What is the Biomarkers Market Size?
The global biomarkers market size is calculated at USD 94.24 billion in 2025 and is predicted to increase from USD 107.90 billion in 2026 to approximately USD 318.76 billion by 2034, expanding at a CAGR of 14.50% from 2025 to 2034.
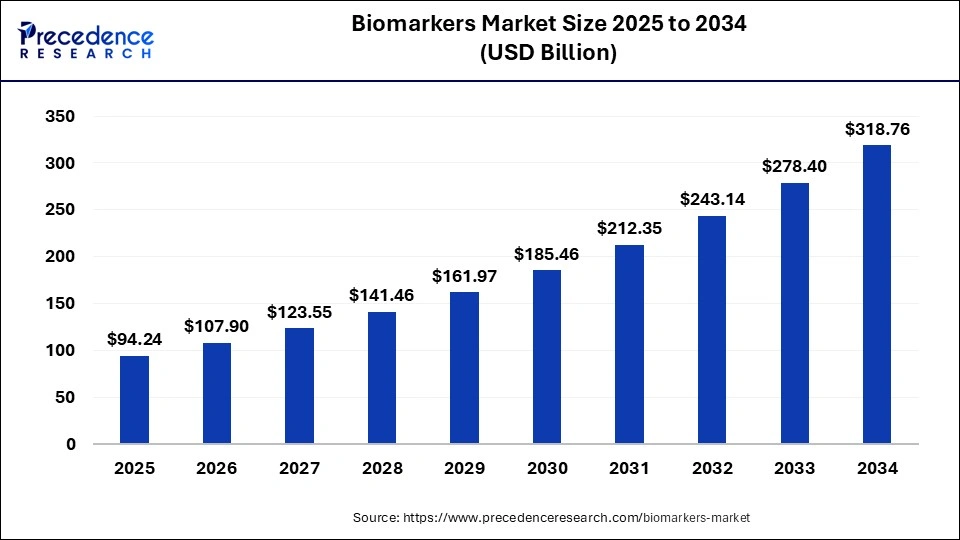
Biomarkers Market Key Takeaways
- North America has held a revenue share of more than 46% in 2024.
- By type, the safety segment has accounted highest revenue share in 2024.
- By application, the drug discovery and development segment will dominate the market in 2024.
- By disease, the cancer segment captured a biggest revenue share of in 2024.
AI in the Market
AI is changing the biomarkers market completely by speeding up the process, increasing precision, and shortening the time necessary for development. It allows scientists to interpret complex data coming from different sources, such as genomics, imaging, and medical history, and then they can find the disease patterns that are not visible. AI is one of the major factors that makes personalized medicine possible by indicating which patients will respond to a particular treatment and also discovering the most valuable biomarkers for precision diagnosis. It makes clinical data analysis more efficient, causes fewer manual mistakes, and grants regulatory compliance. Algorithms powered by AI can generate digital biomarkers from the data that comes from wearables and remote-monitoring devices, thus enhancing the design of clinical trials, recruitment of patients, and prediction of outcomes.
Key Market Insights
Biomarkers have presented a major boost for early detection of cancer which can be a crucial development in the field of cancer diagnostics. Early detection can be a major advantage in the treatment and management of cancer. Biomarkers are vital as molecular markers of the physiological state of a cell at a definite time. Active genes, their corresponding protein products, and supplementary organic compounds produced by the cell generate these markers. As a regular cell progresses through the intricate process of transformation into a cancerous state, biomarkers can prove vital for the screening of early cancer and individuals at risk of evolving cancer. Consistent, non-invasive examinations for the early recognition of cancer improve the likelihoods for successful treatment and preclude the usage of an invasive biopsy.
Biomarkers Market Growth Factors
- Rising incidences of cancer
- Increasing focus on establishment of biomarker-based diagnostic methods
- Growing number of contract research organizations
- High incidence of chronic ailments
- Increasing research on the use of biomarkers in early detection of cancer
- Rapidly expanding applications of biomarkers in diagnostics
Market Scope
| Report Highlights | Details |
| Market Size in 2024 | USD 82.30 Billion |
| Market Size in 2025 | USD 94.24 Billion |
| Market Size by 2034 | USD 318.76 Billion |
| Growth Rate from 2025 to 2034 | CAGR of 14.50% |
| Largest Market | North America |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | By Type, By Application, and By Disease and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
What are the driving factors of the Biomarkers Market in Europe?
Europe fared well in the biomarkers market as it has solid research networks, cross-industry partnerships, and state-funded healthcare innovation programs as its drivers. The focus on personalized medicine and the relaxing of regulations are two major factors in the clinical translation of biomarkers. Besides, the region's academic brilliance as well as its interest in evidence-based healthcare have quickened the process of biomarker validation, leading to steady market growth in both therapeutic and diagnostic applications.
Germany Biomarkers Market Trends
Germany has a memorable part in the marketing of the European market for biomarkers because of its top-notch research facilities, world-class pharmaceutical industry, and well-established clinical infrastructure. Academia and biotech companies work together in mutual collaboration, which is the main driver of biomarker validation and commercialization. The nation's resolve towards precision medicine and pioneering in molecular diagnostics not only solidifies its position but also strengthens its role.
Type Insights
Safety Biomarkers segment contributed the highest market share in 2024 Safety Biomarkers segment recorded the prime market share in the global biomarkers market by type in 2024. Increasing use in the process of drug discovery is the major reason for high market share of safety biomarkers. Other factors such as increasing investment in research and development to develop new diagnostic procedures is expected to increase the usage of the safety biomarkers over the estimate period.
The efficacy biomarkers are projected grow at the highest CAGR during the forecast period mainly due to new product launches in the near future.
Disease Insights
Cancer segment has held the largest market share in 2024. Cancer is one of the most widely studied disorder in the world. Constant research in the cancer field has revealed that, use of biomarkers in early detection of the deadly disease can be a turning point in cancer management. Strong product pipeline of major manufacturers related to cancer biomarkers is expected to further boost the popularity of biomarkers in cancer management.
Neurological Diseases segment will expand at a significant CAGR during the forecast time-frame.
Application Insights
Drug Discovery and Development segment generated the major market share in 2024. The use of biomarkers is on a constant rise in drug discovery and development majorly due to the advantages offered by biomarkers to scaleup the clinical development stage. These factors contribute to the growth of drug discovery and development segment.
Regional Insights
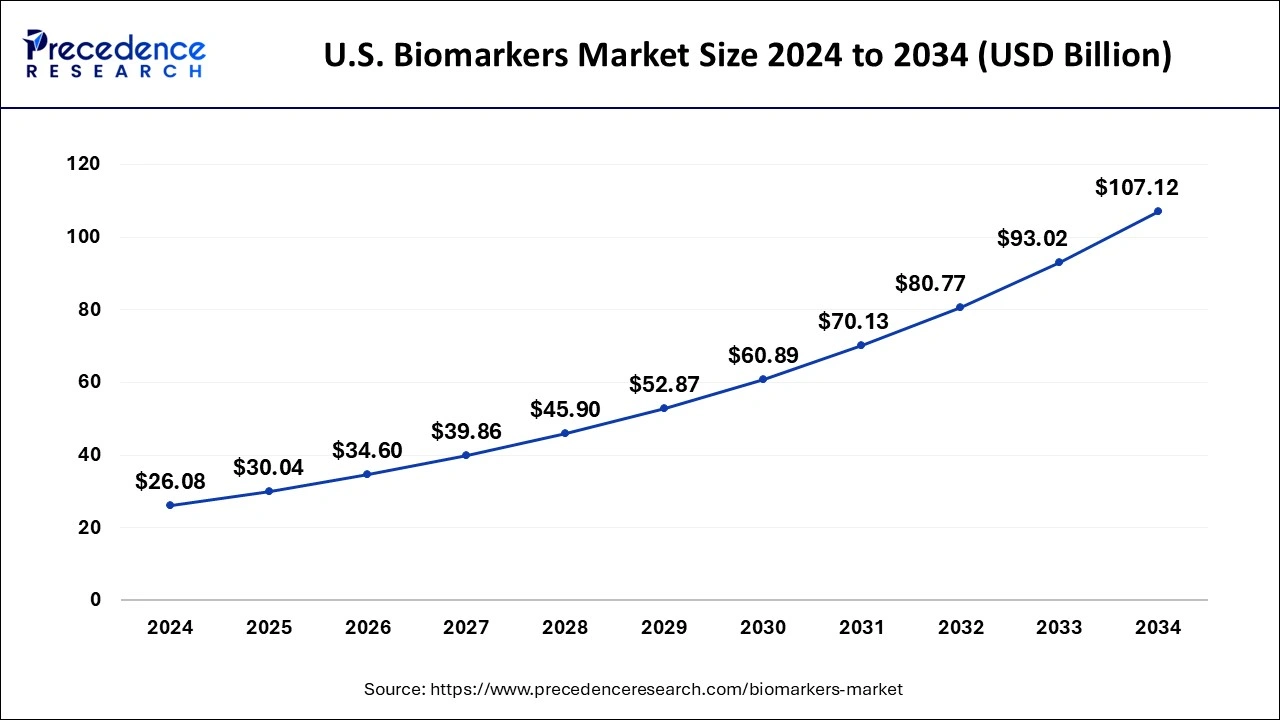
U.S. Biomarkers Market Size and Growth 2025 to 2034
The U.S. biomarkers market size is evaluated at USD 30.04 billion in 2024 and is predicted to be worth around USD 107.12 billion by 2034, rising at a CAGR of 15.20% from 2025 to 2034.
North America doninated the biomarkers market in 2024. The research report covers key trends and prospects of biomarkers products across different geographical regions including North America, Europe, Asia-Pacific, Latin America, and Middle East and Africa. Geographically, biomarkers market is conquered by North America owing to high incidence of cancer and existence of state-of-the-art healthcare infrastructure.
Several prominent players are widely adopting strategies, such as mergers and acquisitions, new product development, investment, and collaboration, to strengthen their position in the industry. For instance, in January 2023, Tempus announced a prospective study in collaboration with AstraZeneca that aims to identify biomarkers of response in patients with small cell lung cancer (SCLC). According to the American Cancer Society, in the U.S., the second-most common cancer is lung cancer, and approximately 13% of people diagnosed with lung cancer have SCLC.
- In October 2024, University of Nevada, Las Vegas (UNLV) received a USD 3.2 million investment from the Alzheimer's Drug Discovery Foundation (ADDF) to develop a biomarker observatory that will capture and categorize emerging information on blood tests, brain scans, and digital devices that help diagnose and monitor Alzheimer's disease.
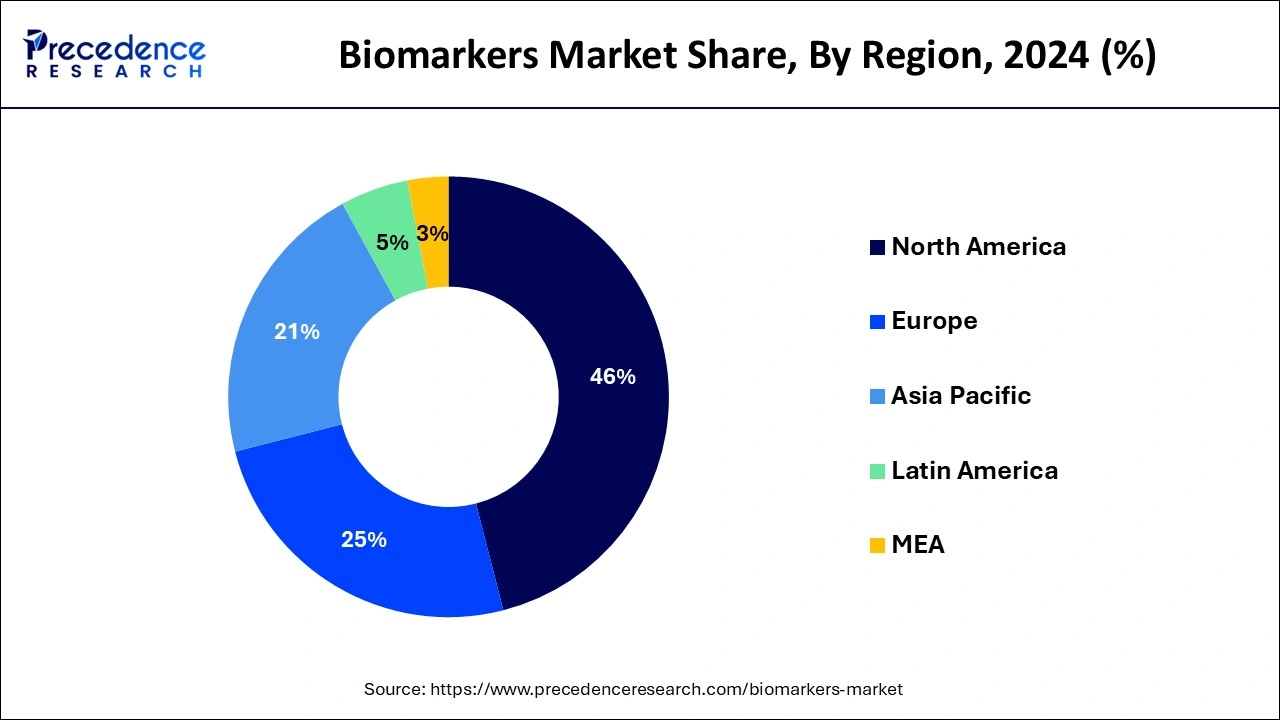
Asia Pacific is expected to expand at a rapid pace during the forecast period. The growth of the region is attributed to the increasing investment in advancing healthcare infrastructure, growing incidence of chronic diseases, increasing demand for personalized medicine, increasing research on the use of biomarkers in the early detection of cancer, and a supportive government framework. For instance, in March 2025, LuNGS Alliance, an initiative led by the Cancer Research and Statistics Foundation (CRSF), announced free Lung NGS (Next Generation Sequencing) biomarker testing for lung cancer patients across India. This initiative is backed by AstraZeneca, Pfizer, and Roche, with 4baseCare serving as the official lab partner. The initiative aims to drive awareness about precision oncology as an essential, accessible, and affordable treatment plan for lung cancer treatment.
How is North America leading the Biomarkers Market?
Because of its advanced healthcare infrastructure, strong research capabilities, and high diagnostic innovation adoption, North America is at the forefront of the market for biomarkers. The market is accelerated by both a favorable regulatory environment and heavy investments from the private and public sectors. The region is a pioneer in the areas of genomics, proteomics, and AI applications; thus, it will keep on revamping its precision medicine and biomarker-based clinical applications over a number of therapeutic areas.
United States Biomarkers Market Trends
The United States continues to be the most dominant country in the biomarker market worldwide, backed by its sophisticated research, regulation, and clinical trial activities. The usage of biomarkers in precision medicine and pharmaceutical companies' drug-making process results in better patient lives. The country where efficient biotech companies and the use of AI technology in the discovery of biomarkers prevail is, in fact, the leading one in the area of diagnosis and therapeutics.
How is Asia-Pacific performing in the Biomarkers Market?
Asia-Pacific is the fastest-growing biomarker market, being driven by the rapid healthcare infrastructure improvement and increasing support for biotech investments. The increasing number of diseases, along with the rising public awareness and availability of inexpensive clinical research, attract the collaboration of global companies. The support given by the respective governments for the genomics and personalized medicine initiatives aids innovation, while the increasing number of patients provides a large market for the biomarker-based therapeutics.
China Biomarkers Market Trends
China's biomarker market is highly dynamic and growing fast due to the influx of healthcare funds, the upgrading of laboratory equipment, and the increased emphasis on gene research. The government's strategies in favor of biomarker adoption, coupled with large-scale health initiatives in the population, are the primary reasons for the wide coverage of the country's strong biotechnology base and local innovation participation. Thus, China is placed well in terms of partnerships with international drug firms, as a major player in both the discovery and application of biomarkers.
Biomarkers Market Companies
- Bio-Rad Laboratories, Inc.
- Qiagen
- Epigenomics AG
- Abbott
- Hoffmann-La Roche Ltd.
- Agilent Technologies, Inc.
- Johnson & Johnson Services, Inc.
- Abbott
- Siemens
- Thermo Fisher Scientific, Inc.
Recent Developments
- In September 2024, Scientists in Chhattisgarh introduced India's first-ever biomarker kit, which could transform the way Covid-19 severity is predicted and managed. Developed by a team at the Multi-Disciplinary Research Unit (MRU) of Dr Bhimrao Ambedkar Hospital in Raipur. This innovation offers early detection of severe COVID-19 cases, promising to enhance patient outcomes and optimise healthcare resources.
- In October 2024, MEDiC Life Sciences, a Silicon Valley biotech startup, announced that it had entered a research collaboration with Hanmi Pharmaceutical, a pioneer biopharma company in Korea. In this collaboration, MEDiC will use its MCAT platform to identify cancer biomarkers for one of Hanmi's clinical assets.
- In July 2024, Biogen Inc., Beckman Coulter, Inc. announced a collaboration with Fujirebio to potentially identify and develop blood-based biomarkers for tau pathology in the brain and to potentially clinically advance and commercialize new tests for tau pathology in Alzheimer's disease (AD).
- In September 2025, Aptamer Group launched a biomarker discovery service, providing researchers with innovative methods to identify and validate new disease markers in pharmaceutical, biotechnology, and diagnostic development.
https://pharmaceuticalmanufacturer.media - In April 2025, the Viromarkers project launches in Rome on April 3-4, uniting over fifty researchers and partners to develop and validate biomarkers for improving patient care in HIV, Hepatitis D, and Cytomegalovirus infections.
https://www.eatg.org
Value Chain Analysis
- R&D (Research and Development): The primary goal of this department is to identify and validate new biomarker molecules as well as to carry out the research studies needed to support their diagnostic and therapeutic application.
Key players: Merck & Co., Johnson & Johnson, Roche - Clinical Trials and Regulatory Approvals: The process starts with human trials to test the biomarker's safety and efficacy. After that, the regulatory approvals required for the product to enter the market are obtained.
Key Players: IQVIA, Syneos Health (Contract Research Organizations), Food and Drug Administration (FDA) - Formulation and Final Dosage Preparation: The final product is a reliable diagnosis or treatment product that is ready for clinical or commercial use, coming from approved biomarker findings.
Key Players: Sun Pharma, Dr. Reddy's Laboratories, Cipla - Packaging and Serialization: This process involves the assigning of unique identification codes, labeling, and packing to ensure that the product can be traced and that counterfeit distribution is prevented.
Key players: Kezzler, Aphena Pharma Solutions, Sharp Services - Distribution to Hospitals, Pharmacies: The logistics involved in distributing genuine biomarker products to the clinics, labs, and pharmacies for the patients are managed efficiently.
Key players: Cardinal Health, AmerisourceBergen, McKesson - Patient Support and Services: The ultimate goal of this offering is to make sure patients are well educated and to give them access to different programs and support for treatment adherence, thus increasing their engagement and optimizing the outcomes of biomarker-driven healthcare.
Key players: Almac, Biomarkers Healthcare Private Limited
Segments Covered In the Report
By Type
- Validation
- Efficacy
- Safety
By Disease
- Neurological Diseases
- Cancer
- Immunological Diseases
- Cardiovascular Diseases
- Others
By Application
- Personalized Medicines
- Drug Discovery & Development
- Diagnostics
- Others
By Geography
- North America
- U.S.
- Canada
- Europe
- Germany
- France
- United Kingdom
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Rest of Latin America
- Middle East & Africa (MEA)
- GCC
- North Africa
- South Africa
- Rest of Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content
.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
