac rhythm devices. By embracing these innovative solutions, hospitals can enhance patient care, streamline workflows for healthcare providers, and ultimately optimize operational efficiency.
The increasing demand for such advanced monitoring devices presents a lucrative opportunity for investment. Acting swiftly to integrate these technologies into healthcare practices can position institutions at the forefront of cardiac care delivery, ensuring they remain competitive in an evolving market landscape. Thereby, the factor of technological advancements is observed to offer a lucrative opportunity to the cardiac rhythm management devices market.
Segment Insights
Product Insights
In the dynamic landscape of the cardiac rhythm management devices market, the defibrillators segment stood as a dominant force in 2025. Defibrillators play a pivotal role in restoring normal heart rhythm by administering an electric charge or current to the heart. In cases of sudden cardiac arrest (SCA), where the heart rhythm ceases, prompt intervention with CPR (cardiopulmonary resuscitation) and a defibrillator is critical for survival. Implantable or wearable defibrillators further enhance patient care by addressing dangerous arrhythmias, effectively correcting irregular heart rates or rhythms.
These devices offer a lifeline to individuals at risk of cardiac events, providing reassurance and potentially life-saving intervention. The widespread presence of automated external defibrillators (AEDs) in public spaces underscores the importance of accessibility and immediate response in emergency situations. AEDs empower bystanders to intervene effectively in cases of SCA, significantly improving the chances of survival. As technological advancements continue to drive innovation within the cardiac rhythm market, defibrillators remain at the forefront, offering versatile solutions to address cardiac emergencies and save lives.
Application Insights
The arrhythmias segment held the largest share of the cardiac rhythm management devices market in 2025. The aging population is more susceptible to cardiac arrhythmias due to age-related changes in the heart's electrical conduction system. With the global population aging rapidly, the prevalence of arrhythmias is expected to rise, driving the demand for cardiac rhythm management devices that can effectively address the unique needs of elderly patients. Continuous advancements in cardiac rhythm management technologies have led to the development of sophisticated devices with enhanced capabilities for diagnosing and treating arrhythmias. These devices offer improved accuracy, reliability, and functionality, enabling clinicians to tailor treatment strategies based on individual patient needs and preferences.
End-use Insights
The hospitals segment led the cardiac rhythm management devices market share of 50% in 2025. The evident requirement of health monitoring devices in hospitals create a significant growth factor for the segment to grow. The rapid adoption of devices with advanced technologies in hospitals along with the number of admitted patients post-surgery in such environments drives the growth of the segment.
The ongoing penetration of advanced wireless cardiac rhythm management devices create a large consumer-base at hospitals. This is observed to be another growth factor for the segment to expand in the upcoming period. Admitted patients post-surgery require continuous monitoring, cardiac rhythm management devices carry the capability to transfer the required data to hospital data storage.
Regional Insights
What is the U.S. Cardiac Rhythm Management Devices Market Size?
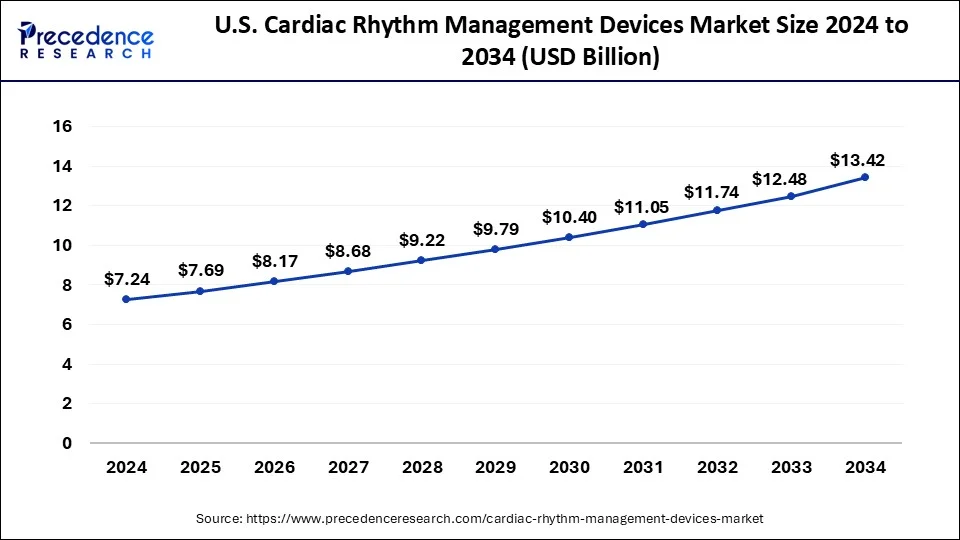
The U.S. cardiac rhythm management devices market size was exhibited at USD 7.69 billion in 2025 and is projected to be worth around USD 14.23 billion by 2035, growing at a CAGR of 6.35%.
North America led the market with the biggest market share of 46% in 2025, leveraging advanced technologies and widespread adoption across various healthcare settings. However, within the realm of cardiac rhythm management, specific considerations and procedures take precedence, driving the market's dynamics in the region.
Adaptive-rate devices, capable of sensing vibration, impedance changes, or the QT interval, present advancements in pacing technology. Nevertheless, concerns regarding mechanical or physiological interference leading to inappropriate high-rate pacing have surfaced. As a precautionary measure, it is recommended to disable adaptive-rate pacing, even in the absence of electrocautery usage during surgical procedures.
- In the United States, a staggering total of 777,173 Cardiac Rhythm Management (CRM) Procedures were conducted in 2022. These procedures are typically performed by interventional cardiologists and electrophysiologists, who implant devices within patients' bodies to regulate cardiac rhythm using electrical pulses.
Key segments within the United States cardiac rhythm management devices market include Cardiac Resynchronization Therapy (CRT), Implantable Cardioverter Defibrillator (ICD), Implantable Loop Recorder Procedures, and Pacemaker Implant Procedures.
North America's prominence in the cardiac rhythm management devices market underscores its commitment to advancing cardiovascular care and ensuring optimal patient outcomes. With a focus on innovation, collaboration between healthcare professionals, and technological advancements, the region continues to lead the way in shaping the future of cardiac rhythm management.
- In November 2023, Medtronic unveiled the United States debut of the Penditure left atrial appendage (LAA) exclusion system. This system features an implantable clip preloaded on a single-use delivery system, designed for LAA management during concomitant cardiac surgery procedures.
- In April 2022, Abbott disclosed that the U.S. Food and Drug Administration (FDA) had granted approval for the Aveir single-chamber (VR) leadless pacemaker. This approval allows for the treatment of patients in the U.S. with slow heart rhythms.
How is Asia-Pacific Growing in the Cardiac Rhythm Management Devices Market?
Asia-Pacific is expected to experience the fastest growth during the predicted timeframe. The market is driven by the rising prevalence of cardiovascular disorders, the burgeoning medical device sector, and the rapidly expanding healthcare sector. The rising adoption of advanced technologies and favorable reimbursement policies potentiate the adoption of cardiac rhythm management devices.
China Market Trends
China leads the market in Asia-Pacific, due to improved access to healthcare services, a higher incidence of cardiovascular disease, and investments in medical device manufacturing. Government initiatives promoting domestic production boosted supply capabilities. Expansion of hospital infrastructure enhanced treatment availability. Rising awareness and early diagnosis programs contributed to increasing demand for cardiac implant devices.
European Rapidly Growing Cardiac Rhythm Management Devices Industry
Europe is expected to witness significant growth in the market. This growth is due to the increasing prevalence of cardiovascular diseases and the rapidly rising geriatric population. These new devices offer improved accuracy and efficiency in monitoring and treating cardiac rhythm disorders. Technological advancements and increasing investments in research and development are also gaining traction, driving market growth further.
Germany Cardiac Rhythm Management Devices Market Trends: The country's growth is driven by rapid technological advancements, which have led to the development of more effective and safe devices. Technological advancements have enabled the development of more advanced devices that offer better monitoring, diagnostic capabilities, and treatment options for patients.
Latin America's Growing Cardiac Rhythm Management Devices Industry
Latin America is expected to witness substantial growth in the upcoming years. This growth is driven by increasing cardiovascular disease prevalence, rising patient awareness, and rapidly expanding access to advanced cardiac care. Supportive government initiatives, improving healthcare infrastructure, and investments by global players are further accelerating market expansion, especially in countries like Brazil and Mexico.
Brazil Cardiac Rhythm Management Devices Market Trends: Brazil's growth in the rhythm management devices industry is driven by increasing prevalence of cardiovascular diseases, rising patient awareness, and expanding access to advanced cardiac care facilities.
MEA's Growing Cardiac Rhythm Management Devices Industry
The Middle East and Africa are expected to witness steady growth in the market due to the rising prevalence of cardiovascular diseases, increasing patient awareness, and the expansion of cardiac screening programs. The market is also witnessing the high adoption of advanced devices such as leadless pacemakers and subcutaneous ICDs, thus accelerating demand. Investments in healthcare infrastructure, clinician training, and government initiatives supporting early detection and treatment are further driving market growth, especially in countries such as Saudi Arabia, the UAE, and South Africa.
Saudi Arabia Cardiac Rhythm Management Devices Market Trends
The Saudi Arabian market is expanding due to the rising prevalence of cardiovascular diseases and an aging population, which is increasing demand for pacemakers, ICDs, and CRT systems. Healthcare infrastructure upgrades, heightened public awareness of heart health, and increased expenditure, especially in major cities like Riyadh, Jeddah, and Dammam, are boosting the adoption of advanced CRM technologies.
Trade Analysis of the Cardiac Rhythm Management Devices Market
- The global trade of pacemakers for stimulating the heart muscles reached $7.45 billion in 2024, representing 7.77% increase from 2023.
- Over the past 5 years, the global trade of pacemakers has witnessed an average annual rise of 6.13%.
- Switzerland, Ireland, and the Netherlands were the top three global exporters of pacemakers for stimulating the heart muscles, with Switzerland exporting $1.79 billion of pacemakers.
- Top importers include the U.S. with $1.85 billion, the Netherlands with $1.05 billion, and Belgium with $547 million in imports.
Cardiac Rhythm Management Devices Market Companies
- Physio-Control, Inc.: Provides defibrillators, automated external defibrillators (AEDs), and emergency response systems for cardiac arrest management and pre-hospital care solutions globally.
BIOTRONIK: Offers pacemakers, ICDs, CRT devices, and remote monitoring solutions with advanced sensor technology for personalised cardiac rhythm management. - Schiller: Delivers ECG systems, defibrillators, stress testing devices, and monitoring solutions supporting diagnosis and management of cardiac rhythm disorders.
- Medtronic: Provides a comprehensive CRM portfolio, including pacemakers, ICDs, CRT devices, and remote cardiac monitoring platforms with AI-enabled diagnostics.
- Abbott: Offers innovative pacemakers, ICDs, insertable cardiac monitors, and remote monitoring systems focused on improving arrhythmia detection and patient outcomes.
- Koninklijke Philips N.V.: Provides cardiac monitoring systems, wearable biosensors, and integrated healthcare solutions for real-time cardiac rhythm tracking and clinical decision support.
- Zoll Medical Corporation: Supplies defibrillators, wearable cardiac monitors, and life-support technologies for emergency care, resuscitation, and continuous cardiac rhythm monitoring applications.
Other Major Key Players
- Physio-Control, Inc. (Stryker)
- Schiller
- Boston Scientific Corporation
- Progetti Srl
- LivaNova Plc
Recent Developments
- In April 2025, Merit Medical Systems announced the U.S. commercial launch of its Ventrax™ Delivery System, the latest addition to its electrophysiology and cardiac rhythm management portfolio. The system is designed to facilitate ablation procedures for treating ventricular tachycardia (VT), a condition contributing to approximately 75% of sudden cardiac deaths in the US. (Source: https://www.stocktitan.net)
- In April 2025, HeartBeam, Inc. announced a strategic collaboration with AccurKardia, an innovator in ECG-based diagnostics technology, bringing together two innovation leaders in ambulatory electrocardiogram (ECG) recording and analysis. The initial collaboration aims to enhance the accessibility of cardiac monitoring solutions and will focus on making AccurKardia's FDA-cleared ECG analysis software, AccurECG™, available on HeartBeam's devices. (Source: https://www.businesswire.com)
- In February 2025, MicroPort CRM, MedTech Zone (AMTZ), and Purple MicroPort signed a Memorandum of Understanding (MoU) to drive the expansion of MicroPort CRM's pacemaker business in India and its neighboring countries. This strategic partnership aims to address the growing need for advanced Cardiac Rhythm Management (CRM) devices in the rapidly expanding cardiovascular markets of India and the surrounding regions. (Source: https://microport.com)
- In January 2024, Medtronic and Cardiac Design Labs collaborated to launch heart rhythm monitoring technology Padma Rhythms, an external loop recorder (ELR) patch designed for comprehensive, long-term heart monitoring and diagnosis. “We are excited to embark on this transformative journey with CDL to reshape cardiac rhythm monitoring in India,” said Michael Blackwell, vice president and managing director of Medtronic India. (Source: https://www.financialexpress.com)
- In November 2024, Abbott, the global healthcare company, announced the launch of AVEIR VR single-chamber ventricular leadless pacemaker for the treatment of patients in India with slow heart rhythms. This pacemaker is now approved by the Central Drugs Standard Control Organization (CDSCO) in India. It is also U.S. Food and Drug Administration (FDA) approved and marks a significant advancement for patient care, bringing new features to patients and their physicians. (Source: http://financialexpress.com)
- In June 2023, Philips and BIOTRONIK formed a strategic alliance to expand care for out-of-hospital cardiology labs.
- In October 2023, MicroPort CRM launched the ULYS ICD and INVICTA Defibrillation Lead in Japan.
- In October 2023, Boston Scientific introduced the LUX-Dx II+ Insertable Cardiac Monitor System.
Segments Covered in the Report
By Product
- Pacemakers
- Implantable
- External
- Defibrillators
- Implantable Cardioverter Defibrillators (ICD)
- S-ICD
- T-ICD
- Implantable Cardioverter Defibrillators (ICD)
- External Defibrillator
- Manual External Defibrillator
- Automatic External Defibrillator
- Wearable Cardioverter Defibrillator
- Cardiac Resynchronization Therapy (CRT)
- CRT-Defibrillator
- CRT-Pacemakers
By Application
- Congestive Heart Failure
- Arrhythmias
- Bradycardia
- Tachycardia
- Others
By End-use
- Hospitals
- Cardiac care centers
- Ambulatory surgical centers
- Others
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
