Uterine Fibroid Treatment Devices Market Size and Forecast 2025 to 2034
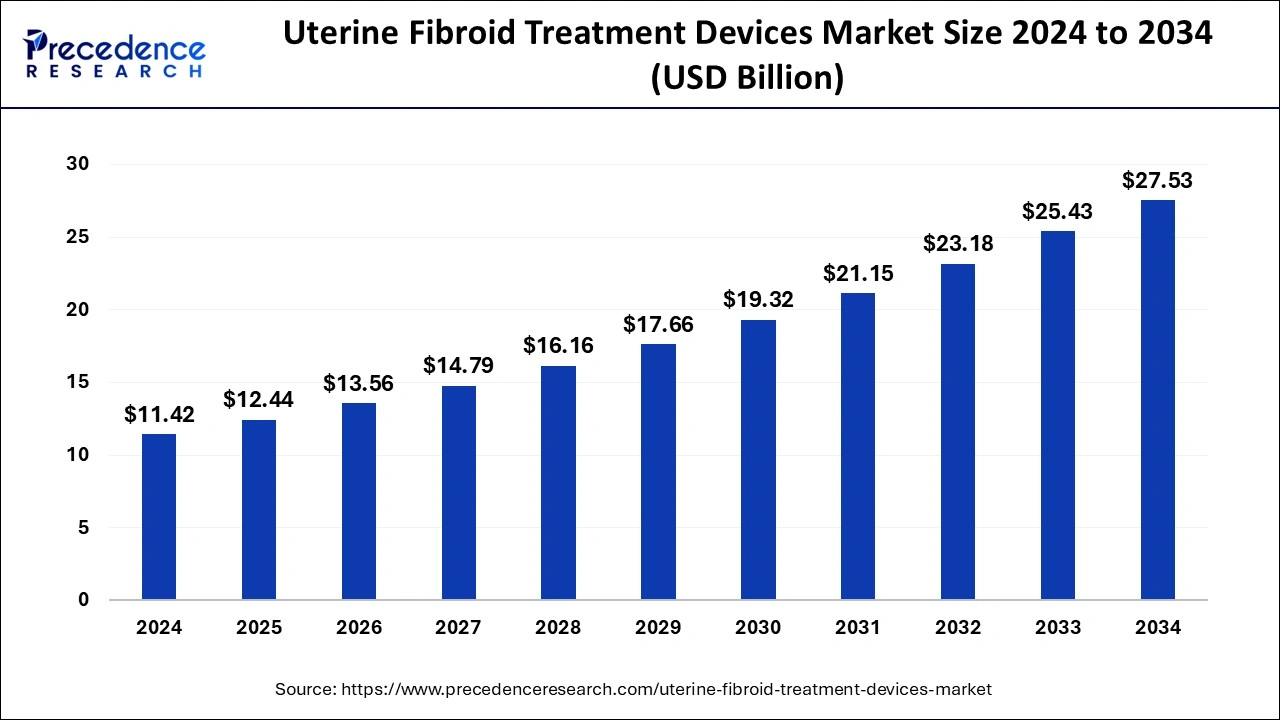
The global uterine fibroid treatment devices market was estimated at USD 11.42 billion in 2024 and is predicted to increase from USD 12.44 billion in 2025 to approximately USD 27.53 billion by 2034, expanding at a CAGR of 9.20% from 2025 to 2034. The uterine fibroid treatment devices market is driven by the growing desire for less invasive medical procedures.
Uterine Fibroid Treatment Devices MarketKey Takeaways
- In terms of revenue, the global uterine fibroid treatment devices market was valued at USD 11.42 billion in 2024.
- It is projected to reach USD 27.53 billion by 2034.
- The market is expected to grow at a CAGR of 9.20% from 2025 to 2034.
- North America held the largest market share of 62% in 2024.
- Asia Pacific is expected to witness a growth rate at a CAGR of 10.8% during the forecast period.
- By technology, the surgical techniques segment held the largest market share of 34% in 2024.
- By technology, the ablation techniques segment is observed to grow at a CAGR of 10.4% during the forecast period.
- By mode of treatment, the invasive treatment segment held the largest market share of 46% in 2024.
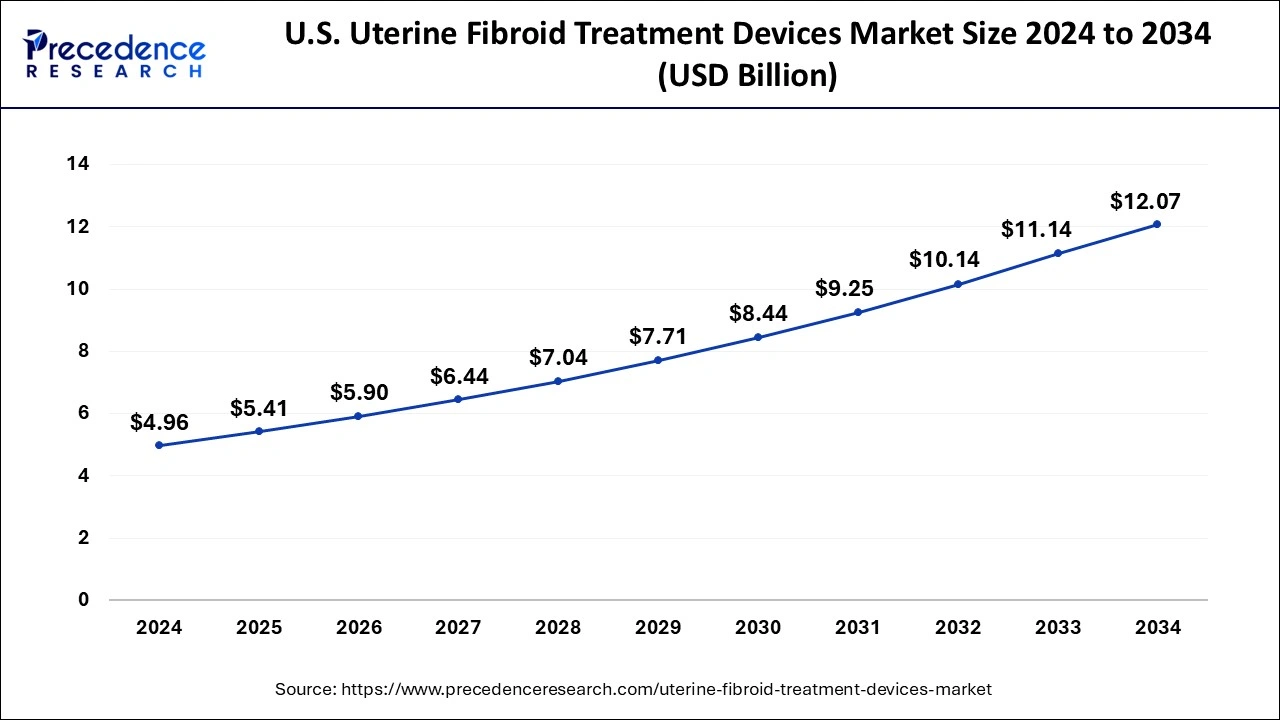
U.S.Uterine Fibroid Treatment Devices Market Size and Growth 2025 to 2034
The U.S. uterine fibroid treatment devices market size was estimated at USD 4.96 billion in 2024 and is anticipated to reach around USD 12.07 billion by 2034, growing at a CAGR of 9.30% from 2025 to 2034.
North America held the largest market share of 62% in 2024 in the uterine fibroid treatment devices market. North America has a highly developed healthcare infrastructure including access to state-of-the-art technologies, well-established medical facilities, and research institutions. This may result in the widespread and early adoption of novel uterine fibroid treatment technologies. The regulatory landscape is frequently seen as transparent but strict. Investing extensively in research and development and enabling quicker approval processes for innovative uterine fibroid treatment devices can boost confidence in patients and healthcare professionals. This dedication to innovation may lead to developing cutting-edge and successful uterine fibroids treatment tools.
- For instance, in January 2024, Pfizer Canada ULC, in collaboration with Sumitomo Pharma Canada, Inc., is pleased to announce that MYFEMBREE is now prescribed in Canada for the treatment of heavy menstrual bleeding linked to uterine fibroids in pre-menopausal women and mild to severe pain related to endometriosis in these women.
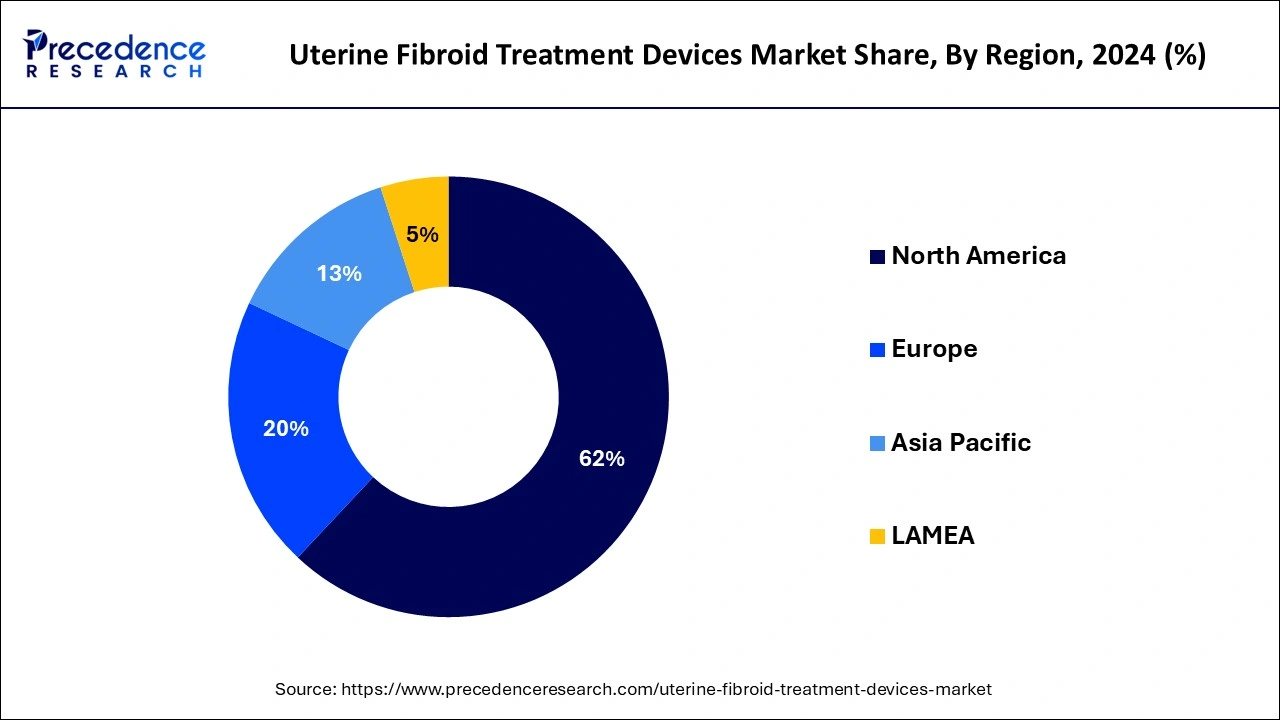
Asia-Pacific is observed to expand at a CAGR of 10.8% during the forecast period. The demand for treatment alternatives is rising as uterine fibroids are becoming more common in the Asia-Pacific area. The need for treatment devices may increase due to increased knowledge about uterine fibroids and improvements in diagnostic methods that lead to the identification of more cases. Initiatives and policies by governments in the Asia-Pacific area that support healthcare could favor growth.
- For instance, in July 2023, Femtech in India as the US Food and Drug Administration granted Nesa Medtech 510(k) clearance from the FDA for its fibroid mapping tool. For patients with uterine fibroids, its Fibroid Mapping Reviewer Application aids in diagnosis and interventional procedure planning. Using ultrasound scans creates a three-dimensional uterus model that allows doctors to find and measure fibroids.
Market Overview
Non-cancerous growths called uterine fibroids form in the uterus's muscular wall. The uterine fibroid treatment devices market offers devices for treating uterine fibroids are utilized in a range of non-invasive and minimally invasive methods to manage these symptoms while maintaining uterine integrity. Typical device types include embolization devices, which stop blood flow to fibroids and cause them to shrink; myomectomy devices, which remove fibroids surgically; and ablation devices, which destroy fibroids using heat, radiofrequency radiation, or ultrasound.
Organon is a pharmaceutical business that focuses on women's health and has been trying to reinvent hysterectomy. The goal of Claria Medical is to produce a medical device that will enable the removal of the uterus to be completed more quickly and safely than with existing operations by using a minimally invasive laparoscopic technique. Organon brought Claria and its hardware entirely in January 2023 for an upfront $8 million expenditure from its R&D budget. The National Science Foundation and the National Institutes of Health have already awarded grants to Claria, a pre-series A business based in the San Francisco area.
Uterine Fibroid Treatment Devices MarketGrowth Factors
- The surge in the uterine fibroid treatment devices market is not merely a random occurrence. It is a direct result of the significant role played by improved diagnostic techniques and increased awareness. These advancements are opening new opportunities and paving the way for a promising future in the healthcare industry.
- Another exciting trend shaping the market is the growing popularity of devices customized to the requirements and preferences of specific patients. Coupled with the choice of minimally invasive procedures among younger women, this trend creates anticipation about the market's future direction.
- Younger women prefer minimally invasive procedures that maintain fertility.
- The uterine fibroid treatment devices market is witnessing a significant expansion, driven by the development of novel, more potent instruments such as radiofrequency ablation (RFA) and MRI-guided focused ultrasound (MRgFUS). These innovative solutions are not just gaining popularity, but they are also proving their effectiveness in treating fibroids, thus reinforcing confidence in the market's growth potential.
- Because fibroids are more common in women in this age range, there is a larger pool of possible patients.
- Government healthcare efforts and rising disposable incomes support uterine fibroid treatment devices market's expansion.
- Patient advocacy groups and educational efforts increase public knowledge about fibroids and available treatments.
- Overall, treatment expenses are decreased by devices that give quicker healing times and shorter hospital stays.
- The market adoption of a particular item might be significantly impacted by favorable insurance coverage.
Market Scope
| Report Coverage | Details |
| Growth Rate from 2025 to 2034 | CAGR of 9.20% |
| Market Size in 2025 | USD 12.44 Billion |
| Market Size by 2034 | USD 27.53 Billion |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | By Technology and By Mode of Treatment |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Growing awareness of symptoms and potential complications
There is a growing need for efficient and minimally invasive treatment alternatives as more women become aware of the symptoms and dangers of uterine fibroids. Because of increased early detection rates brought about by this understanding, there is a growing need for sophisticated medical devices that can offer focused and effective management strategies for uterine fibroids. As a result, the uterine fibroid treatment devices market for devices used to treat uterine fibroids is growing as patients and healthcare professionals look for cutting-edge solutions to this prevalent gynecological ailment.
Shift towards minimally invasive and non-invasive procedures
Incisions made during minimally invasive operations are usually smaller or nonexistent, which lowers the risk of bleeding, infection, and other problems that come with more standard surgical methods. Because they believe that less intrusive treatments will result in a quicker recovery, less scarring, and a lower risk of problems, patients frequently favor them. Minimally invasive methods cause less damage to the surrounding tissues, which lessens postoperative pain and suffering. Because focused ultrasound doesn't involve physical penetration, it further reduces discomfort compared to invasive procedures.
Restraint
Obtaining regulatory approval is a complex and lengthy process
Medical devices must receive regulatory approval before being marketed and utilized for patient care to guarantee their efficacy and safety. The regulatory process frequently entails stringent testing, clinical studies, and documentation regarding products used to treat uterine fibroids, like minimally invasive surgical instruments or novel therapeutic technologies.
Regulatory bodies like the European Medicines Agency (EMA) in Europe or the food and drug administration (FDA) in the United States often receive extensive data on device performance, safety profiles, and clinical results. These regulatory agencies review the provided data to determine whether the device satisfies safety requirements and helps treat uterine fibroids.
Opportunities
Increasing awareness of treatment options
As awareness increases, more people learn about the range of available uterine fibroid therapies, which boosts the market's growth. In addition to helping patients look for workable answers, this increased knowledge opens doors for medical professionals and device makers to satisfy the growing need for cutting-edge and varied treatment alternatives. Ultimately, greater awareness promotes technical developments and enhances patient results while aiding the general expansion and growth of the uterine fibroid treatment devices market.
Competitive landscape
Medtronic
A multinational leader in medical technology, Medtronic designs and produces a range of medical tools and treatments. In the meantime, Medtronic works in many medical specialties, such as neurology, diabetes, cardiology, and more. Medtronic has shown interest in creating products or programs about treating uterine fibroids and medical equipment and technologies for women's health.
- In November 2022, The TruClear system, a mechanically operated hysteroscopic tissue removal tool for the safe and efficient treatment of intrauterine abnormalities (IUA), was recently introduced by India Medtronic Private Limited. IUA is frequently used to diagnose conditions, including fibroids, polyps, adhesions, cancers, hyperplasia, and retained products of conception.
Technology Insights
The surgical techniques segment held the largest market share of 34% in 2024in the uterine fibroid treatment devices market during the forecast period. Uterine fibroids can frequently be removed or reduced with great effectiveness by surgical treatments, including myomectomy and hysterectomy. These processes offer a thorough and long-lasting remedy by going straight after the illness's underlying cause. Most surgeons are very skilled and experienced in performing uterine fibroid procedures. Their inclination for these operations is influenced by their experience with surgical techniques, which raises the adoption rate.
The ablation techniques segment is observed to grow at a CAGR of 10.4% in the uterine fibroid treatment devices market during the forecast period. Ablation procedures include using radiofrequency, microwave, or laser energy, among other approaches, to remove or destroy uterine fibroids. Compared to typical surgical operations, this strategy has various advantages, such as less intrusive procedures, shorter recovery times, and decreased risk.
Ablation procedures are becoming increasingly popular among patients since they frequently lead to reduced length of stay in the hospital, decreased pain following surgery, and a quicker return to regular activities. Furthermore, technological developments have produced ablation devices that are more accurate and effective, improving the treatment's total effectiveness.
Mode of Treatment Insights
The invasive treatment segment held the largest market share of 46% in 2024 in the uterine fibroid treatment devices market. More efficacy is frequently seen in invasive fibroid excision or symptom reduction therapies. Myomectomy and hysterectomy are two surgical procedures that directly address the existence of fibroids and can lead to a more thorough cure of symptoms. Because they address the underlying causes of uterine fibroids, invasive treatments frequently offer complete cures. Fibroids can be reduced or removed using surgical and minimally invasive methods, giving patients long-term symptom relief.
The non-invasive treatment segment is observed to grow at a significant pace during the forecast period. In contrast to typical surgical procedures, non-invasive treatments minimize risks, shorten recovery times, and improve patient satisfaction overall. Because they may target and eliminate uterine fibroids without requiring incisions or anesthesia, technologies, including magnetic resonance-guided focused ultrasound (MRgFUS), radiofrequency ablation, and focused ultrasound, have become increasingly popular. Reduced side effects, a desire for a quicker recovery, and an increasing need for minimally invasive procedures are the main drivers of this growing need for non-invasive solutions.
Uterine Fibroid Treatment Devices Market Companies
- Medtronic
- Stryker Corporation
- Boston Scientific Corporation
- INSIGHTEC
- CooperSurgical Inc.
- Hologic, Inc.
- Lumenis
Recent Developments
- In November 2023, to increase the commercialization of its sonata treatment, which targets uterine fibroids, Gynesonics announced an investment of $67 million.
- In June 2022, The Healthcare Products Regulatory Agency (MHRA) and UK Medicines have authorized the marketing of Yselty, an oral GnRH antagonist, to treat mild to severe signs and symptoms of uterine fibroids (UF) in adult women of reproductive years. ObsEva SA is a biopharmaceutical company developing and commercializing novel health therapies for women.
Segments Covered in the Report
By Technology
- Surgical Techniques
- Hysterectomy
- Myomectomy
- Laparoscopic Techniques
- Laparoscopic Myomectomy
- Myolysis
- Ablation Techniques
- Microwave Ablation
- Hydrothermal Ablation
- Cryoablation
- Ultrasound Ablation
- High Intensity Focused Ultrasound (HIFU)
- MRI-guided Focused Ultrasound (MRgFUS)
- Other Ablation Techniques
- Embolization Techniques
By Mode of Treatment
- Invasive Treatment
- Minimally Invasive Treatment
- Non-invasive Treatment
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content



 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
