What is In Vitro Diagnostics Quality Controls Market Size in 2026?
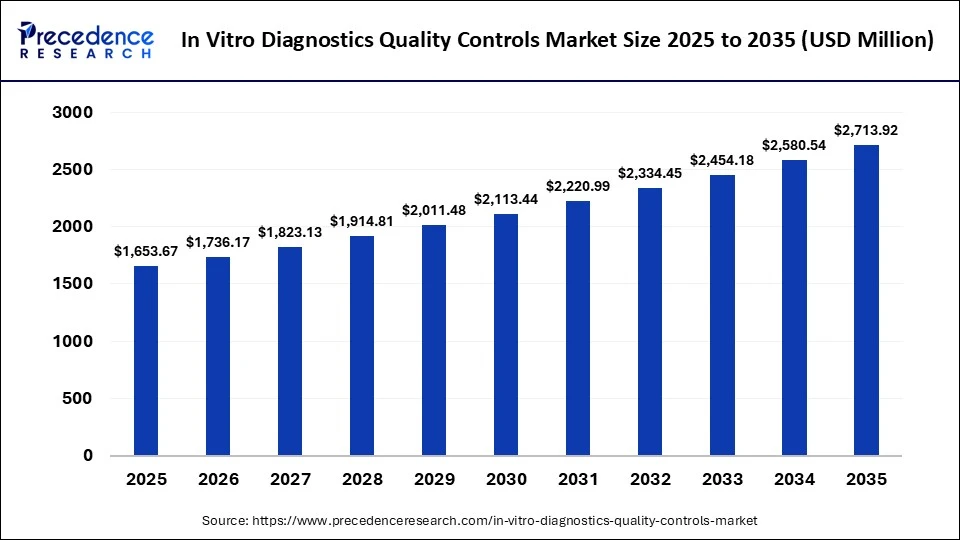
The global in vitro diagnostics quality controls market size was calculated at USD 1,653.67 million in 2025 and is predicted to increase from USD 1,736.17 million in 2026 to approximately USD 2,713.92 million by 2035, expanding at a CAGR of 5.09% from 2026 to 2035. The market is driven by the increasing use of advanced diagnostic solutions in research laboratories, coupled with the rapid expansion of the microbiology sector.
Market Overview
The global in vitro diagnostics quality controls market deals with the production and distribution of reference materials, reagents, and analytical solutions used to ensure the accuracy, reliability, and consistency of laboratory diagnostic tests. Quality control products detect the performance of diagnostic instruments and assays across haematology, microbiology, clinical chemistry, immunoassays, molecular diagnostics, and other laboratory disciplines. These solutions are essential for laboratory accreditation, regulatory compliance, and patient safety in clinical testing environments.
What is the Role of AI in In Vitro Diagnostics Quality Controls?
Artificial Intelligence plays a crucial role in in vitro diagnostics quality controls by improving precision in diagnostic results, lowering human errors, and improving compliance in QC centers. Also, AI is used to monitor lab instruments to identify patterns for predicting future component failure, reduce waste, and enhance the consistency of reagent production. In recent times, AI has been deployed in laboratories to reduce mistakes in pre-analytical, analytical, and post-analytical phases. Additionally, AI supports automated trend analysis and predictive insights, helping laboratories maintain compliance with regulatory standards and improve overall diagnostic reliability.
- In May 2025, Diagnostics.ai launched a CE-IVDR-certified transparent AI platform. This AI-enabled platform is designed to enhance the capabilities of molecular diagnostics.
Major Market Trends
- Partnerships: Several pharma companies are partnering with diagnostic solution providers to develop advanced in-vitro diagnostics solutions.
- Opening of Research Laboratories: The governments of different countries are investing significantly in opening new research laboratories to check the efficacy of novel therapeutics.
- Surging Prevalence of Genetic Diseases: The growing cases of genetic diseases in various nations, such as Japan, the UK, and Canada, have increased the demand for advanced testing solutions.
- Deployment of Cloud-based Data Management Solutions in Hospitals: Nowadays, modern hospitals have started deploying cloud-based data management solutions to facilitate real-time monitoring, peer benchmarking, and data analytics for identifying trends in different patients.
- Rising Adoption of Third-Party Controls by Laboratories: The laboratories are increasingly using third-party controls more than OEM controls, as it offers superior flexibility across multiple analyzer platforms, by delivering unbiased performance data, and cost-effectiveness.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1,653.67 Million |
| Market Size in 2026 | USD 1,736.17 Million |
| Market Size by 2035 | USD 2,713.92 Million |
| Market Growth Rate from 2026 to 2035 | CAGR of 5.09% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product & Service, Manufacturer Type, Technology, Application, End User, and region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Analysis
Product & Service Insights
Why Did the Quality Control Products Segment Dominate the Market?
The quality control products segment dominated the in vitro diagnostics quality controls market in 2025, capturing the largest share of 56%. This is mainly due to their wider application in clinical laboratories to enhance testing accuracy and ensure regulatory compliance. Laboratories are engaged in performing quality control to validate the equipment performance and check the reliability of reagents, which increases the demand for third-party and OEM-manufactured quality control products. Growing regulatory requirements, increasing adoption of advanced diagnostic technologies, and the need to minimize false positives and negatives have further reinforced the segment's leading position.
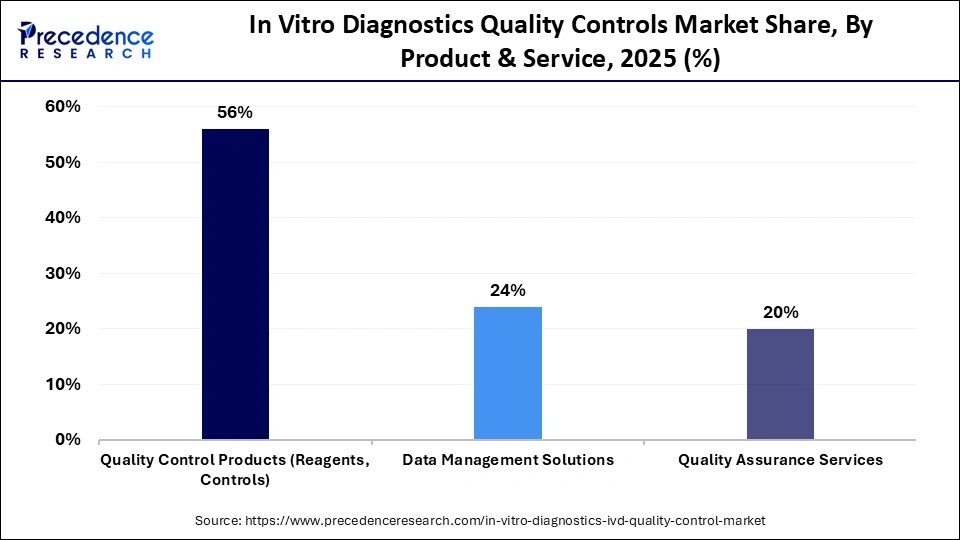
The data management solutions segment is expected to grow at the highest CAGR during the forecast period, owing to the surging adoption of automated solutions in the laboratories to enhance real-time monitoring. With the rising volume of quality testing, the laboratories have started deploying a wide range of advanced informatics platforms to streamline QC data analysis and reduce human errors. Data management solutions help in enabling trend analysis and automated flagging, which in turn improves the overall operational efficiency in laboratories.
Manufacturer Type Insights
What Made Third-Party Controls the Leading Segment in the Market?
The third-party controls segment led the in vitro diagnostics quality controls market with a major share of 58% in 2025. This is because these controls provide independent and unbiased verification of assay performance, ensuring greater accuracy and reliability than in-house or kit-based controls. They help laboratories meet stringent regulatory standards, maintain consistency across multiple instruments and platforms, and reduce the risk of errors in diagnostic testing. The flexibility to use them across a wide range of assays and compatibility with different diagnostic systems further strengthens their market dominance.
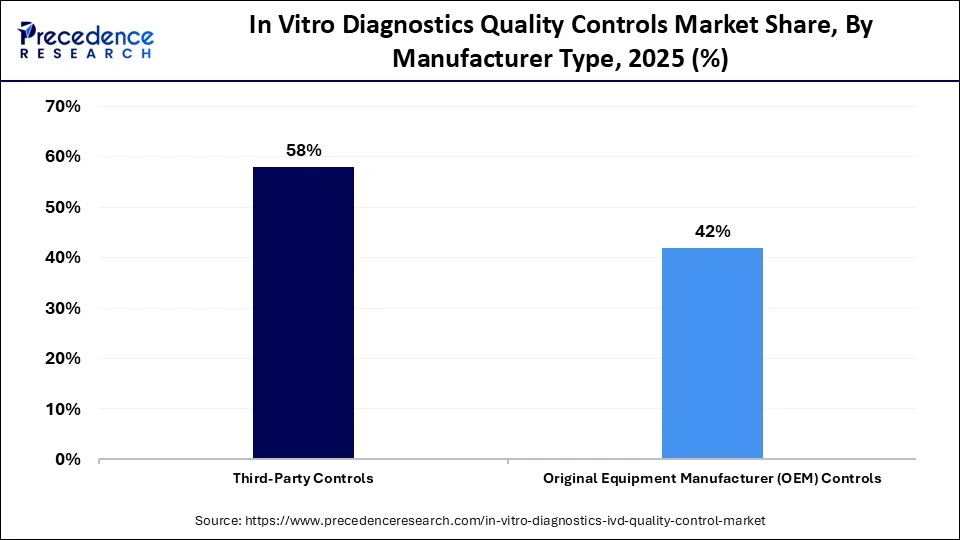
The original equipment manufacturer (OEM) controls segment is expected to expand at a significant CAGR between 2026 and 2035. This is mainly due to the rising demand for integrated solutions directly from diagnostic instrument manufacturers. OEM controls are designed to be fully compatible with specific analyzers, ensuring seamless operation, higher accuracy, and reduced risk of errors. Additionally, partnerships between OEMs and laboratories, along with the increasing adoption of advanced automated diagnostic systems, are driving the segment's growth. With the surging adoption of automated solutions in laboratories, the application of OEM-manufactured controls has grown significantly to reduce technical complexities.
Technology Insights
Why Did the Immunoassay Diagnostics Segment Lead the In Vitro Diagnostics Quality Controls Market?
The immunoassay diagnostics segment led the in vitro diagnostics quality controls market in 2025, holding a 29% share. The dominance of the segment is attributed to the increased testing volume and wide application of immunoassay in food safety, clinical diagnostics, and environmental monitoring. Immunoassays are mainly used to detect several chronic diseases, hormone levels, tumor markers, and autoimmune diseases. The sensitivity of these assays requires strict calibration and regular validation to provide accurate results, which in turn increases the demand for QC tools.
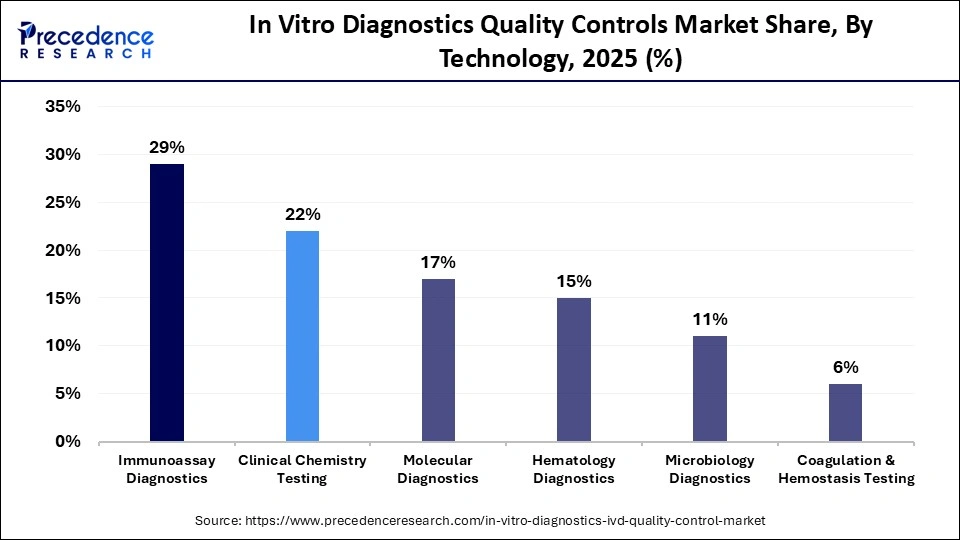
The molecular diagnostics segment is expected to grow with the fastest CAGR during the forecast period due to the growing adoption of real-time PCR and next-gen sequencing technologies for testing cancers and infectious diseases. Molecular assays are highly complex and sensitive, which requires frequent quality control to prevent contamination. The top companies dealing in molecular diagnostics comprise Thermo Fisher Scientific and Roche Diagnostics.
Application Insights
Why Did the Infectious Disease Testing Segment Dominate the Market?
The infectious disease testing segment dominated the in vitro diagnostics quality controls market with around 27% share in 2025. This is mainly due to the rising need for accurate diagnosis and the rapid adoption of molecular diagnostics (PCR) technology in laboratories to combat the spread of infectious diseases. Infectious disease assays include HIV, hepatitis, respiratory pathogens, and viral outbreaks, which require strict quality control to maintain authenticity.
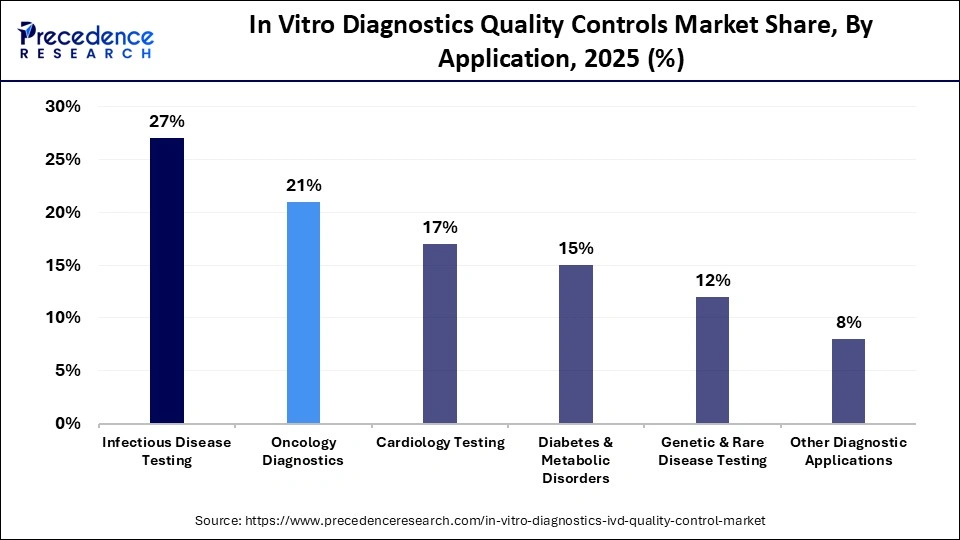
The genetic & rare disease testing segment is expected to grow with the highest CAGR during the forecast period. This is because hospitals are adopting next-gen sequencing and exome sequencing technologies for detecting a wide range of inherited disorders. Genetic disease testing involves stringent validation to deliver analytical accuracy and maintain regulatory standards, which increases the demand for third-party QC solutions.
End User Insights
What Made Hospitals the Leading Segment in the In Vitro Diagnostics Quality Controls Market?
The hospitals segment dominated the in vitro diagnostics quality controls market with around 38% share in 2025, as they are equipped with modern laboratories to provide a wide range of blood tests. Hospitals are engaged in conducting a large volume of testing across several sectors, including hematology, molecular diagnostics, clinical chemistry, and immunoassays, which increases the demand for advanced quality control procedures to check the efficacy of existing equipment.
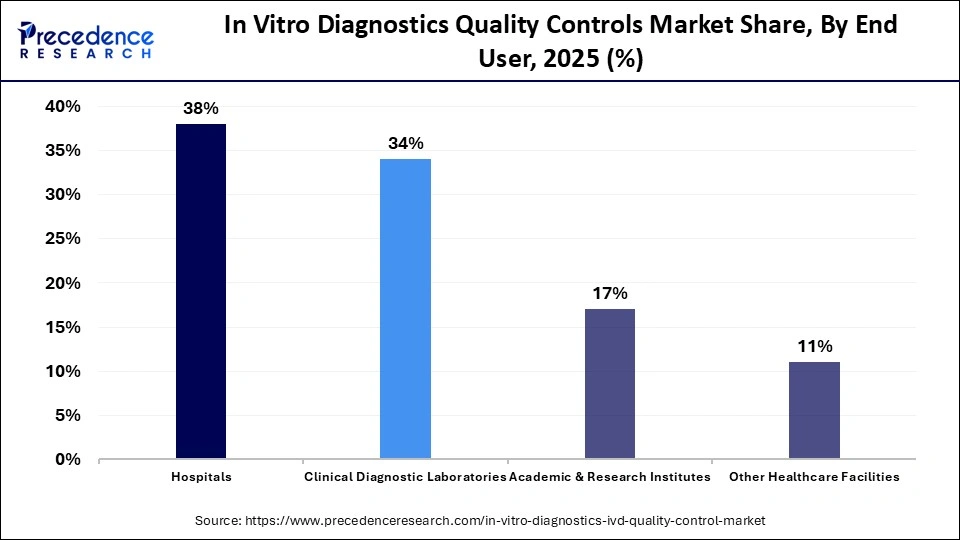
The clinical diagnostic laboratories segment is expected to grow with the highest CAGR during the forecast period, owing to the rise in the number of independent laboratories in different regions, as well as the surging prevalence of infectious diseases globally. These laboratories adopt high-class equipment and advanced automated systems to manage testing of metabolic disorders, which in turn increases the application of quality control services to maintain accuracy.
Regional Insights
What Made North America the Dominant Region in the In Vitro Diagnostics Quality Controls Market?
North America dominated the in vitro diagnostics quality controls market while holding a major share of around 41% in 2025. The region's dominant position in the market is attributed to the availability of highly established healthcare infrastructure, along with the surging cases of cardiovascular diseases. The growing demand for laboratory information management systems (LIMS) and advanced QC solutions launched by prominent companies, including Revvity and Bio-Rad Laboratories, is contributing to industrial development. Additionally, the rapid investment by hospitals to integrate advanced testing solutions, as well as strict regulatory standards set by healthcare organizations, further propel the market expansion.
- In June 2025, Revvity launched three Mimix reference standards for IVD use. This solution is designed to monitor next-generation sequencing (NGS) or droplet digital polymerase chain reaction (ddPCR) assays in the laboratories of the U.S.
U.S In Vitro Diagnostics Quality Controls Market Size and Growth 2026 to 2035
The U.S. in vitro diagnostics quality controls market size is calculated at USD 0.43 billion in 2025 and is expected to reach nearly USD 0.61 billion in 2035, accelerating at a strong CAGR of 3.56% between 2026 to 2035.
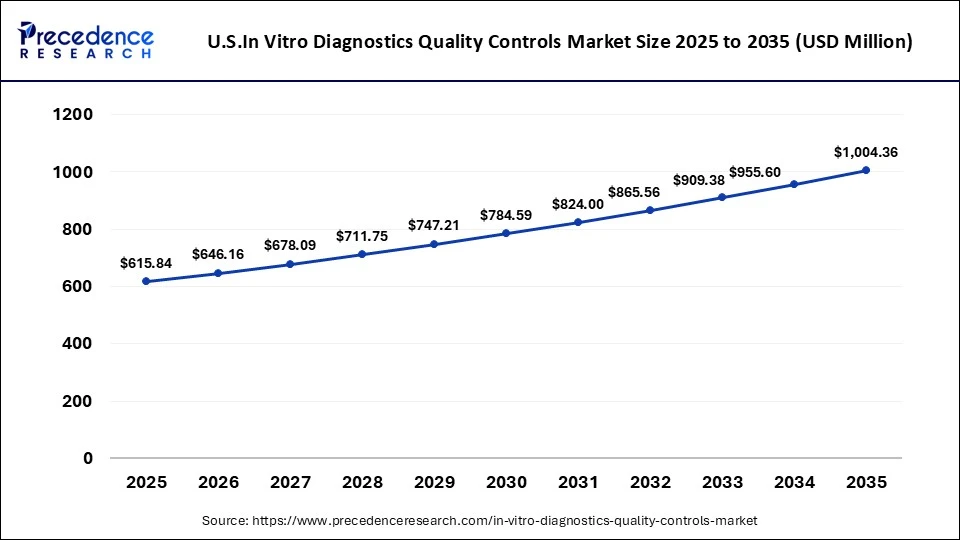
The U.S. is leading the North American in vitro diagnostics quality controls market, owing to the increase in the number of biotech laboratories, coupled with the growing prevalence of prostate cancer and breast cancer. Also, the growing emphasis of the government on opening new research laboratories and the surging adoption of third-party control services by hospitals have driven industrial expansion.
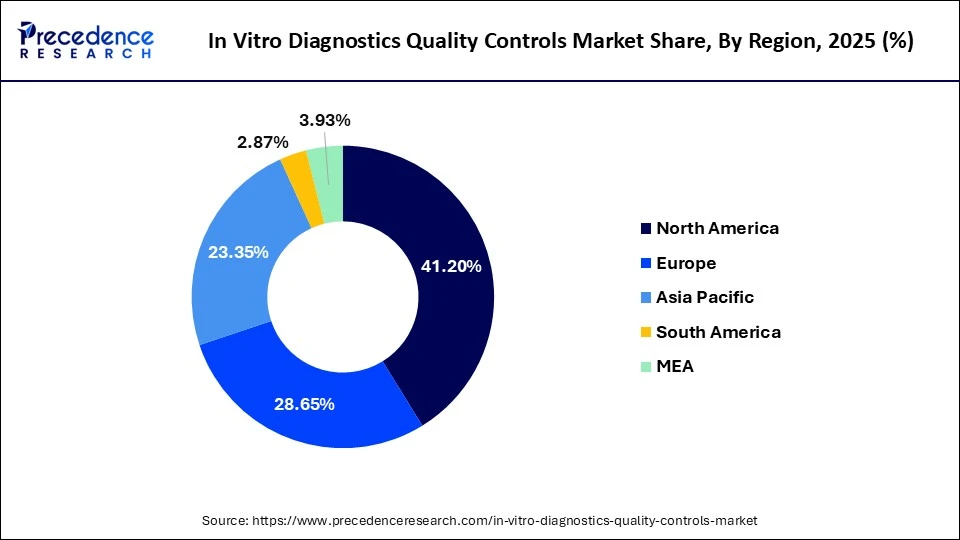
Why is Asia Pacific Expanding with the Highest CAGR in the Market?
Asia Pacific is expected to expand with the highest CAGR during the forecast period, due to the rising preference of consumers to visit independent laboratories regularly for testing their blood samples, coupled with the rapid deployment of advanced software in QC centers for getting accurate results. Companies such as Sysmex Corporation, Seegene Inc., and Eiken Chemical Co., Ltd., which are engaged in delivering IVD QC services, as well as the rapid establishment of public hospitals in remote areas of Japan, Australia, and South Korea, have contributed to the market expansion.
- In November 2025, the Western Australian Government announced to invest around US$ 1.5 billion. This investment is made to construct three public hospitals in different parts of Australia.
China Market Analysis
China is a major contributor to the Asia Pacific in vitro diagnostics quality controls market, owing to the digital transformation across the healthcare sector, along with the outbreak of different types of viral diseases. Additionally, the rapid investment by the laboratories to adopt AI-based QC solutions to check the efficacy of testing equipment has bolstered the market expansion.
Top Companies in the In Vitro Diagnostics Quality Controls Market
- Siemens Healthineers AG
- Abbott
- Bio-Rad Laboratories
- Danaher
- Randox Laboratories Ltd
- Helena Laboratories Corporation
- Thermo Fisher Scientific Inc
- SeraCare Life Sciences Inc
- BD
- Merck KGaA
- QIAGEN
- Hologic, Inc.
- Bio-Techne
- Fortress Diagnostics
- Microbiologics
- Trina Bio Reactive AG
- ConeBioproducts
- Biorex Diagnostics
- F. Hoffmann-La Roche Ltd
- Sysmex Corporation
- QuidelOrtho Corporation
Recent Developments
- In December 2025, Leica Biosystems launched a pathology portfolio. This pathology portfolio consists of Aperio GT 180 DX scanner, Aperio CS5 DX scanner, and Aperio iQC DX software that finds application across the in-vitro diagnostics sector.
(Source: https://www.prnewswire.com) - In October 2025, Fujifilm launched the FW500 clinical chemistry analyzer. The FW500 clinical chemistry analyzer is designed to support diagnostic laboratories with enhanced precision and superior efficiency. (Source: https://www.digitalhealthnews.com)
- In May 2025, Codix Pharma opened a new production unit in Nigeria. This new manufacturing center is inaugurated to produce a wide range of in-vitro diagnostic (IVD) kits for the end-users of Africa. (Source: https://guardian.ng)
Segments Covered in the Report
By Product & Service
- Quality Control Products (Reagents, Controls)
- Serum/plasma-based controls
- Whole blood controls
- Data Management Solutions
- Quality Assurance Services
By Manufacturer Type
- Third-Party Controls
- Original Equipment Manufacturer (OEM) Controls
By Technology
- Immunoassay Diagnostics
- Clinical Chemistry Testing
- Molecular Diagnostics
- Hematology Diagnostics
- Microbiology Diagnostics
- Coagulation & Hemostasis Testing
By Application
- Infectious Disease Testing
- Oncology Diagnostics
- Cardiology Testing
- Diabetes & Metabolic Disorders
- Genetic & Rare Disease Testing
- Other Diagnostic Applications
By End User
- Hospitals
- Clinical Diagnostic Laboratories
- Academic & Research Institutes
- Other Healthcare Facilities
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
