What is the Uterine Fibroid Drugs Market Size?
The global uterine fibroid drugs market size is calculated at USD 3.16 billion in 2025 and is predicted to increase from USD 3.61 billion in 2026 to approximately USD 11.93 billion by 2035, expanding at a CAGR of 14.21% from 2026 to 2035. This market is growing due to the increasing prevalence of fibroids among women of reproductive age and the rising demand for non-surgical, effective treatment options.
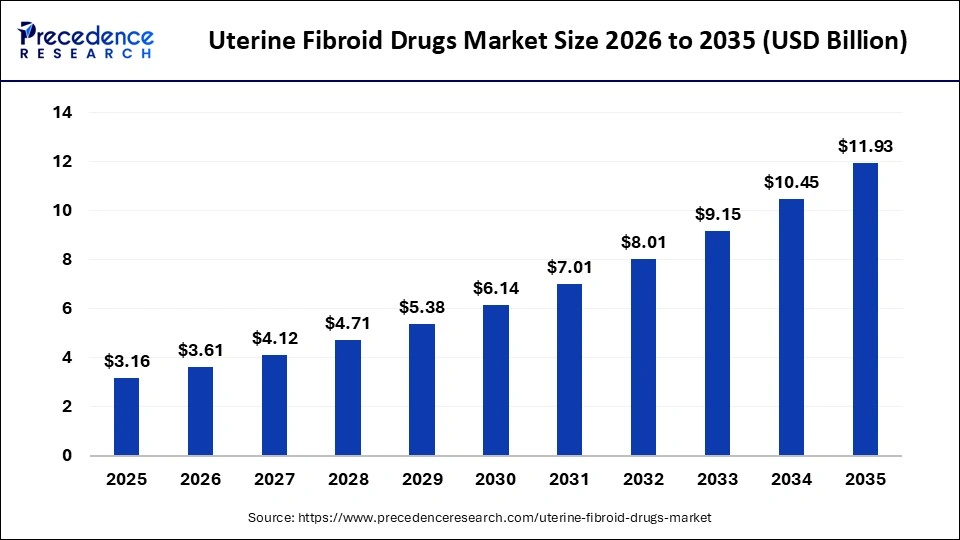
Market Highlights
- North America dominated the uterine fibroid drugs market, having the biggest share of 35% in 2025.
- The Asia Pacific is expected to grow at a notable CAGR between 2026 and 2035.
- By product type, the GnRH agonists and antagonists segment contributed to the largest market share of 35% in 2025.
- By product type, the selective progesterone receptor modulators (SPERMs) segment is growing at a notable CAGR between 2026 and 2035.
- By deployment type, the hospital/clinical-based prescriptions segment held the major market share of 55% in 2025.
- By deployment type, the retail pharmacy/drug stores segment is poised to grow at a strong CAGR between 2026 and 2035.
- By application, the symptomatic treatment (bleeding, pain) segment accounted for the largest market share of 50% in 2025.
- By application, the fertility & reproductive health segment is expanding at a notable CAGR between 2026 and 2035.
- By technology/mode of action, the hormonal modulations segment contributed the biggest share of 45% in 2025.
- By technology/mode of action, the GnRH receptor targeting segment will grow at a solid CAGR between 2026 and 2035.
- By end-user, the hospitals & specialty clinics segment recorded more than 50% of market share in 2025.
- By end-user, the gynecology & women's health centers segment is growing at a strong CAGR between 2026 and 2035.
Market Overview
Why is the Uterine Fibroid Drugs Market Growing So Rapidly?
The uterine fibroid drugs market is expanding as more women seek non-surgical pharmaceutical treatments to reduce symptoms such as heavy bleeding, pain, and pelvic pressure. Many patients prefer medical management because it allows them to avoid invasive procedures and maintain daily activities with fewer disruptions. This shift increases demand for therapies that offer predictable symptom control and support long-term quality of life.
Adoption is also rising due to better clinical awareness and improved diagnostic pathways. More healthcare providers use modern imaging tools and updated guidelines to identify fibroids earlier, which leads to earlier treatment discussions and a broader range of therapeutic options. Targeted hormonal treatments play a central role in this market. These therapies regulate hormone levels that influence fibroid growth, helping reduce bleeding and discomfort while maintaining a favourable safety profile.
Pharmaceutical companies continue to invest in clinical research and innovation to develop safer long-term treatment options. Research efforts focus on improving effectiveness, reducing side effects, and extending treatment duration without compromising safety.
Market Trends
- The preference for non-surgical treatments is increasing rapidly. Patients want less invasive, faster recovery options.
- Targeted hormonal therapies are gaining adoption. These offer better symptom control with fewer side effects.
- The focus on long-term symptom control is rising. Drugs are being designed to prevent recurrence and heavy bleeding.
- Movement toward personalized treatment plans is growing. Therapies are tailored based on age, fertility goals, and symptom severity.
- Increase in clinical research and new drug approvals. Pharmaceutical companies are investing heavily in innovation and safety.
- Better insurance and reimbursement support boost access. Coverage is improving drug-based treatments over surgical procedures.
Key Technological Shifts
| Technological Shift | Description | Impact on Market |
| Oral GnRH antagonists | Development of once-daily oral therapies replacing injectable hormones | Higher patient compliance and faster adoption |
| Add back therapy combinations | Use of hormonal add-back formulations to reduce side effects | Better safety profile and long-term usage feasibility |
| Non-hormonal drug development | Pipeline focused on targeting fibroid growth without hormonal disruption | Expands treatment options for patients with contraindications |
| Personalized dosing algorithms | AI/clinical data used to design individualized treatment plans | More effective symptom control and reduced adverse events |
| Novel drug delivery systems | Long-acting formulations, sustained release mechanisms | Improved therapeutic outcomes and reduced dosing frequency |
| Digital monitoring platforms | Apps and digital tools to track pain, bleeding, and treatment response | Better patient engagement and real-time care adjustment |
Uterine Fibroid Drugs Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 3.16 Billion |
| Market Size in 2026 | USD 3.61 Billion |
| Market Size by 2035 | USD 11.93 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 14.21% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product Type, Deployment Type, Application, Technology/Mode of Action, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Which Future Demands Will Shape the Uterine Fibroid Drugs Market?
| Future Demand | Description | Why it Matters |
| Non-surgical drug therapies | Increased preference for oral, minimally invasive treatments | Reduces surgery burden and recovery time |
| Long-term symptom control solutions | Drugs that reduce recurrence, pain, and heavy bleeding | Improves patient quality of life and adherence |
| Better safety and tolerability | Low side effects, hormone-sparing therapies | Expands patient eligibility and acceptance |
| Affordable treatment alternatives | Cost-effective drugs for wider access | Enables adoption in emerging markets |
| Strong insurance and reimbursement support | Coverage for drug-based therapies | Boosts uptake of newer therapeutics |
| Faster regulatory approvals | Streamlined pathways for innovative drugs | Speeds commercialization and patient access |
Uterine Fibroid Drugs Market Segmental Insights
[[segment_insights]]
Uterine Fibroid Drugs Market Regional Insights
[[regional_insights]]
Top Uterine Fibroid Drugs Market Companies
- AbbVie Inc
- Pfizer Inc
- Bayer AG
- Takeda Pharmaceutical Company Limited
- Gedeon Richter Plc.
- Myovant Sciences Ltd.
- Allergan (AbbVie subsidiary)
- Ferring Pharmaceuticals
- HRA Pharma
- Johnson & Johnson
- Novartis AG
- Astellas Pharma Inc.
- Eisai Co., Ltd.
- Lupin Limited
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (Viatris)
- Cadila Healthcare Limited
- Biocon Limited
- Glenmark Pharmaceuticals
- Sanofi S.A.
Recent Developments
- In February 2025, Kissei Pharmaceutical submitted a new drug application in Japan for Linzagolix, an oral GnRH antagonist designed to treat uterine fibroids. This regulatory step reflects growing investment in non-surgical and hormone-targeted therapies that improve patient quality of life. The move is expected to accelerate commercial rollout across Asia as approvals progress.(Source: https://www.kissei.co.jp )
- In August 2024, Theramex announced that NICE issued guidance recommending Linzagolix for adults with moderate to severe uterine fibroid symptoms in the UK. This update expands access to drug-based treatment through national health services, reducing reliance on surgery. It also positions Linzagolix as a competitive option in Europe's women's health market.(Source: https://www.pharmiweb.com )
- In September 2024, Theramex launched Yselty in Germany, marking its first commercial availability in the European market. This launch supports broader regional adoption of non-surgical therapy options, including treatments targeting heavy bleeding.
Uterine Fibroid Drugs Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
