Immunoprecipitation is a powerful technique used to isolate and analyze specific proteins with high precision. It plays a vital role in molecular biology, enabling researchers to study protein interactions, biomarkers, and disease mechanisms.
Not every discovery begins with complexity; sometimes, it starts by quietly pulling one protein out of the crowd and finally paying attention to it.
Immunoprecipitation is a fundamental method involved in the isolation and purification of specific proteins and is a useful tool in this field in the disciplines of molecular biology and immunology. Its philosophy is based on the outstanding specificity of antibody-antigen interactions, allowing the precise capture of target molecules amidst a milieu of other biomolecules. In the present review, we describe the fundamentals, procedures, and essentials of the immunoprecipitation method.
Principles of Immunoprecipitation
Immunoprecipitation relies on the remarkable specificity of antibodies to their respective antigens. These Y-shaped proteins are antibodies produced by the immune system in response to foreign entities known as antigens present in the body. Each antibody molecule has two identical binding sites at the ends of its Y structure, allowing for the precise identification and attachment to target antigens.
The immunoprecipitation method consists of the formation of immune complexes through the interaction between particular antibodies and their specific antigens. It is performed in carefully optimized conditions, typically in a buffered solution to mimic physiological conditions. They are isolated using solid support matrices that bind to the constant region of antibody molecules upon forming these antibody-antigen complexes.
Step-by-Step Explanation: How it Actually Happens
- Sample Preparation: Setting the Stage: Lysis of cells or tissues releases the protein content from them. Although this is a simple phase, it implies all succeeding steps are based on it. High stress may interfere with protein interactions; lower stress could permit undesirable contaminants to remain.
- Antibody Binding: The Moment of Specificity: An antibody is incorporated into the lysate to methodically search for and bind its target. Binding occurs via precise molecular interactions upon recognition. This is the critical step, and an antibody that is not properly selected does not fail overtly but quietly sabotages it.
- Capture Using Beads: Making the Invisible Visible: These have been coated with Protein A or G to specifically bind the antibody-protein complex. These beads act as supports that allow for the physical separation of the complex from its environment. Without this stage, much would go unseen.
- Washing: The Art of Letting Go: The washing step rids the complex of non-specific proteins loosely associated with the complex. Here is the compromise too gentle may leave deposits of impurities, and too much rigor can give your protein a chance of slipping away.
- Elution: Releasing the Target: Eventually, conditions like pH or ionic strength are adjusted to pull that protein away from its molecule-bound antibody to be removed for subsequent analysis.
What is the Immunoprecipitation Market Size in 2026?
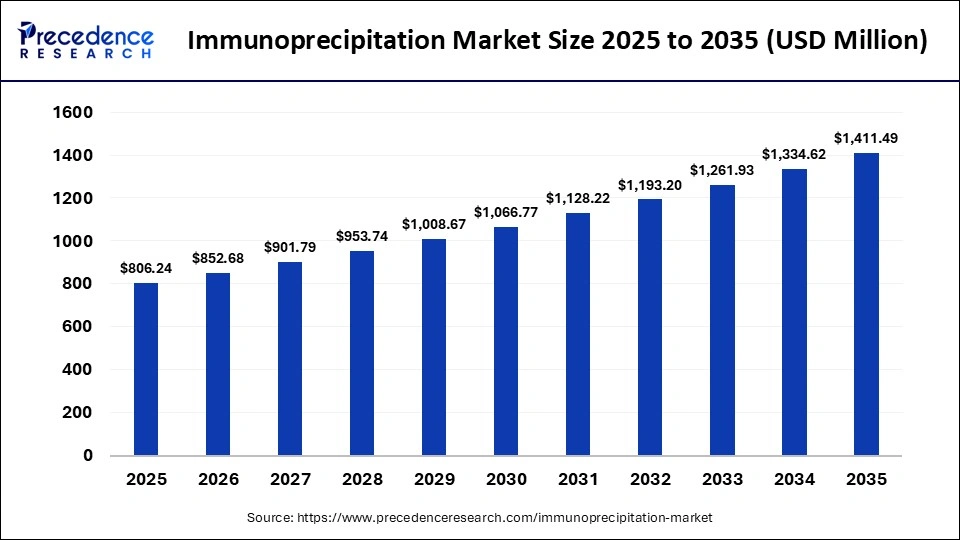
The global immunoprecipitation market was valued at USD 806.24 billion in 2025 and is projected to grow from USD 852.68 billion in 2026 to approximately USD 1,411.49 billion by 2035, registering a CAGR of 5.76% during the forecast period from 2026 to 2035. As researchers increasingly focus on complex protein interactions, immunoprecipitation is becoming an essential technique across a wide range of applications, including modern proteomics, molecular diagnostics, and targeted drug development.
Get a Free Sample Report with Key Market Trends: https://www.precedenceresearch.com/sample/8209
Crucial Step in Immunoprecipitation: Antibody Binding
The first important step after preparation is the process of targeted antigen attachment. This antibody to antigen interaction builds its building block for use as part of immunoprecipitation, and is responsible for selectivity and success in protein acquisition.
- Antibody Selection: To select a primary antibody that can accurately detect the target protein, it is necessary to choose a primary antibody that can recognize the target protein in the sample over a wide range of proteins. Choose antibodies that are validated to be used in immunoprecipitation and characterized based on specificity and affinity.
- Incubation: The sample lysate is incubated with the selected antibody under gentle agitation at an appropriate temperature for a specified duration. This allows sufficient time for the antibody to bind to its cognate antigen and form stable immune complexes.
- Crosslinking: In certain applications, particularly when working with weakly interacting proteins, crosslinking agents such as formaldehyde or glutaraldehyde may be used to stabilize the antibody-antigen complexes and prevent dissociation during subsequent washing steps.
Variants of Immunoprecipitation
- Co-Immunoprecipitation (Co-IP): Co-Immunoprecipitation is a technique in which a single molecule can be isolated while its respective interaction partners are captured. If protein A binds protein B within a cell, isolating A generally takes B along. This property makes Co-IP critical to study protein–protein interactions, describing possible signaling cascades, and understanding molecular complexes.
- Chromatin Immunoprecipitation (Ch-IP): Interactions of Proteins and DNA: Chromatin Immunoprecipitation expands this notion to include the genome by identifying the sites where proteins linked to DNA interact with that DNA. The process of isolation is done by cross-linking the protein-DNA complexes and identifying them with a specific antibody. As a result, Ch-IP has greatly deepened our understanding of gene regulation.
- RNA Immunoprecipitation (R-IP): Engaging with RNA: Immunoprecipitation in RNA is focused on protein interactions with RNA molecules. This also illustrates how these proteins affect RNA stability, translation pathways, and cell localization.
- Cross-Linking Methods (CL-IP and Variants): Detailed Mapping: Extensive, sophisticated methods, including the CLIP and PAR-CLIP, combine immunoprecipitation, sequencing, and interaction with ribonucleotide site-matching techniques for identification at the near-nucleotide level.
Why Immunoprecipitation Still Matters
Because of this, immunoprecipitation is considered a very important technology in the world of high-throughput omics and automation. However, immunoprecipitation continues to have a lot of vitality for a few reasons.
- Unmatched Specificity: Immunoprecipitation uses specific-target recognition, not general characteristic recognition. This allows researchers to examine proteins in their own native environments while preserving their interactions and modifications.
- Adaptability Across Various Applications: Whether verifying protein expression, investigating post-translational modifications, or exploring interaction networks, immunoprecipitation readily adapts to suit a spectrum of experimental demands.
- Suitability for Subsequent Analyses: The immunoprecipitation yields, whether in biological or molecular aspects, can be easily studied using western blotting, mass spectrometry, or sequencing. This is the point where it falls along a tight link between traditional biochemistry and contemporary molecular biology.
Applications: Where Immunoprecipitation Shows Its Strength
- Discovering Disease Biomarkers: Immunoprecipitation helps in isolating proteins present in low quantities during diseases that would otherwise be missed. This allows scientists to pick up on subtle molecular changes, particularly important in cancer or autoimmune diseases, where early biomarkers often remain undetected.
- Validation of Drug-Target Interactions: Immunoprecipitation gives concrete evidence of a drug's actions on the proteins of its target rather than simply assuming that drugs hit their target. This improves therapeutic intervention informed by more accurate, unverified information.
- Examining Post-Translational Modifications: Few proteins maintain their original structure, so immunoprecipitation has allowed isolation of modified forms (e.g., phosphorylated, ubiquitinated, or acetylated) with less noise than other methods, allowing a simpler identification of how these minor chemical modifications can affect function.
- Investigating Viral-Host Interactions: Indeed, during an infection, when viruses can take on host proteins for survival, immunoprecipitation plays a pivotal role in reflecting these transient interactions and revealing how pathogens utilize cellular mechanisms, as well as providing opportunities for potential therapeutic intervention
- Studying Epigenetic Regulation through Chromatin Analysis: By using chromatin immunoprecipitation to isolate DNA-bound proteins and examine gene regulation processes, they can find not so much whether those protein regulation patterns follow some sequence of DNA as what kind of environmental state a gene can be found in.
Strengths and Limitations: A Balanced View
Strengths
- Very specific results as antibody-antigen recognition occurs.
- Opportunity of studying proteins under near-native conditions.
- Versatility in several experiments.
Limitations
- Contaminants may be introduced with non-specific binding.
- The results are largely sensitive to antibody quality.
- Optimization can be time-consuming.
- Some protein interactions might be lost during washing steps.
The Subtle Variables That Change Everything
- Antibody choice: Not all antibodies are created equal; high specificity and high affinity matter a great deal.
- Bead choice: Magnetic beads are easier to handle-agarose beads have greater binding capacity.
- Buffer composition: Magnetic beads are easier to handle-agarose beads have greater binding capacity.
- Temperature and time: These affect binding efficiency and background noise.
Emerging Trends: Where Immunoprecipitation is Heading
- Convergence with Mass Spectrometry: Immunoprecipitation has now matured from being a purely isolated process to one not only involving mass spectrometry to analyze entire protein complexes in one go. This move pushes the method away from a targeted approach towards exploratory research, using data to generate insights instead of building hypotheses.
- Shift Toward Magnetic Bead-Based Automation: In the era of traditional immunoprecipitation, magnetic bead methods offer improvements for separating components and reducing variability. The change makes the technique more reproducible, less dependent on specific skills, and is more appropriate for automated workflows that don't rely on exact execution or timing.
- Rise of Recombinant Antibodies and Nanobodies: Engineered alternatives, including recombinant antibodies and nanobodies, are becoming increasingly complementary-and occasionally replacements-for traditional antibodies. Such advancements lead to an improvement in specificity, batch quality, and stability, and address long-standing issues associated with uneven antibody performance in immunoprecipitation.
- Integration into Multi-Omics Workflows: Immunoprecipitation is gradually integrating into other proteomic, transcriptomic, and genomic approaches for researchers. This integration supports scientists in associating isolated protein activity with larger cellular processes instead of studying it in a vacuum.
- Improved Cross-Linking and Stabilization Techniques: Advances in the field of cross-linking chemistry allow researchers to maintain weak or transient interactions at work, which might fade away during the washing process. This improvement permits immunoprecipitation techniques to provide a more accurate insight into cellular activities at any particular moment.
The Future of Immunoprecipitation
Immunoprecipitation does not evolve via a complete overhaul, but rather through improvement. We are creating an improvement process that is more streamlined, more precise, and less dependent on laboratory personnel’s subjective judgment. With better antibody quality and greater efficiency, together with stronger integration with mass spectrometry, it will merge comfortably into broader workflows.
The evolution isn’t the basic idea so much as the reassurance around it; there’ll be fewer worries about specificity and trade-offs with respect to sensitivity. Additionally, immunoprecipitation will become more common in clinical settings where small sample sizes can be meaningful. Going forward, this technique won’t be seeking to increase its own reach, but rather will work on the application of what it can do and will be working at much higher levels of performance, reliability, and consistency.
Expert Advise
Our experts suggest that immunoprecipitation is an emerging trend in the biotechnology sector, with the growing demand for personalized medicines, the development of targeted therapeutics, and increasing investments for research purposes. Researchers focus on developing novel immunoprecipitation methods with the integration of AI, introducing automation, minimizing tedious tasks, and facilitating data analysis. They also prioritize antibody specificity and affinity, as poor-quality antibodies are common causes of non-specific binding and inconsistent results. In addition, robotic liquid handling systems, miniaturization, and integration with multi-omics technologies drive the future of immunoprecipitation.
About the Authors

Aditi Shivarkar
Aditi, Vice President at Precedence Research, brings over 15 years of expertise at the intersection of technology, innovation, and strategic market intelligence. A visionary leader, she excels in transforming complex data into actionable insights that empower businesses to thrive in dynamic markets. Her leadership combines analytical precision with forward-thinking strategy, driving measurable growth, competitive advantage, and lasting impact across industries.

Aman Singh
Aman Singh with over 13 years of progressive expertise at the intersection of technology, innovation, and strategic market intelligence, Aman Singh stands as a leading authority in global research and consulting. Renowned for his ability to decode complex technological transformations, he provides forward-looking insights that drive strategic decision-making. At Precedence Research, Aman leads a global team of analysts, fostering a culture of research excellence, analytical precision, and visionary thinking.

Piyush Pawar
Piyush Pawar brings over a decade of experience as Senior Manager, Sales & Business Growth, acting as the essential liaison between clients and our research authors. He translates sophisticated insights into practical strategies, ensuring client objectives are met with precision. Piyush’s expertise in market dynamics, relationship management, and strategic execution enables organizations to leverage intelligence effectively, achieving operational excellence, innovation, and sustained growth.





 Request Consultation
Request Consultation

