What is the Genetic Toxicology Testing Market Size in 2026?
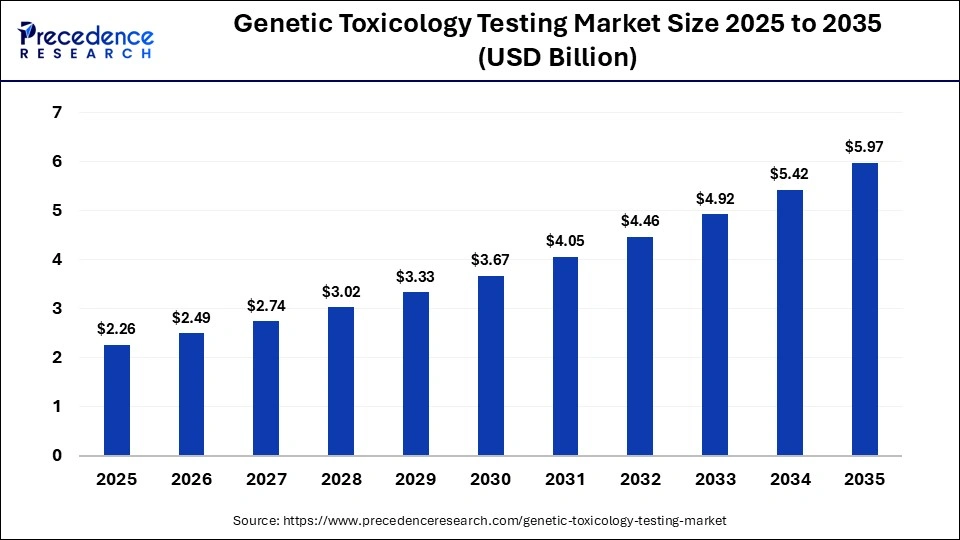
The global genetic toxicology testing market size accounted for USD 2.26 billion in 2025 and is predicted to increase from USD 2.49 billion in 2026 to approximately USD 5.97 billion by 2035, expanding at a CAGR of 10.20% from 2026 to 2035. Increasing regulatory focus on safety, advancements in testing technology, and the integration of artificial intelligence are key drivers of market growth.
Key Takeaways
- North America led the genetic toxicology testing market in 2025 with approximately 39% share.
- Asia-Pacific is expected to be the fastest-growing region in the forecasted period, with a CAGR of nearly 10.5%.
- By product, the services segment dominated the market with approximately 48% share in 2025.
- By product, the reagents and consumables segment is expected to grow at the fastest CAGR of 8.5% in the market between 2026 and 2035.
- By test type, the in vitro testing segment held a dominant position in the market with a share of approximately 62% in 2025.
- By test type, the in vivo testing segment is expected to grow with the highest CAGR of 8.8% in the market during the studied years.
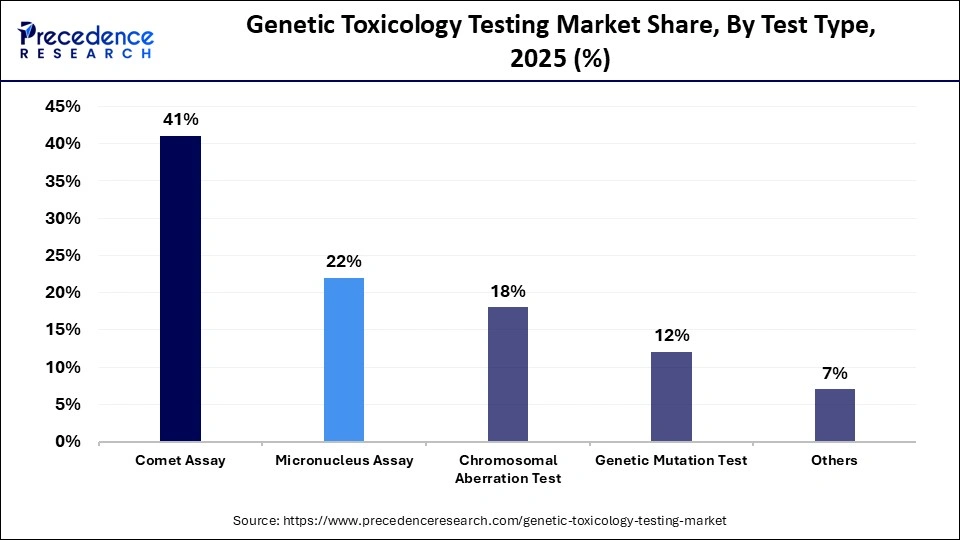
- By assay type, the comet assay segment registered its dominance over the global genetic toxicology testing market with a share of 41% in 2025.
- By assay type, the genetic mutation test segment is expected to expand rapidly in the market with a CAGR of 9.0% in the coming years.
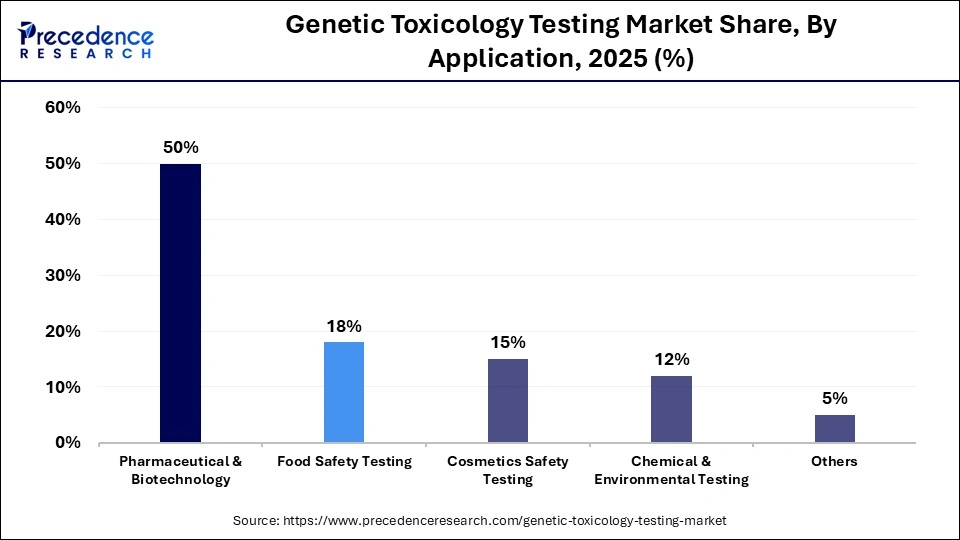
- By application, the pharmaceutical and biotechnology segment held the largest revenue share of 50% in the market in 2025.
- By application, the food safety testing segment is expected to witness the fastest growth in the market with a CAGR of 9.4% over the forecast period.
- By end-user, the pharmaceutical and biotechnology companies segment contributed the biggest revenue share of 45% in the market in 2025.
- By end-user, the contract research organizations (CROs) segment is expected to gain the highest market share with a CAGR of 9.2% between 2026 and 2035.
Genetic Toxicology Testing Market Overview
Genetic toxicology testing evaluates the potential of chemicals, pharmaceuticals , food additives, and environmental substances to damage genetic material such as DNA or chromosomes. These tests identify mutations, chromosomal aberrations, or other genetic alterations that may lead to cancer or hereditary diseases. Genetic toxicology testing is widely used during drug development, chemical safety assessment, and regulatory approval processes. Pharmaceutical, biotechnology , food, and cosmetic industries rely on these tests to ensure product safety and compliance with global regulatory standards.
Impact of Artificial Intelligence on Genetic Toxicology Testing
Artificial intelligence (AI) is transforming the domain of genetic toxicology by enabling faster, more accurate, and equitable diagnostic techniques. Drug developers are progressively utilizing AI for both discovery and safety trials to attain swifter and more economical outcomes, aligning with global initiatives aimed at decreasing animal testing.
Commonly employed AI and statistical techniques in this domain include k-nearest neighbors, which classifies a test chemical by locating training chemicals that exhibit the closest similarity; multiple linear regression, which employs several explanatory variables to forecast the outcome of a response variable through a multivariate linear equation; and the Naïve Bayes classifier, which relies on Bayes' theorem and presumes conditional independence among molecular descriptors (i.e., explanatory variables). The primary objective of incorporating AI into toxicology is to enhance accuracy and quality, reduce time and expenses, and lessen animal testing and cruelty.
Genetic Toxicology Testing Market: Key Trends
- Rising demand for Food Safety Assessments: Food safety assessment is emerging as a key trend in the toxicology testing industry. Growing consumer preference for additive-free, preservative-free, and allergen-free food is increasing the use of genetic toxicology testing to ensure that ingredients do not pose potential genetic health risks.
- AI-assisted Predictive Toxicology: The integration of AI-assisted predictive tools and advanced software platforms, such as Comet assay analysis systems, in silico modeling, and bioinformatics-based toxicity screening, increases the use of genetic toxicology testing in pharmaceutical and biotechnology research, while improving the accuracy and reliability of results.
- Environmental Genotoxicity Assessment: Genetic toxicology testing is increasingly applied in environmental monitoring as governments and research institutions focus on pollution-related health risks. Methods such as the micronucleus test help detect DNA damage caused by industrial chemicals, heavy metals, radiation, and air pollutants.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 2.26 Billion |
| Market Size in 2026 | USD 2.49 Billion |
| Market Size by 2035 | USD 5.97 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 10.20% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Assay Type, Application, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segmental Insights
Product Insights
How the Services Segment Dominated the Genetic Toxicology Testing Market?
The services segment dominated the market with a share of 48% in 2025, driven by rising demand for outsourced services, the increasing complexity of testing procedures, and the need for specialized expertise. Service providers offer a broad range of genetic toxicology testing solutions, including in vitro and in vivo tests, genotoxicity assays, and regulatory consulting. In addition, growing regulatory requirements and the intricate nature of genetic toxicology testing are encouraging companies to partner with specialized providers that offer advanced infrastructure and technical capabilities. As a result, many organizations are increasingly outsourcing their testing needs to these expert service providers, further promoting market growth.
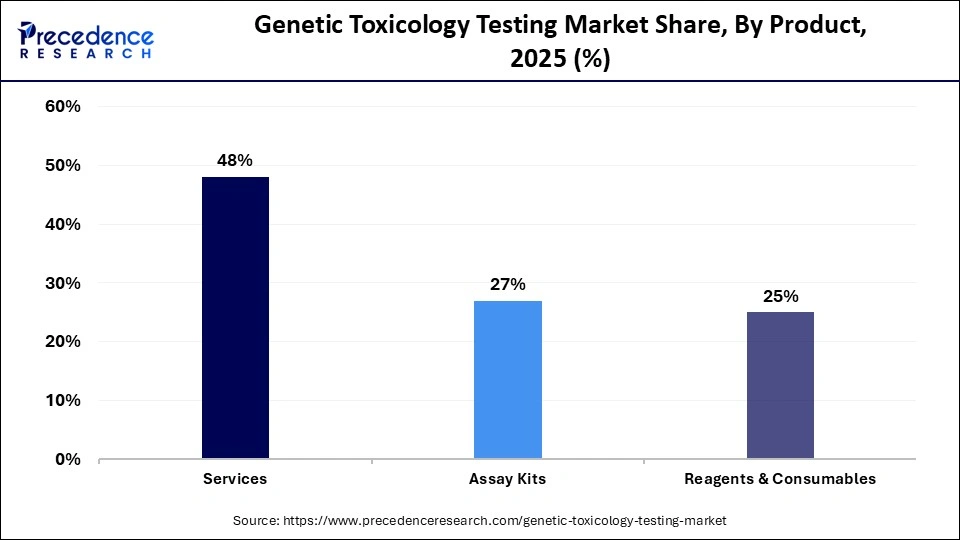
The reagents and consumables segment is expected to show the fastest growth with a CAGR of 8.5% over the forecast period, driven by rising demand for modern equipment, high-purity reagents, and high-quality consumables. Having the most suitable products in the laboratory is essential for achieving reliable and reproducible results. Over the past several decades, scientific research has become increasingly dependent on sterile, single-use products. Key fields that commonly rely on laboratory consumables include biochemistry, food and drug inspection, academic chemistry laboratories, clinical diagnostics, and hospitals, supporting segment growth.
Test Type Insights
Which Test Type Segment Dominated the Genetic Toxicology Testing Market?
The in vitro testing segment accounted for the highest revenue share of 62% in the market in 2025, driven by the increasing adoption of in vitro approaches in genetic toxicology testing. In vitro testing does not require live subjects; instead, studies and assays are performed on biological materials (such as cells and tissues) in a controlled laboratory environment. Generally, in vitro studies refer to work conducted on cells, tissues, or other biological components isolated from the living organism of interest.
In the context of genetic toxicology, in vitro assays are widely used to detect DNA damage, chromosomal aberrations, gene mutations, and other genotoxic effects of chemicals and drug candidates. In vitro testing offers several advantages, including reduced testing time, ease of use, cost-effectiveness, support for high-throughput screening (HTS), and a higher level of scalability. As a result, it enables more efficient early-stage safety evaluation and helps to minimize unnecessary use of vivo studies and animal testing in subsequent phases of toxicological assessment.
The in vivo testing segment is expected to gain the highest market share with a CAGR of 8.8% between 2026 and 2035, driven by the increasing demand for accurate and reliable testing methodologies, ongoing technological advancements, and the need to understand how an entire living organism responds to therapeutic or chemical exposure. In vivo studies are essential for evaluating the pharmacokinetics of substances within living systems, including drug absorption, metabolism, distribution, and elimination, while also assessing safety and efficacy.
Because these studies are conducted on whole biological systems, they provide more comprehensive and physiologically relevant data compared to isolated testing models. In addition, in vivo testing follows strict regulatory protocols and standardized testing frameworks, which further enhance the reliability of the results. However, modern drug development typically integrates both in vivo and in vitro models for toxicological evaluation before obtaining regulatory approval from agencies such as the U.S. FDA.
Assay Type Insights
Why Did the Comet Assay Segment Dominate the Genetic Toxicology Testing Market?
The comet assay segment contributed the biggest revenue share of 41% in the market in 2025, as the comet assay is a relatively simple, sensitive, and quantitative method for studying DNA damage (including oxidative damage) and repair at the level of individual cells. Also known as single-cell gel electrophoresis, this technique is based on agarose-embedded DNA electrophoresis.
It is used to determine DNA damage in isolated cells. In addition, the development of advanced software has improved the accuracy and efficiency of comet assay analysis. For example, Comet Assay IV is a semi-automated system with single-click tools for quick, reliable DNA damage data analysis. These advantages have strengthened the adoption of comet assays and contributed significantly to the growth of this market segment.
The genetic mutation test segment is expected to witness the fastest growth in the market with a CAGR of 9.0% over the forecast period, driven by heightened awareness of genetic disorders, a growing demand for personalized medicine, advancements in genomics and biotechnology, more stringent regulatory requirements, and the rising incidence of various diseases. DNA testing offers comprehensive insights into an individual's genetic composition. It aids in confirming whether a person has or is predisposed to a specific illness, evaluating the risk of developing certain conditions, and identifying whether an individual carries a specific altered gene that may be inherited by their offspring, thus facilitating the growth of this segment.
Application Insights
Which Application Segment Dominated the Genetic Toxicology Testing Market?
The pharmaceutical and biotechnology segment held the largest revenue share of 50% in the market in 2025, as the extensive use of genetic toxicology studies throughout drug discovery and development pipelines. Companies also increasingly rely on these tests during early screening and preclinical safety assessment. In addition, the need to evaluate mutagenicity, carcinogenicity, and reproductive toxicity before initiating clinical trials remains a critical regulatory requirement.
Ongoing technological advancements in molecular biology techniques, including improved nucleic acid synthesis, gene cloning, and advanced analytical tools, are enhancing the precision and efficiency of genetic toxicology testing methods. As regulatory authorities continue to strengthen safety standards and demand comprehensive toxicological data packages, pharmaceutical and biotechnology companies are increasing their investments in robust genetic toxicology testing solutions, thereby supporting the growth of this application segment.
The food safety testing segment is expected to expand rapidly in the market with a CAGR of 9.4% in the coming years, driven by a strengthening regulatory framework for food safety and increasing consumer awareness regarding food quality and nutritional value. Rising demand for high-quality ingredients and customized food products tailored to specific health conditions is further contributing to this trend, as concerns related to food allergens and potential genetic toxicity continue to grow.
Demand for advanced testing is also increasing, with major players such as Eurofins Scientific and SGS SA focusing on expanding their food testing services. As a result, food safety testing is gaining significant traction and emerging as a key growth driver within the market.
End-User Insights
Which End-User Segment Led the Genetic Toxicology Testing Market?
The pharmaceutical and biotechnology companies segment led the market with a share of 45% in 2025, due to the critical role these tests play in drug discovery and preclinical development. Early identification of genotoxic and mutagenic effects helps minimize costly failures in later stages of clinical trials, making genetic toxicology an essential component of safety assessment.
Regulatory authorities also mandate comprehensive genotoxicity evaluation as part of submissions such as Investigational New Drug (IND) and New Drug Application (NDA) filings before approval by agencies like the U.S. FDA. Consequently, pharmaceutical and biotechnology companies widely employ genetic toxicology testing for DNA damage assessment, mutagenicity studies, and chromosomal aberration analysis during preclinical evaluation, supported by specialized research expertise and strict regulatory compliance requirements.
The contract research organizations (CROs) segment is expected to grow with the highest CAGR of 9.2% in the market during the studied years, driven by the rising trend of outsourcing research and testing activities by major pharmaceutical and biotechnology companies. CROs manage all or part of trial-related tasks, including specialized genetic toxicology studies that may be difficult or costly to perform in-house.
Operating under strict regulatory and ethical frameworks and supported by Institutional Review Boards (IRBs) and independent ethics committees, CROs offer specialized expertise, dedicated facilities, and efficient study management, making them increasingly preferred partners for toxicological research.
Regional Insights
North America Genetic Toxicology Testing Market Size and Growth 2026 to 2035
The North America genetic toxicology testing market size is estimated at USD 881.40 million in 2025 and is projected to reach approximately USD 2,385.15 million by 2035, with a 10.34% CAGR from 2026 to 2035.
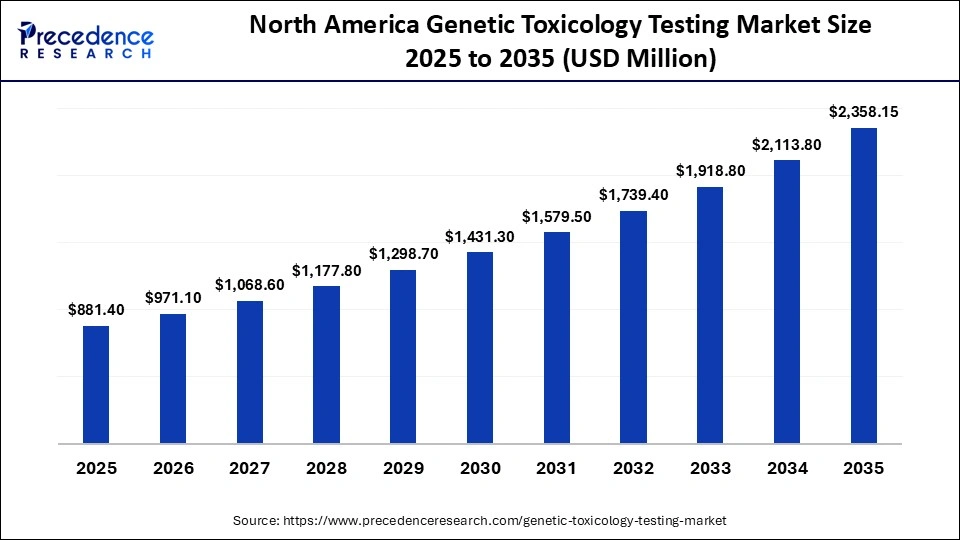
Why North America Dominated the Genetic Toxicology Testing Market?
North America held a major market share of 39% in 2025, driven by growing drug development activities, significant investments in pharmaceutical pipelines, and the presence of key industry players that enhance access to genetic toxicology testing services for pharmaceutical and biotechnology companies across the region. The regional market is further supported by a well-established base of pharmaceutical and biotechnology companies, along with specialized firms that focus on genetic testing solutions.
U.S. Genetic Toxicology Testing Market Size and Growth 2026 to 2035
The U.S. genetic toxicology testing market size is calculated at USD 661.05 million in 2025 and is expected to reach nearly USD 1,780.40 million in 2035, accelerating at a strong CAGR of 10.42% between 2026 and 2035.
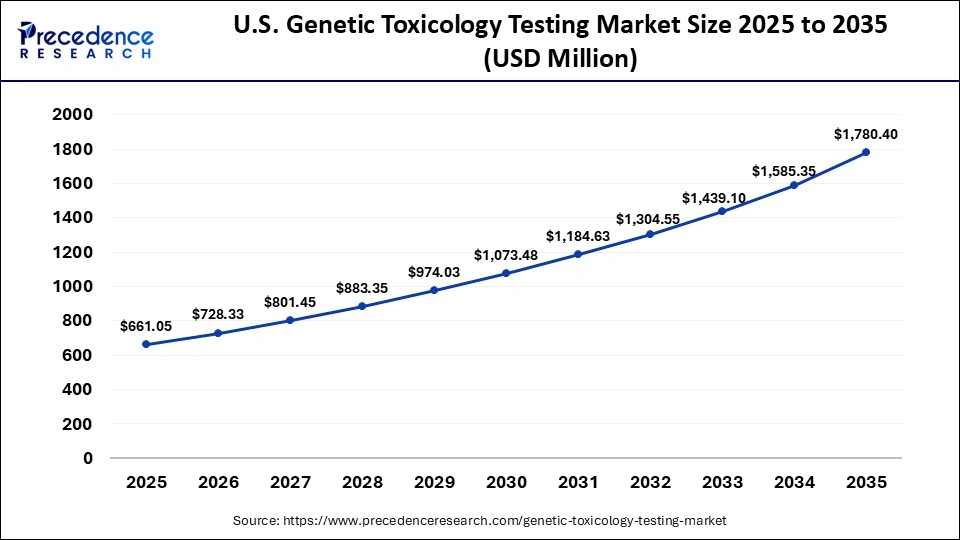
U.S. Market Trends
The U.S. leads the market in North America, due to the presence of key players, a robust research infrastructure, and increasing R&D investments. In addition, the strict regulatory framework in the U.S., particularly the guidelines provided by the U.S. FDA, continues to increase the demand for genetic toxicology testing in drug safety evaluation. Major companies such as Thermo Fisher Scientific, Charles River Laboratories, and Laboratory Corporation of America Holdings also play an important role in strengthening the regional market and supporting its growth.
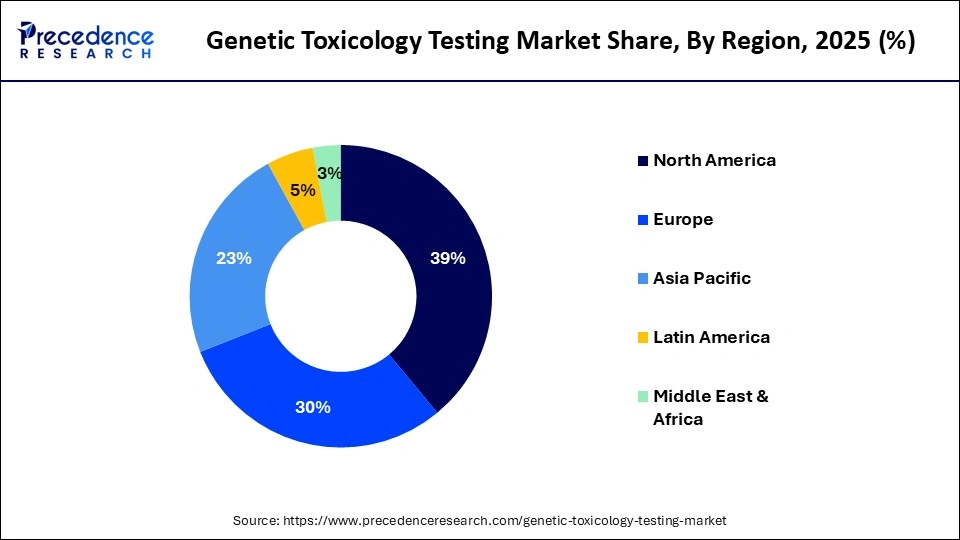
How is Asia-Pacific Growing in the Genetic Toxicology Testing Market?
Asia-Pacific is expected to experience the fastest growth during the predicted timeframe. This growth is primarily driven by increasing investment in research and development, a stronger focus on scientific innovation, and rising awareness of genetic testing and its role in evaluating product safety and potential genotoxic effects.
Industries such as pharmaceuticals, biotechnology, food, and cosmetics are placing greater emphasis on assessing safety, genotoxic potential, and efficacy during product development, which is significantly increasing the demand for genetic toxicology testing across the region. Additionally, evolving disease patterns and the growing prevalence of genetically influenced and lifestyle-related disorders are encouraging more comprehensive toxicological assessments. Regulatory oversight in the region is also becoming more structured and stringent.
India Market Trends
In India, the Central Drugs Standard Control Organization (CDSCO) plays a key role in ensuring compliance with safety and quality standards across regulated industries. CDSCO enforces strict evaluation frameworks for product safety and clinical validation. Strengthening regulatory standards, along with increasing public awareness of scientifically validated and safety-tested products, is expected to further accelerate the adoption of genetic toxicology testing across the Asia Pacific region.
Genetic Toxicology Testing Market Companies
- Thermo Fisher Scientific
- Charles River Laboratories
- Eurofins Scientific
- SGS SA
- Syngene International
- Inotiv
- Gentronix
- Creative Bioarray
- Toxys
- Xenometrix AG
- MB Research Laboratories
- Nelson Laboratories
- BioRad Laboratories
- Danaher Corporation
- Merck KGaA
Recent Developments
- In January 2026, Variant Bio, a private biotechnology company, introduced an AI-driven platform that analyzed genetic data to identify new drug candidates for advancement into human clinical trials.(Source: https://www.reuters.com )
- In October 2025, AIG Hospitals introduced a DNA-based drug response test, marking a significant step forward in precision medicine in India. In collaboration with GenepoweRx, the hospital had launched the country's first affordable pharmacogenomics test, a genetic screening tool that helped doctors identify which medications were likely to be most effective for each patient based on their DNA profile. (Source: https://newsmeter.in )
- In April 2025, Shed, a wellness solutions company, launched a new line of at-home genetic testing kits designed to provide personalized health insights based on an individual's DNA analysis. These kits focused on how genetics influenced responses to nutrition, fitness, sleep, and stress management. (Source: https://clpmag.com )
Segment Covered in the Report
By Product
- Services
- Assay Kits
- Reagents & Consumables
By Test Type
- In Vitro Testing
- In Vivo Testing
By Assay Type
- Comet Assay
- Micronucleus Assay
- Chromosomal Aberration Test
- Genetic Mutation Test
- Others
By Application
- Pharmaceutical & Biotechnology
- Food Safety Testing
- Cosmetics Safety Testing
- Chemical & Environmental Testing
- Others
By End-User
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations (CROs)
- Academic & Research Institutes
- Regulatory & Testing Laboratories
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Free Sample
Get a Free Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting