What is the Specimen Collection Cards Market Size?
The global specimen collection cards market size accounted for USD 502.11 million in 2025 and is predicted to increase from USD 528.82 million in 2026 to approximately USD 843.15 million by 2035, expanding at a CAGR of 5.32% from 2026 to 2035. The market is driven by the expansion of modern healthcare infrastructure, the increasing prevalence of infectious diseases worldwide, the growing need for newborn screening, and ongoing technological advancements in specimen collection cards.
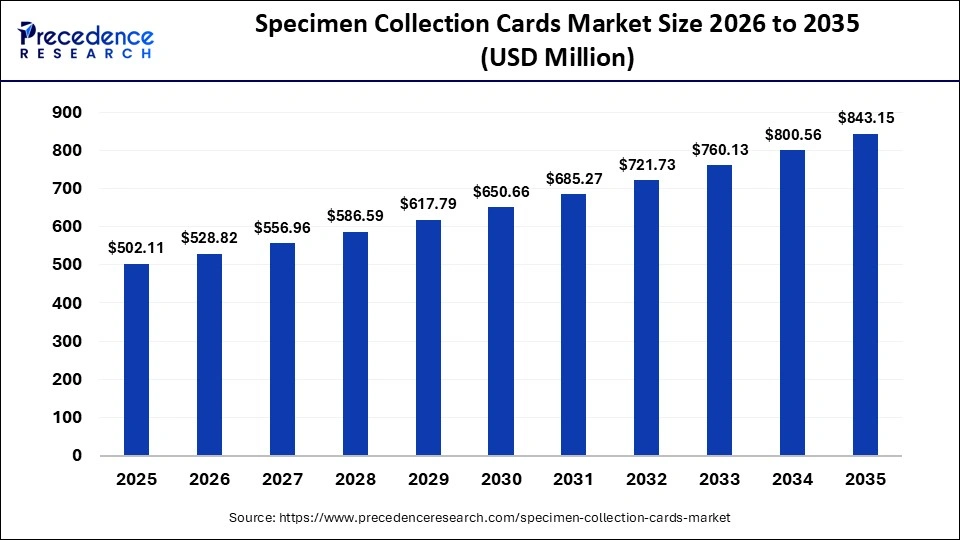
Market Highlights
- North America dominated the market, holding the largest market share of 34.5% in 2025.
- The Asia Pacific is expected to grow at the fastest CAGR of 6.1% between 2026 and 2035.
- By specimen type, the blood segment held the largest market share of 89% in 2025.
- By specimen type, the saliva segment is expected to grow at a remarkable CAGR of 6% between 2026 and 2035.
- By material / card substrate, the cotton & cellulose-based segment contributed the largest market share of 60% in 2025.
- By material / card substrate, the fiber-based / synthetic segment is growing at a CAGR of 20% between 2026 and 2035.
- By application, the newborn screening (NBS) segment accounted for the highest market share of 22.6% in 2025.
- By application, the forensics & research segment is projected to expand at the fastest CAGR between 2026 and 2035.
- By distribution channel, the offline segment captured the biggest share of 80% in the Specimen collection cards market in 2025.
- By distribution channel, the online segment is expanding at a solid CAGR between 2026 and 2035.
- By end-use sector, the hospitals & clinics segment held the major market share of 64% in 2025.
- By end-use sector, the research institutions & others segment is expected to grow at a noteworthy CAGR of 12% between 2026 and 2035.
Rise of Specimen Collection Cards: Spearheading the Future of Diagnostics
Specimen collection cards are specifically designed absorbent cards or filter-paper substrates used to collect, stabilize, store, transport, and analyze biological specimens such as blood, saliva, urine, buccal cells n diagnostics, screening, research, forensics, and therapeutic monitoring. They enable ambient-temperature transport, long-term sample stability, reduced bio-hazard risk, and simplified logistics, especially for remote, decentralized, or point-of-care settings.
How are AI-driven innovations reshaping the specimen collection cards market?
As technology continues to advance, artificial Intelligence (AI) integration is greatly accelerating the growth of the specimen collection cards market by reducing human error, boosting efficiency, and enabling quicker, more reliable diagnostics. AI-powered algorithms and machine vision can quickly evaluate the quality of a specimen immediately after collection, including checking fill volume, detecting hemolysis and clotting in blood samples, and accurately confirming the absence of contaminants to ensure the sample is suitable for testing. AI also helps manage the large datasets produced, facilitating smooth identification and rapid sample tracking, which leads to fewer misidentification errors.
Specimen Collection Cards Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 502.11 Million |
| Market Size in 2026 | USD 528.82 Million |
| Market Size by 2035 | USD 843.15 Million |
| Market Growth Rate from 2026 to 2035 | CAGR of 5.32% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Specimen Type, Material, Application, Distribution Channel, End-Use Sector, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Specimen Collection Cards Market Segment Insights
[[segment_insights]]
Specimen Collection Cards Market Regional Insights
[[regional_insights]]
Regulatory Landscape
| Country/Region | Regulatory Body | Key Regulations | Focus Areas | Notable Notes |
| U.S. | U.S. Food and Drug Administration (FDA) | The Federal Food, Drug, and Cosmetic Act (FD&C Act) | Device Classification, Convenience Kits, Performance, and Validation | Under the FDAs regulatory the specimen collection cards are regulated as medical devices, generally falling into the lowest-risk Class I category. The FDA also monitors market trends such as the rise of home-based testing and miniaturization. |
| European Union | European Commission, the Medical Device Coordination Group (MDCG) | Regulation (EU) 2017/746 on in vitro diagnostic medical devices (IVDR) | CE Marking, Risk-Based Classification, Quality Management System (QMS), Technical Documentation, Registration and Traceability (EUDAMED), and Labeling and Instructions for Use | Manufacturers and economic operators in the EU specimen collection cards market must focus on the following regulatory areas. |
| India | Central Drugs Standard Control Organization (CDSCO) | The Drugs and Cosmetics Act, 1940 | Mandatory Licensing, Quality Management Systems (QMS), Performance Evaluation, and Post-Market Surveillance | The Central Drugs Standard Control Organization (CDSCO), which functions under the Ministry of Health and Family Welfare, ensures that manufacturers or importers are subject to a streamlined licensing process. |
Specimen Collection Cards Market Companies
[[market_company]]
Other Major Companies
- Ahlstrom (Ahlstrom-Munksjo)
- ARCHIMED Life Science GmbH
- GenTegra LLC
- FortiusBio
- CENTOGENE N.V.
- Whatman (Cytiva)
- Shimadzu Corporation
- Spot On Sciences
- Sedia Biosciences Corporation
- HemaXis
Recent Developments
- In February 2025, Ahlstrom, a global leader in fiber-based specialty materials, announced that its biological sample collection cards had been officially classified as FDA Class 1 medical devices. This designation by the US Food and Drug Administration (FDA) affirms the safety, reliability, and compliance of GenSaver 2.0, GenSaver Color 2.0, AutoCollect, 5 circles, BioSample, HemaSep Strip, HemaSep Punch, and Lipid Saver sample collection cards with the highest industry standards. These products are designed for the secure and efficient collection, transportation, and ambient-temperature storage of biological samples.(Source: https://www.ahlstrom.com)
- In February 2025, Tufts researchers collaborate with scientists in South Africa to test a more precise medical device that more accurately measures HIV viral loads and detects drug-resistant mutations. In a clinical pilot with 75 South African patients living with HIV, the Tufts research teams device, called a plasma spot card, is used. While this technology helps track someones adherence to their drug regimen or monitor disease progression, the most frequently used devices dont control how much blood they collect, which leads to inaccurate readings of a persons infection.(Source: https://now.tufts.edu)
Specimen Collection Cards Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
