Johnson & Johnson Expands Stroke Care Portfolio with Cereglide System to Enhance Precision in Clot Removal
Johnson & Johnson has expanded its stroke care portfolio with the introduction of the Cereglide 42 and Cereglide 57 aspiration catheters, alongside the Innerglide 7 delivery aid, all designed to improve access and remove distally located clots more consistently. These innovations align with the company’s aspiration-first approach, further enhancing physician capabilities in stroke treatment and improving patient outcomes in the U.S. market.
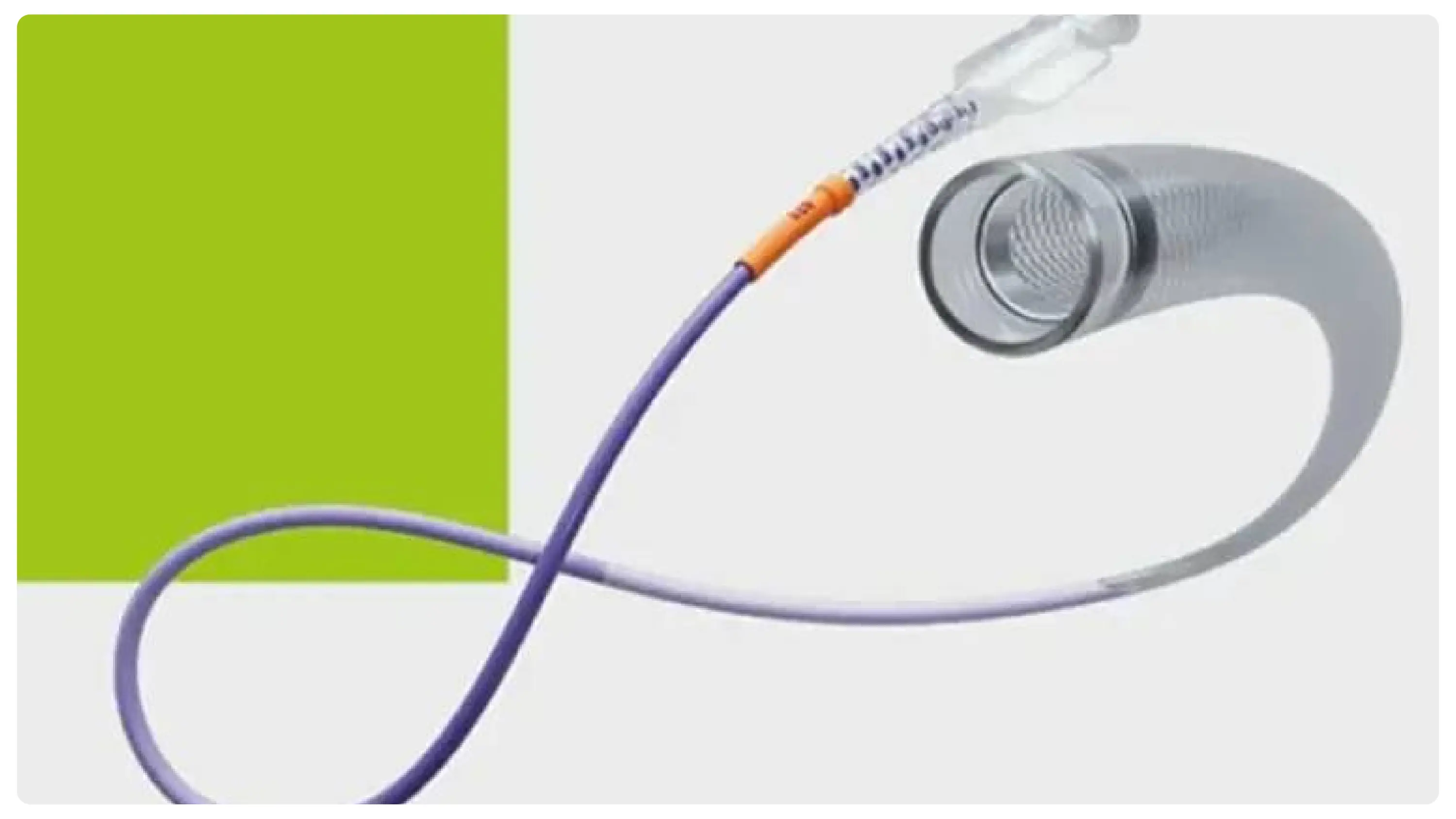
Enhancing Neurovascular Access with Advanced Device Engineering
The Cereglide system is an introduction that is indicative of an active attempt to solve the long-standing problems in the neurointerventional capabilities. The design of these catheters is such that they are easily incorporated into the current clinical processes, and the procedures would have to be adjusted less. These include a hydrophilic coating, which minimizes friction during navigation, and radiopaque tip markers.
This results in augmented fluoroscopic visibility, allowing placement in a difficult anatomical area with precision. Furthermore, the system's incorporation of sophisticated trackability technology facilitates sound access to the M2 portion of the middle cerebral artery, which is a critical goal in most stroke treatment.
According to the Precedence Research, the thrombus aspiration catheter market was valued at USD 2.20 billion in 2025 and is expected to reach USD 4.66 billion by 2035, expanding at a CAGR of 7.80% from 2026 to 2035, driven by increasing cases of stroke and demand for minimally invasive procedures.
Christian Cuzick, Worldwide President of Neurovascular at Johnson & Johnson MedTech, stated, “The expansion of the Cereglide portfolio reflects our commitment to addressing real-world procedural challenges in stroke care. By delivering consistent performance across device sizes and simplifying decision-making for physicians, we aim to improve treatment efficiency and ultimately enhance patient outcomes.”
Industry Impact and Future Outlook
These devices are introduced into the international market at a time when there is an increasing interest globally in enhancing stroke care outcomes. Especially to the management of ischemic stroke, which continues to be a major cause of disability and mortality around the world. Cereglide 42 and 57 are specifically crafted to be used with aspiration-first thrombectomy methods. They are gradually replacing thrombectomy as a first-line treatment in stroke patients.
The overall increase in the use of such technologies indicates a tendency in treating neurovascular patients toward more sophisticated and patient-centered approaches. The requirements for equipment with accuracy, trustworthiness, and user-friendliness are likely to increase. New technologies such as the Cereglide system have thus become a pivotal point in determining the future of stroke treatment. Additionally, the Cereglide portfolio expansion is a positive move towards this end, as it makes it clear that Johnson & Johnson could be considered as one of the leading innovators in neurovascular care.