What is the Cell and Gene Therapy Market Size?
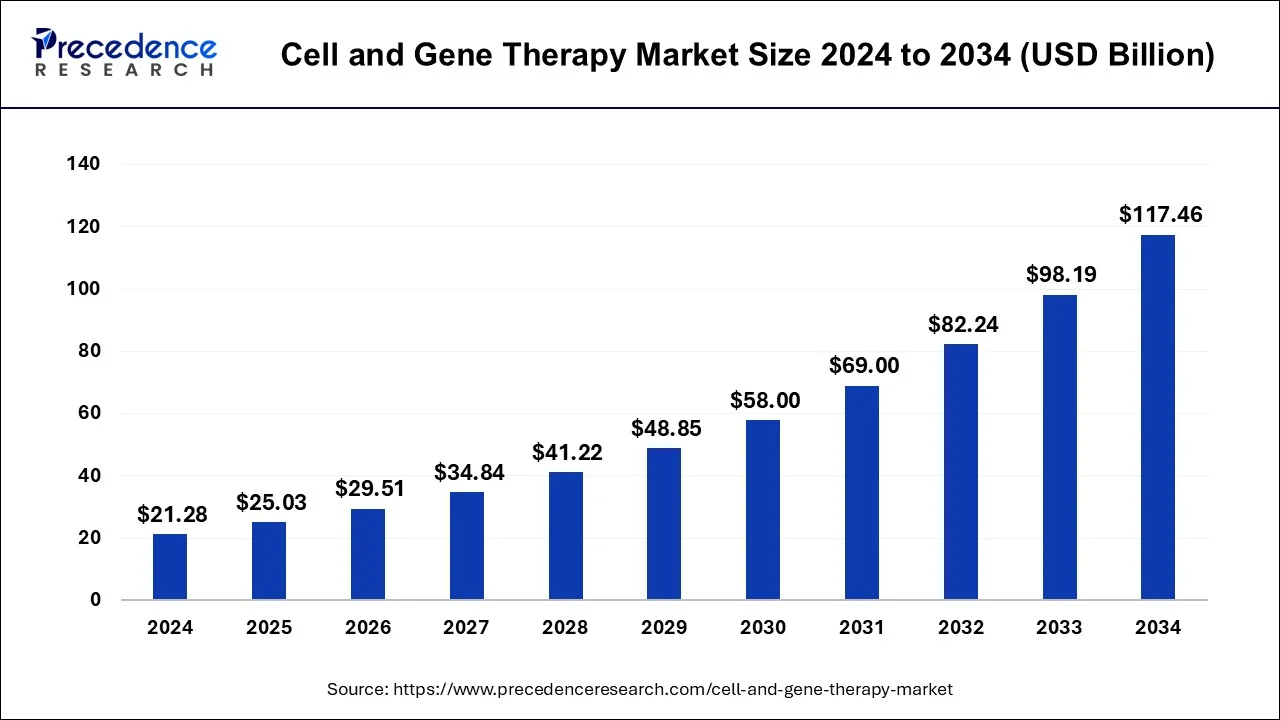
The global cell and gene therapy market size was estimated at USD 8.94 billion in 2025 and is predicted to increase from USD 10.44billion in 2026 to approximately USD 47.18 billion by 2035, expanding at a CAGR of 18.1% from 2026 to 2035.
Cell and Gene Therapy Market Key Takeaways
- In terms of revenue, the global cell and gene therapy market was valued at USD 8.94billion in 2025.
- It is projected to reach USD 45.24billion by 2035.
- The market is expected to grow at a CAGR of 17.60% from 2026 to 2035.
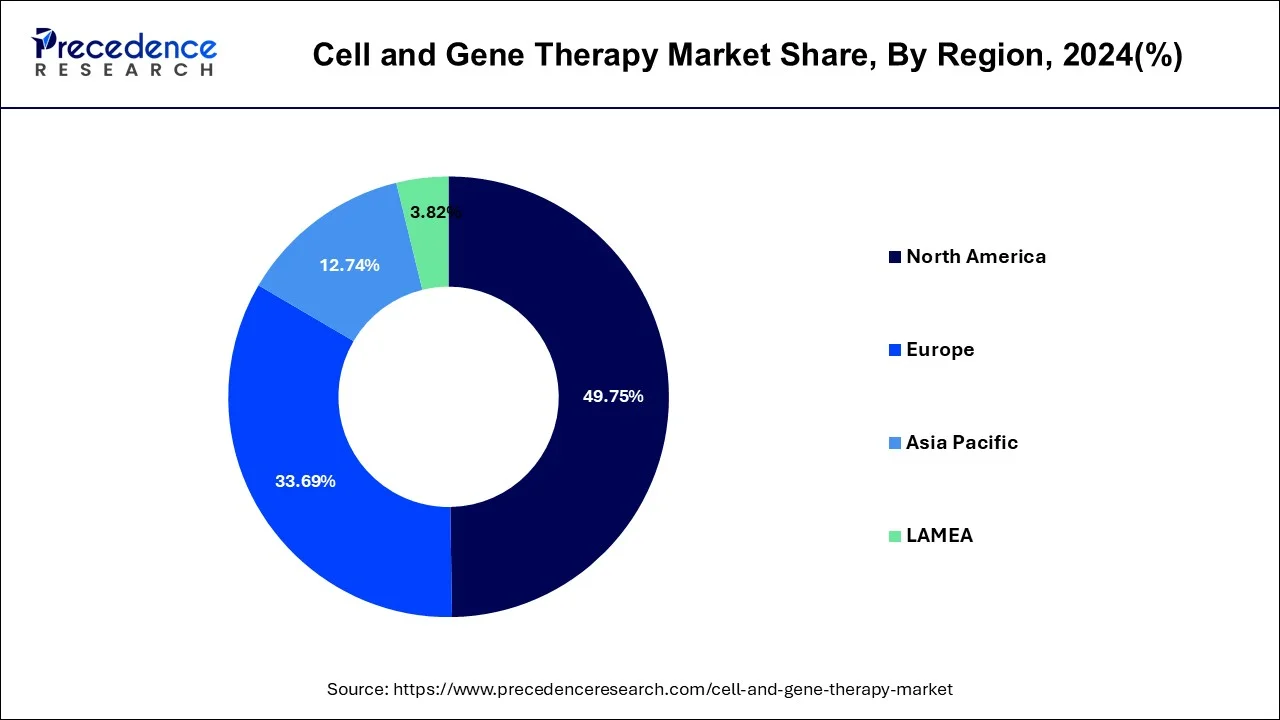
- North America region generated a revenue share of around 50.26% in 2025.
- By therapy type, the cell therapy segment led the market with a 65% share in 2025.
- By therapy type, the gene therapy segment held the second largest share of 35% in the market in 2025.
- By indication, the oncology segment led the market with a 39% share in 2025.
- By indication, the musculoskeletal/orthopedic disorders segment held the second largest share of 21% in the market in 2025.
- By vector type, the viral vectors segment led the market with a 72% share in 2025.
- By vector type, the non-viral vectors segment held the second largest share of 28% in the market in 2025.
- By manufacturing type, the in-house segment led the market with a 46% share in 2025.
- By manufacturing type, the contract development & manufacturing organizations (CDMOs) segment held the second largest share of 39% in the market in 2025.
- By end use, the hospitals & specialty clinics segment led the market with a 63% share in 2025.
- By end use, the cancer treatment centers segment held the second largest share of 24% in the market in 2025.
What is Cell and Gene Therapy Manufacturing?
Cell and gene therapies (CGTs) provide substantial advancements in patient care by treating or perhaps curing a variety of illnesses that have previously been untreatable by small molecule and biological medicines. The FDA in the United States has approved more than 20 CGTs during the past 20 years, and many of these one-time therapies range in price from US$37,500 to US$2 million per shot. Given the high financial outlay and patient expectations of these life-saving pharmaceuticals, it is essential that manufacturers provide integrated services across the whole supply chain to enable efficient biomanufacturing processes and smooth logistics and minimize uptake barriers.
High-profile mergers and acquisitions, such as those involving bluebird bio/BioMarin, Celgene/Juno Therapeutics, Gilead Sciences/Kite, Novartis/AveXis and the CDMO CELLforCURE, Roche/Spark Therapeutics, and Smith & Nephew/Osiris Therapeutics, have been made recently as a result of the CGT market's expansion.
Many bio/pharma businesses have re-invested in R&D, In order to standardize vector manufacturing and purification, utilize forward engineering approaches in cell therapies, improve cryopreservation of cellular samples, and explore the development of off-the-shelf allogeneic cell solutions.
In order to increase their manufacturing capabilities, pharmaceutical companies are now closely examining their internal capabilities and either investing in their own manufacturing facilities or outsourcing to contract development and manufacturing organizations (CDMOs) or contract manufacturing organizations (CMOs).
Lack of money, infrastructure, and capacity forces small biotech companies developing cell and gene therapies to form strategic collaborations with contract manufacturers. This has fueled cell and gene therapies (CGTs) market expansion. Additionally, investments in this area are growing enormously, which further fuels the market.
How is AI contributing to the Cell and Gene Therapy Manufacturing Industry?
To improve the manufacturing of cell and gene therapy, AI enhances the speed of the development process and stabilizes the complex production conditions. It optimizes the condition of processes with real-time manufacturing information and enhances uniformity with anticipatory learning. AI is also able to anticipate equipment failures long before they happen, as well as facilitate smooth production changes on a large scale.
Market Outlook
- Industry Growth Overview: Clinical momentum keeps the industry growing. Automation enhances effectiveness throughout manufacturing settings.
- Global Expansion: Infrastructure is being expanded in Germany, the UK, and Canada to supply regional manufacturing requirements.
- Major investors: Thermo Fisher Scientific, Lonza Group, Catalent, WuXi AppTec, Fujifilm Holdings, increase capacity and technologies.
Market Scope
| Report Coverage | Details |
| Market Size by 2035 | USD 47.18 Billion |
| Market Size in 2025 | USD 8.94 Billion |
| Market Size in 2026 | USD 10.44 Billion |
| Growth Rate from 2026 to 2035 | CAGR of 18.1% |
| Largest Market | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | By Therapy Type, By Indication, By Vector Type, By Manufacturing Type, By End Use, andRegion |
| Regions Covered | Asia Pacific, North America, Europe, Latin America, Middle East and Africa |
Market Dynamics
Drivers
The COVID-19 impact, which had earlier resulted in restrictive containment measures involving social estrangement, remote work, and the closure of commercial activities that resulted in operational challenges, is largely to blame for the growth. The companies are now resuming their operations and adjusting to the new normal while recovering from the impact.
The market has been primarily driven by an exponential increase in the clinical pipeline and an increase in the number of regulatory approvals for innovative medicines.
Market participants are working hard to increase their market presence as they take into account the significant growth opportunities in the contract development of cellular and gene-modified medicines. Additionally, bio producers are forming strategic partnerships with contract producers to quicken the R&D of their candidate programs. The emergence of several new competitors and the expansion of product development capabilities as a result of the growing demand for CMO/CDMO services have had a favorable impact on market revenue.
In order to advance the production of cell and gene therapies, several novel techniques are being introduced. For instance, the potential of single-use technology in production workflows is being investigated by the makers. This method is becoming more popular in this field since it can expedite development while also cutting costs and production times. In the upcoming years, market growth is predicted to be supported by such technological developments in space.
The growing demand for cutting-edge treatments has increased market competition among participants. Companies that manufacture cell therapies and CDMOs are inking agreements in order to hasten the development of their products and acquire a competitive edge. Additionally, ongoing clinical research projects have driven the contract manufacturing segment's revenue growth. Additionally, increase in outsourcing of the manufacturing process for cell and gene therapy also supports segment growth.
Restraints
There are many difficulties in the development, including safety and efficacy problems, drawn-out clinical study protocols, strict regulatory frameworks, and high prices of cell and gene therapy. Some of the key elements include the capacity to simultaneously treat heterogeneous systems with a variety of cells, high gene transfer efficiency, low cell toxicity, single cell specificity to the intended target, and single cell specificity.
Several CAR T-cell therapies have been approved, but according to data from the World Health Organization (WHO), one in five cancer patients who are qualified for them die as they wait for a production facility. In contrast to the past, when many of these autologous products took about a month to generate, some of them may now be made in less than two weeks.
The development of more sophisticated gene-transfer tools with CARs (such as transposon, CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats), among others) and the use of centralized organization with standardized apheresis centers are two approaches being investigated by the health sector (collection and reinfusion).
Opportunities
A number of gene treatments have received approval, mostly for the treatment of rare diseases Numerous businesses, including Astellas Gene Therapies, Bayer, ArrowHead Pharmaceuticals, Bluebird Bio, Intellia Therapeutics, Krystal Biotech, MeiraGTx, Regenxbio, Roche, Rocket Pharmaceuticals, Sangamo Therapeutics, Vertex Pharmaceuticals, Verve Therapeutics, and Voyager Therapeutics, are researching novel gene therapy vectors to boost levels of gene expression/protein production, decrease immunogenicity, and improve durability.
Segment Insights
The Cell Therapy Segment Dominated The Cell and Gene Therapy Market In 2025 With A 65% Share
The cell therapy segment dominated the market with a 65% share in 2025. The focus on treating blood cancers and the surging rate of autoimmune disorders increases demand for cell therapy. The heavy investment in manufacturing cell therapy and the need for structural tissue repairs increases adoption of cell therapy. The increased use of stem cell therapies and strong results of cell therapy drive segment growth.
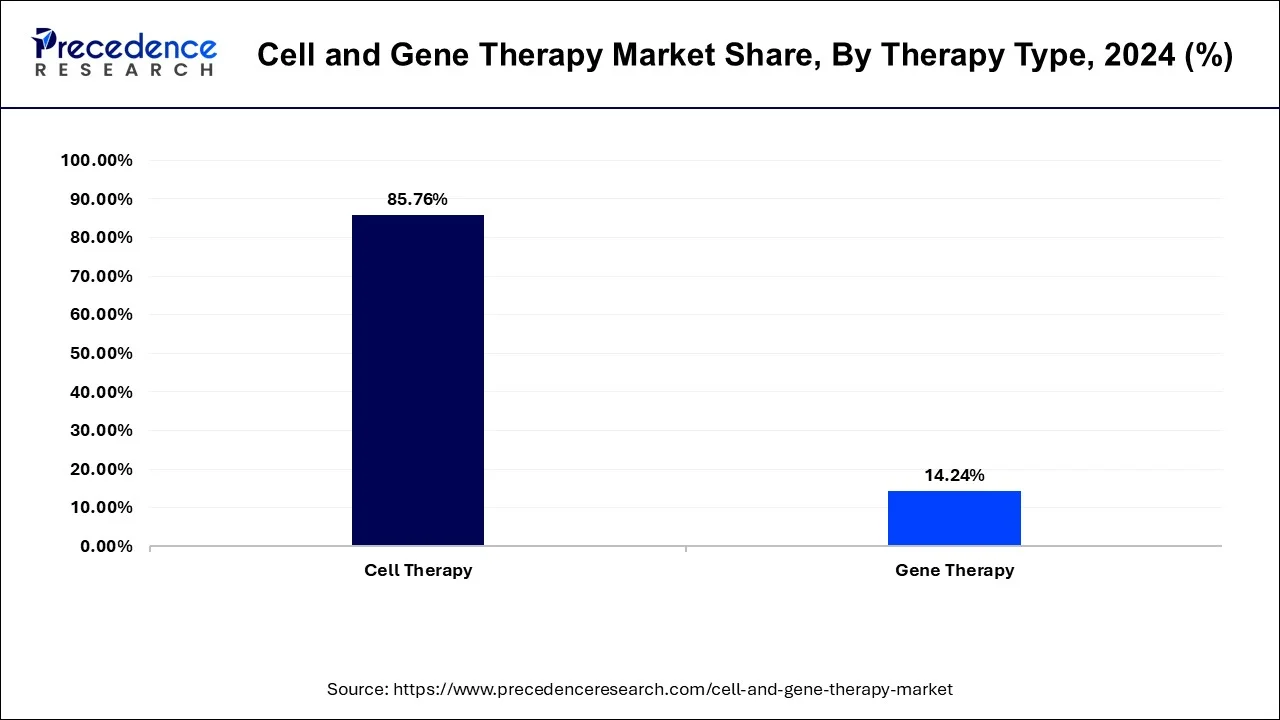
The gene therapy segment held the second largest share of 35% in the market in 2025. The rising number of rare genetic disorders and the expansion of in vivo treatments increases adoption of gene therapy. The prevalence of untreatable genetic disorders and the growing collaborative partnerships for gene therapy help with expansion. The ongoing advancements in gene editing technologies support the segment growth.
Cell and Gene Therapy Market By Therapy Type, 2022-2024 (USD Million)
| Therapy Type | 2023 | 2024 | 2025 |
| Cell Therapy | 4,454.4 | 5,061.6 | 5,792.7 |
| Gene Therapy | 2,381.3 | 2,728.7 | 3,149.1 |
Indication Insights
The Oncology Segment Dominated The Cell and Gene Therapy Market In 2025 With A 39% Share
The oncology segment dominated the market with a 39% share in 2025. The growing number of cancer patients and the expansion of autologous therapies increases adoption rate of cell and gene therapy. The targeted personalization and the high rate of hematological cancers use cell and gene therapy. The huge R&D investment and rapid innovation in CAR-T treatments drive the segment growth.
The musculoskeletal/orthopedic disorders segment held the second largest share of 21% in the market in 2025. The increased treatment gap in orthopedic disorders and the innovations in iPSCs increases adoption of cell & gene therapy. The focus on soft tissue repair and the treatment of non-union fractures increases the use of cell and gene therapy. The rise of age-based orthopedic disorders supports segment growth.
The genetic disorders segment held the third largest share of 12% in the market in 2025. The growing prevalence of sickle cell anemia and the surge in genomic understanding increases adoption of cell and gene therapy. The development of customized genetic disorder therapies helps with expansion. The innovations in gene editing and the increased diagnosis of genetic disorders boost segment growth.
Vector Type Insights
The Viral Vectors Segment Dominated The Cell and Gene Therapy Market In 2025 With A 72% Share
The viral vectors segment dominated the market with a 72% share in 2025. The growing in vivo gene delivery and the need to deliver therapeutic genes increases adoption of viral vectors. The growing use of cancer immunotherapy and the rise in modifications of ex vivo cells increase demand for viral vectors. The high efficacy, stable clinical pipeline, and targeted delivery of viral vectors drive segment growth.
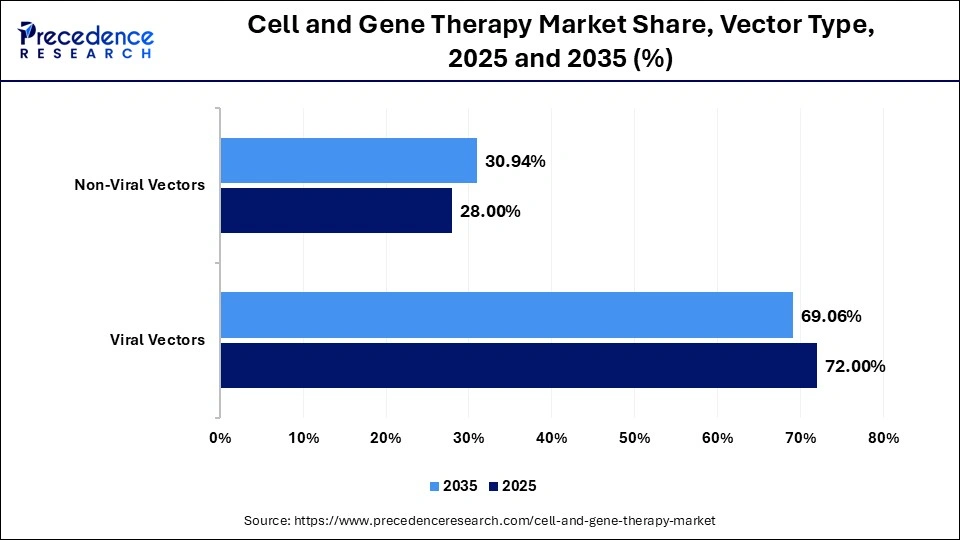
The non-viral vectors segment held the second largest share of 28% in the market in 2025. The increased development of CRISPR genome editing and the larger capacity of genetic cargo increases adoption of non-viral vectors. The explosive growth of mRNA technology helps with expansion. The lower cost, superior safety, lower immunogenicity, and reduced toxicity of non-viral vectors support segment growth.
Manufacturing Type Insights
The In-House Segment Dominated The Cell and Gene Therapy Market In 2025 With A 46% Share
The in-house segment dominated the market with a 46% share in 2025. The higher sensitivity of cell & gene therapy and the rising clinical trials increase demand for in-house manufacturing. The proprietary processes of CGT and the increased commercial production of CGT increase the adoption of in-house manufacturing. The flexibility, cost control, and regulatory compliance of in-house manufacturing drive segment growth.
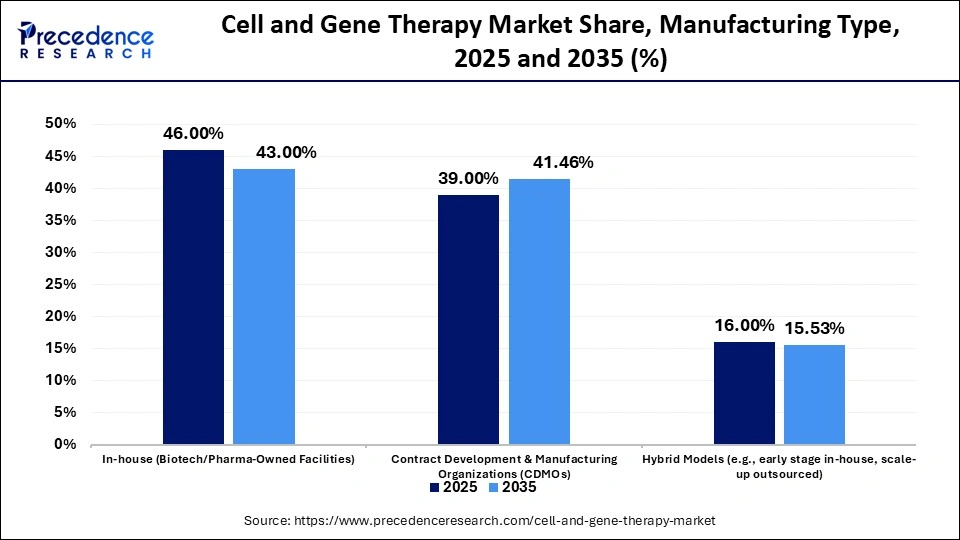
The contract development & manufacturing organizations (CDMOs) segment held the second largest share of 39% in the market in 2025. The customization of cell processing and lower operational cost increases adoption of CDMOs. The robust growth in the CGT pipeline and the need for specialized infrastructure for cell therapy increases adoption of CDMOs. The partnerships of biopharmaceutical companies and CDMOs support segment growth.
The hybrid models segment held the third largest share of 16% in the market in 2025. The focus on eliminating the transportation of fragile materials and faster turnaround time increases the adoption of hybrid models. The need to lower batch failures and the rise in modular manufacturing increases adoption of hybrid models. The balancing capacity, enhanced logistics, and operational scalability of hybrid models boost segment growth.
End Use Insights
The Hospitality & Specialty Clinics Segment Dominated The Cell and Gene Therapy Market In 2025 With A 63% Share
The hospitality & specialty clinics segment dominated the market with a 63% share in 2025. The requirement of sterile facilities for CGT therapy and the need for targeted cancer care increases use of hospitality and specialty clinics. The growing phase 3 clinical trial and specialized storage facilities in specialty clinics help with expansion. The safety protocols, multidisciplinary expertise, and specialized administration in hospitality & specialty clinics drive segment growth.
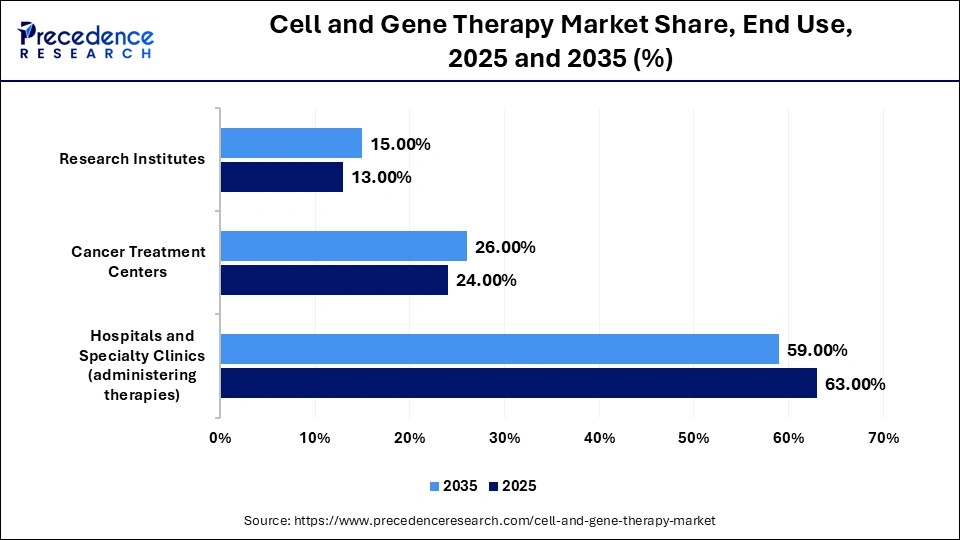
The cancer treatment centers segment held the second largest share of 24% in the market in 2025. The increased use of cell therapies for the treatment of blood cancers and the expansion of TIL therapies increases use of cancer treatment centers. The individual genetic profiles of therapies help with expansion. The high demand for extensive therapeutic clinical testing supports the growth of the segment.
The research institutes segment held the third largest share of 13% in the market in 2025. The development of delivery systems and the rising therapies pipeline increases the adoption of research institutes. The complex technical hurdles and the need for advanced therapies increases adoption of research institutes. The massive funding and supportive frameworks for research institutes boost segment growth.
Cell and Gene Therapy Market By End Use, 2022-2024 (USD Million)
| By End Use | 2023 | 2024 | 2025 |
| Hospitals and Specialty Clinics (administering therapies) | 4,331.2 | 4,908.1 | 5,601.5 |
| Cancer Treatment Centers | 1,604.3 | 1,847.4 | 2,142.4 |
| Research Institutes | 900.2 | 1,034.8 | 1,197.9 |
Regional Insights
What is the U.S. Cell and Gene Therapy Market Size?
The U.S. cell and gene therapy market size was estimated at USD 4.09 billion in 2025 and is predicted to be worth around USD 20 billion by 2035, at a CAGR of 17.21% from 2026 to 2035.
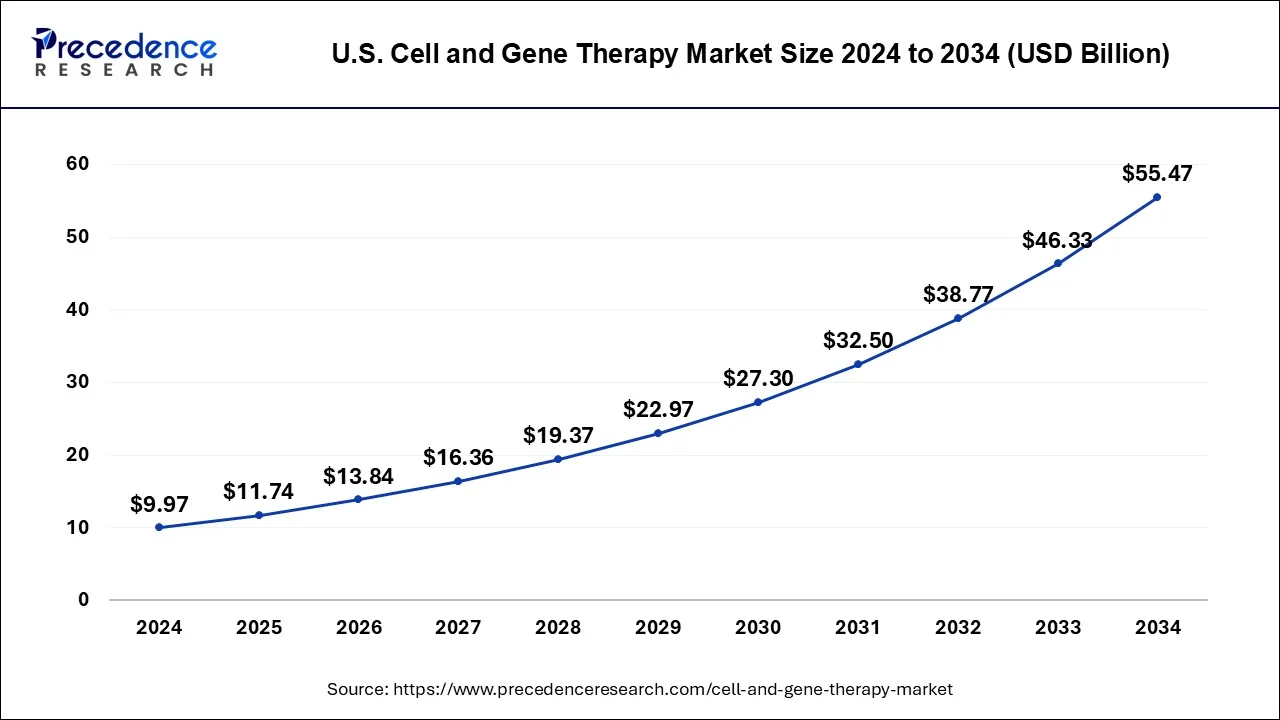
A significant number of ongoing clinical trials and a rise in corporate involvement in gene and cell therapy R&D are the primary drivers of regional market expansion. The North America region accounted for more than 50% of revenue share in 2025.
North America scored first for having the greatest number of gene therapy clinical trials , with more than 400 enterprises in the region actively engaged in the development of cell and gene therapy products for a variety of disorders.
Numerous brand-new collaborative research and innovation initiatives have been launched across Europe under the Horizon 2021 initiative. One of these endeavors is testing gene therapy utilizing viral vectors. This is expected to accelerate the growth of cell and gene therapy manufacturing services across Europe. The rise of the market is anticipated to be aided by the developed infrastructure and skilled labor force in European countries.
Value Chain Analysis of the Cell and Gene Therapy Manufacturing Market
- R&D: The effective identification of therapeutic targets and the development of viral vectors or cellular modification targeting a particular genetic disorder.
Key players: Novartis, Gilead (Kite Pharma), CRISPR Therapeutics - Clinical Trials and Regulatory Approvals: Therapies that are tested in humans and are shown to be safe and gain approval from authorities such as the FDA or EMA.
Key players: IQVIA, Charles River Laboratories - Packaging and Serialization: To make sure that items are kept in temperature-controlled packaging marked with labels that do not allow counterfeiting but allow tracking of items in supply chains.
Key players: Marken (UPS), Cryoport Systems, Almac
Cell and Gene Therapy Market Companies

- Merck KGaA: Provides bioprocessing equipment, specialized culture media, and viral vectors services that assist in the stable production of therapies in a variety of manufacturing settings.
- Novartis AG: Global manufacturing system for manufacturing CAR-T products and gene therapies based on automated bioreactor systems to supply in large quantities on a commercial basis.
Other Major Key Players
- Alnylam Pharmaceuticals Inc.
- Amgen Inc.
- Biogen Inc.
- CORESTEM Inc.
- Dendreon Pharmaceuticals LLC.
- Helixmith Co. Ltd.
- JCR Pharmaceuticals Co. Ltd.
- Kolon TissueGene Inc.
- Novartis AG
- Pfizer Inc.
Recent Developments
- In November 2025, BRIC-THSTI, in partnership with Miltenyi Biotec and BIRAC, launched India's inaugural hands-on training program on Cell and Gene Therapy Manufacturing. The program emphasizes CAR-T cell therapy techniques. ( https://www.expresspharma.in )
- In November 2025, Bharat Biotech launched "Nucelion Therapeutics," a subsidiary in Hyderabad's Genome Valley. The subsidiary serves as a CRDMO for advanced cell and gene therapies, offering comprehensive solutions for global life science innovators. ( https://www.gktoday.in )
- McKinsey will launch a new Digital Capability Center to quicken the development of cell and gene therapies (CGT). The Digital Capability Center will focus on accelerating operational excellence and digital transformations in biopharmaceutical manufacturing. The Digital Capability Center will join McKinsey's rapidly growing global network of Digital Capability Centers, immersive learning environments that inspire and prepare organizations to deliver sustainable performance improvement from operational-excellence and tech-enabled transformations.
- on october 3, 2022, Alexion's genomic medicines annuoced the acquisition of LogicBio's technology, by this acquiestuion they are looking to incorpate knowledgeable team for preclinical development, and team for research and development on rare diseases. LogicBio has developed a number of technical systems for the delivery and insertion of genes to address genetic disorders. Additionally, they will built a platform to improve the creation of viral vectors.
- On October 12, 2022, Moderna disclosed that Merck(MSD) has decided to exercise its $250 million option to co-develop and commercialize PCV mRNA-4157/V940. The vaccine is now being tested in a phase II clinical trial as adjuvant therapy for patients with high-risk melanoma in combination with pembrolizumab, Merck's programmed cell death protein 1 (PD-1) antibody.
- On October 3, 2022, Pfizer announced that they have successfully purchased Biohaven Pharmaceuticals which is a migraine drug manufacturing company. They recently manufactured NURTEC ODT (rimegepant) which is approved for both acute therapy and episodic migraine prevention in adults. With Pfizer's global reach and this acquisition, they will be able to provide migraine patients with more treatment alternatives.
Segments Covered in the Report
By Therapy Type
- Cell Therapy
- Stem Cell Therapy
- Hematopoietic Stem Cells (HSC)
- Mesenchymal Stem Cells (MSC)
- Induced Pluripotent Stem Cells (iPSC)
- Immune Cell Therapy
- CAR-T Cell Therapy
- TCR (T-cell Receptor) Therapy – targets intracellular antigens
- TIL (Tumor Infiltrating Lymphocyte) Therapy
- NK (Natural Killer) Cell Therapy
- Macrophage Therapies
- Dendritic Cell Therapy
- Others
- Stem Cell Therapy
- Gene Therapy
- In-vivo Gene Therapy
- Ex-vivo Gene Therapy
By Indication / Therapeutic Area
- Oncology
- Hematologic Malignancies
- Acute Lymphoblastic Leukemia (ALL)
- Non-Hodgkin Lymphoma (NHL)
- Chronic Lymphocytic Leukemia (CLL)
- Solid Tumors
- Glioblastoma
- Pancreatic Cancer
- Melanoma
- Prostate, Breast, Lung Cancer
- Others
- Head and Neck cancer
- Sarcomas
- Ovarian cancer (with experimental CGT trials)
- Hematologic Malignancies
- Genetic Disorders
- Hemophilia A and B
- Spinal Muscular Atrophy (SMA)
- Duchenne Muscular Dystrophy (DMD)
- Beta-thalassemia
- Sickle Cell Disease
- X-linked Adrenoleukodystrophy (X-ALD)
- Others
- Neurological Disorders
- Parkinson's Disease
- Alzheimer's Disease
- Batten Disease
- Huntington's Disease
- ALS (Amyotrophic Lateral Sclerosis)
- Others
- Cardiovascular Diseases
- Heart Failure
- Myocardial Infarction
- Peripheral Artery Disease
- Critical Limb Ischemia
- Others
- Ophthalmology
- Musculoskeletal / Orthopedic Disorders
- Infectious Diseases
- Metabolic Disorders
- Others
By Vector Type (Gene Delivery Method)
- Viral Vectors
- Adeno-Associated Virus (AAV)
- Lentivirus
- Retrovirus
- Herpes Simplex Virus (HSV)
- Adenovirus
- Others
- Non-Viral Vectors
- Lipid Nanoparticles (LNPs)
- Naked DNA/RNA Plasmids
- Electroporation
- Gene gun / microinjection
- CRISPR-Cas Delivery Systems (non-viral)
- Others
By Manufacturing Type
- In-house (Biotech/Pharma-Owned Facilities)
- Contract Development & Manufacturing Organizations (CDMOs)
- Hybrid Models (e.g., early stage in-house, scale-up outsourced)
By End Use
- Hospitals and Specialty Clinics (administering therapies)
- Cancer Treatment Centers
- Research Institutes
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- MEA
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
