What is the Cancer Gene Therapy Market Size?
The global cancer gene therapy market size was estimated at USD 4.14 billion in 2025 and is projected to hit around USD 22.32 billion by 2035, poised to grow at a CAGR of 18.35% during the forecast period from 2026 to 2035.
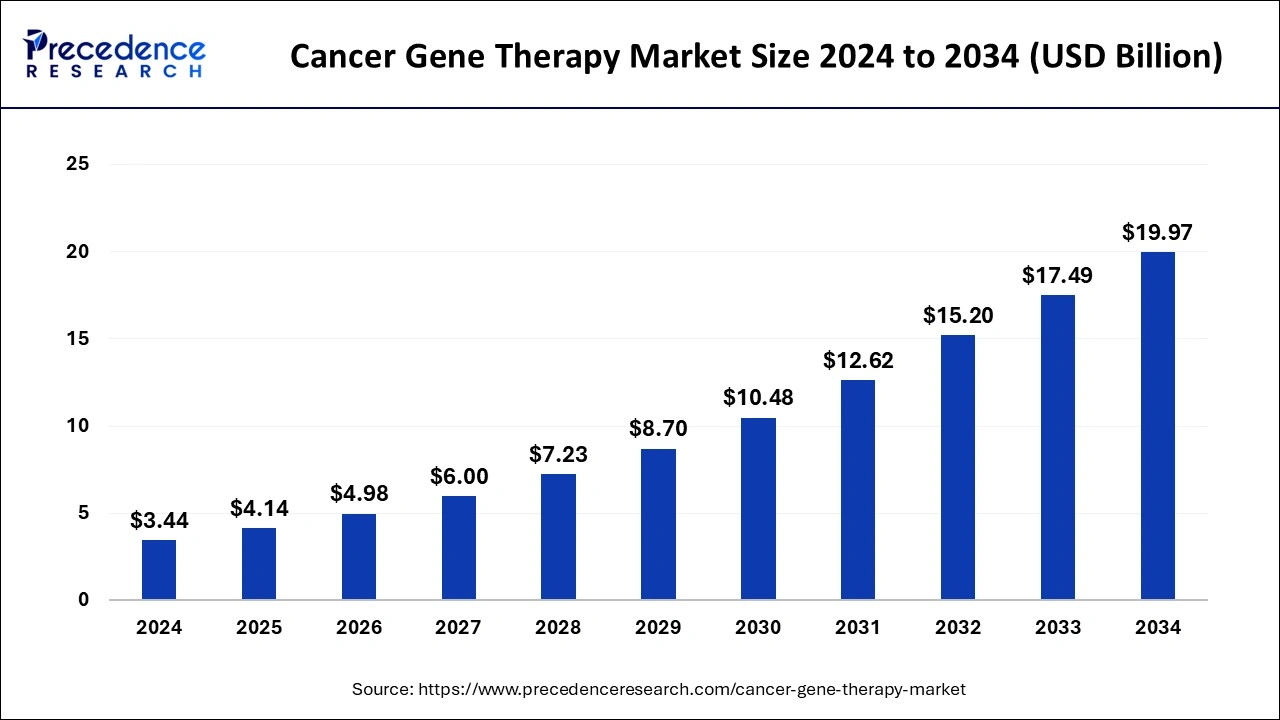
Cancer Gene Therapy Market Key Takeaways
- North America has generated more than 62% of revenue share in 2025.
- By Therapy, the gene-induced immunotherapy segment has contributed more than 41% of the revenue share in 2025.
- By End-User, the biopharmaceutical companies segment had the biggest revenue share of around 46% in 2025.
Market Overview
Cancer is a group of diseases that involve abnormal cell growth which can spread to respective parts of the body. Cancer can spread throughout the human body. Gene therapy is a kind of treatment in which the genes that are not normal or are missing in the patient's cells are replaced with normal genes. Cancer gene therapy is a technique for treating cancers where the therapeutic DNA is introduced in the gene of the individual suffering from cancer.
Due to a high success rate in preclinical as well as clinical trials, cancer gene therapy is gaining high popularity all over the world. There are numerous techniques utilized in cancer gene therapy. In one of the gene therapy techniques, either the mutated gene is replaced with a healthy gene, or the gene is inactivated if its function is abnormal. In a newly developed technique, new genes can be introduced in the body of the patient to help fight against cancer cells.
Further, the ongoing extensive research and development (R&D) strategies implemented by biopharmaceutical firms for producing novel therapeutic drugs are driving the market growth notably.
The market players can aim towards expansions, collaborations, joint ventures, acquisitions, and partnerships to advance capabilities in gene therapy. This would help in yielding effective therapeutic drugs for treating different kinds of cancers. In April 2022, GSK plc announced the acquisition of Sierra Oncology for £1.6 billion ($1.9 billion). This acquisition would help GSK plc in enhancing its capabilities with respect to targeted therapies for treating rare forms of cancer.
Biotechnology firms are evaluating novel gene therapy vectors for increasing levels of protein production/gene expression, reducing immunogenicity, and improving durability.
The top cancers in terms of the count of new cases in 2020 all over the world were Lung Cancer (2,206,771 cases), Breast Cancer (2,261,419 cases), Prostate Cancer (1,414,259 cases), Colorectal Cancer (1,931,590 cases), Stomach Cancer (1,089,103 cases), and Liver cancer (905,677 cases). In 2018, there were around 134,632 new cancer cases and 89,042 cancer-related fatalities. Breast and liver cancers were among the most common tumors in terms of incidence and mortality. The high prevalence of breast cancer cases enhances the scope for CRISPR/Cas9-based gene editing for breast cancer therapy and VISA-claudin4-BikDD gene therapy.
Market Scope
| Report Coverage | Details |
| Growth Rate from 2026 to 2035 | CAGR of 19.23% |
| Market Size in 2025 | USD 4.14 Billion |
| Market Size by 2035 | USD 22.32 Billion |
| Largest Market | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | By Therapy and By End-User |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Cancer is a leading cause of death all over the world. As per the Singapore Cancer Registry Annual Report 2019, there were 78,204 cancer cases reported in Singapore during the period from 2015 to 2019. Out of these 78,204 cancer cases, 12,940 females and 15,605 males died of cancer. Thus, such a high count of fatalities has created considerable scope for gene therapies needed for treating cancer.
The growth of the cancer gene therapy market is attributable to key factors such as the increasing demand for gene therapy and the rising incidence of cancer cases across the world. Also, with the advancements in gene therapy, it is expected to become a preferred choice for the treatment of all kinds of cancers as well as neoplastic disorders.
However, the high cost associated with respective gene therapies combined with undesirable immune responses is anticipated to hinder the cancer gene therapy market growth during the forecast period.
Moreover, in order to gain further access to the regional markets, companies can undertake various strategic initiatives including the expansion of existing product portfolio. These initiatives may result in more intensive rivalry over the forecast period. Additionally, rapidly evolving cancer gene therapies can make the existing cancer gene therapies obsolete very soon. This further intensifies the market competition among the prominent players.
Since the count of cancer gene therapy providers (suppliers) is much less in comparison to the count of buyers, the bargaining power of suppliers is notably higher as compared to the bargaining power of buyers. Owing to considerable profit margins, the threat of new entrants in the cancer gene therapy market is moderate as of now.
As per our research, the global gene therapy market was valued at $ 7.74 billion in 2023 and is predicted to account for over $38.76 billion by 2032. The global gene therapy market will grow at a CAGR of 20.2% from 2023 to 2032. Thus, with growing cancer cases and high growth in the overall gene therapy market, the cancer gene therapy market is expected to boost accordingly.
Segment Insights
Therapy Insights
Based on therapy, the global cancer gene therapy market is segmented into gene-induced immunotherapy, oncolytic virotherapy, and gene transfer. The gene-induced immunotherapy segment held the largest market share in 2025. The gene-induced immunotherapy segment is expected to hold a significant market share during the forecast period.
The dominant share of the gene-induced immunotherapy segment is attributable to research activities aiming to decrease the prevalence of numerous forms of cancer by strengthening the immune system.
Most of the gene therapies for the treatment of cancers are structured on the basis of immunotherapy elements. For example, PROVENGE (by Dendreon Corporation) is an autologous cellular immunotherapy that is aimed at stimulating the patient's immune system against prostate cancer.
Considering the level of efficacy and the favorable outcomes provided by oncolytic virotherapy, it is predicted to grow at the highest compounded annual growth rate (CAGR) over the study period. Among the most effective cancer treatments, oncolytic virotherapy has emerged as one of the top therapies. Oncolytic viruses are able to combat cancer cells without affecting healthy cells. Furthermore, lucrative funding for the research on oncolytic virotherapy is augmenting the oncolytic virotherapy segment growth. The researchers at the Center for Nuclear Receptors and Cell Signaling at the University of Houston got a grant of $1.8 million from the National Institutes of Health to work on oncolytic virotherapy in July 2022.
End User Insights
Based on end user, the global cancer gene therapy market is segmented into hospitals, biopharmaceutical companies, diagnostic centers, research institutes, and others. The biopharmaceutical companies segment had the highest revenue share in 2025. The biopharmaceutical companies segment is expected to dominate the market during the study period.
This growth of the biopharmaceutical companies segment is attributable to the rising global prevalence of distinct kinds of cancers owing to various environmental, hereditary, and lifestyle risk parameters.
The growing adoption of elemental gene therapy alternatives by biopharmaceutical firms to formulate cancer therapeutic regimes is propelling segment growth. Many novel gene therapies are under distinct phases of trials and biopharmaceutical companies are working to market them in different continents all over the world.
The biopharmaceutical companies segment is also estimated to be the fastest-growing segment over the forecast period. The rising prevalence of malignant tumors is a significant factor that is accelerating the market growth. Also, the growing interest in oncology therapeutics research and development (R&D) activities has led to an increase in the count of U.S. food and drug administration approvals of gene therapy drugs. Abecma, Tecratus, and Kymriah are some of the recently approved FDA gene therapy medications.
Regional Insights
What is the U.S. Cancer Gene Therapy Market Size?
The U.S. cancer gene therapy market size was estimated at USD 1.79 billion in 2025 and is projected to surpass around USD 10.04 billion by 2035 at a CAGR of 18.82% from 2026 to 2035.
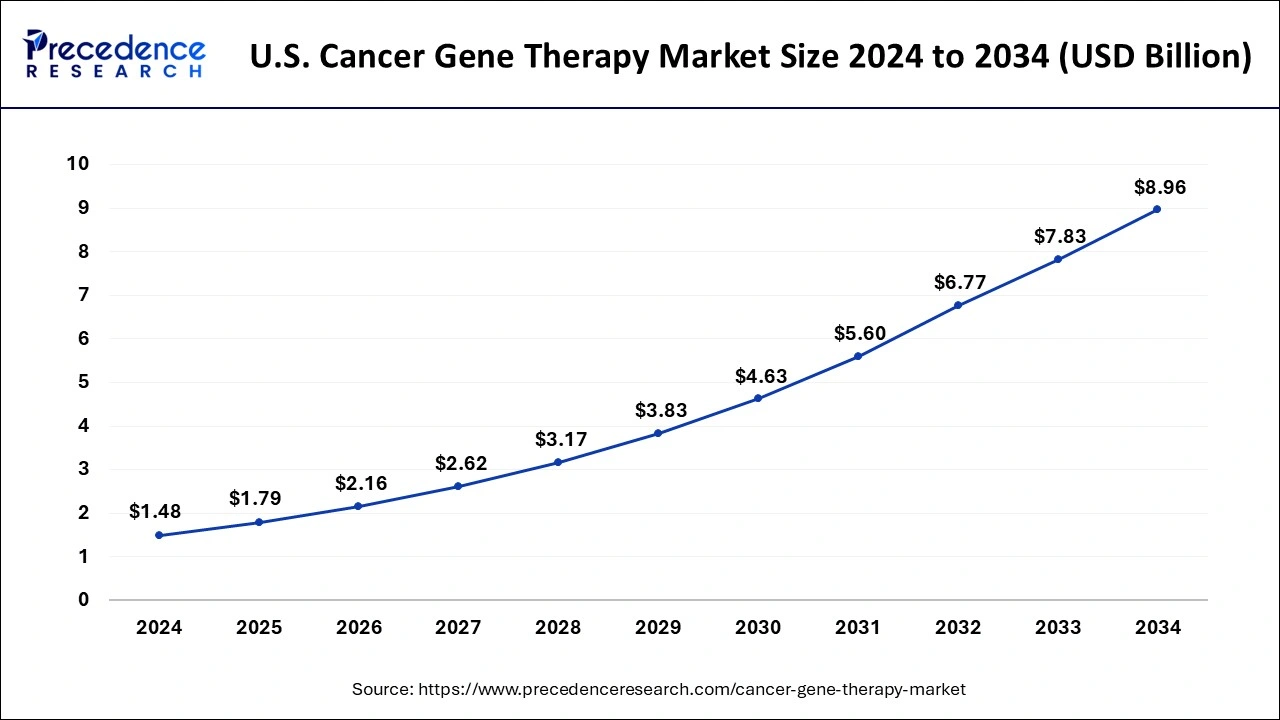
The cancer gene therapy market is spread across North America, Europe, Asia Pacific (APAC), the Middle East and Africa, and Latin America. North America held the largest share of the global cancer gene therapy market in 2025. The U.S. held the largest share followed by Canada and Mexico in 2023. North America is the most technically advanced continent owing to the presence of nations with high gross domestic product (GDP), the existence of favorable economic policies, an ecosystem of well-established biopharmaceutical infrastructure, and early adoption of advanced medical technologies.
U.S. Market Trends
The cancer gene therapy market is dominated by the U.S. due to the well-developed healthcare infrastructure, extensive research and development, and the early adoption of CAR-T and TIL therapies. Growth is supported by powerful FDA approvals, powerful reimbursement models, and plenty of clinical trials. The region enjoys huge biopharma investments and personalized oncology innovation leadership.
The European cancer gene therapy market is segmented into Germany, France, the United Kingdom, Italy, and the Rest of Europe. The awareness of the advantages of cancer gene therapies is considerably high across the European region. Germany is expected to hold the largest share in the Europen cancer gene therapy market during the study period. General government spending in the European Union (EU) on health accounted for $1,273.68 (€1,073 billion) or 8.0 % of the gross domestic product (GDP) in the financial year 2020. In June 2022, Elicera Therapeutics AB, a clinical-stage cell and gene therapy company received funding of $2.67 million (€2.5 million) from the European Innovation Council (EIC) Accelerator Programme. Rising funding in the medical domain is anticipated to support the cancer gene therapy market remarkably.
Germany Market Trends
Germany is a fast-rising market, as the prevalence of cancer and the acceptance of the use of gene-based immunotherapies are increasing. Innovation is promoted by favorable EMA regulations, robust biotechnology investment, and industry-academia partnerships. Germany is at the forefront of clinical trials, which concentrate on CAR-T clinical trials, oncolytic viruses, and regenerative medicine breakthroughs.
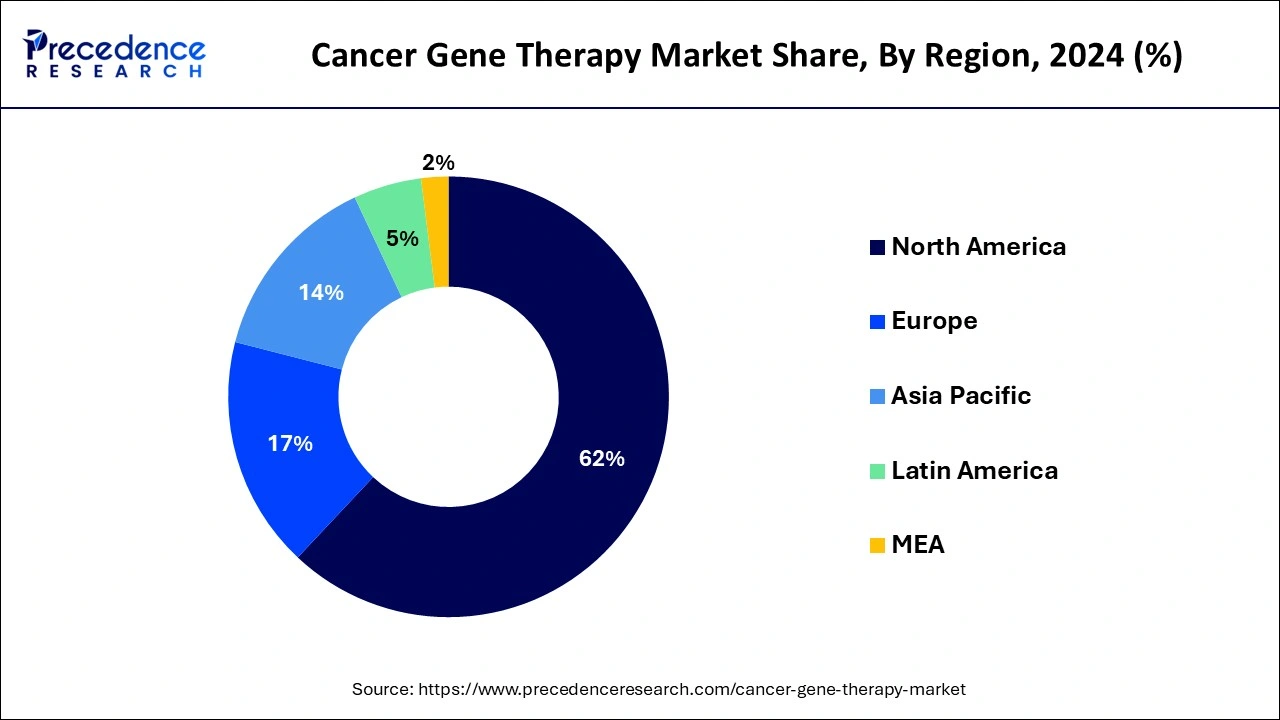
The cancer gene therapy market in the Asia Pacific (APAC) region is segmented into India, China, Japan, South Korea, and the rest of the Asia Pacific (APAC) region. China dominated the Asia Pacific region followed by Japan and India in 2023.
China Market Trends
The region is the fastest-growing because of the high number of patients, rising cancer cases, and the enhancement of healthcare facilities in China. Growth is supported by strong R&D investments, expedited regulatory routes, and local GMP production. The growing interest in CAR-T therapies and government-funded biotech programs further enhances the market growth.
The Latin America, Middle East, and African (LAMEA) cancer gene therapy market is segmented into North Africa, South Africa, Saudi Arabia, Brazil, Argentina, and the Rest of LAMEA. The Middle East and the Latin America region are anticipated to have notable growth in the cancer gene therapy market during the forecast period. Brazil held the largest share in the LAMEA region in 2023. Due to low literacy, uncertainty, and civil war in African countries, the cancer gene therapy market in Africa is expected to grow at a comparatively slow rate.
Brazil Market Trends
The Brazilian market is growing at a steady pace, as the level of cancer awareness and biotechnology infrastructure is improving. Brazil is at the forefront of regional growth in terms of research funding and the increased attention to new-fangled treatments. The increasing cancer burden is generating a need to find innovative gene therapies, which has been helped along by slow increases in the health care system and access to treatment.
UAE Market Trends
The UAE market is emerging on the basis of investment in healthcare infrastructure and biotechnology. GCC countries, in particular, the UAE, are facilitating gene therapy through public-private partnerships. Rising attention to innovative medical technologies, better access to healthcare, and the implementation of new treatment methods are stimulating the development of the market in the region.
Cancer Gene Therapy Market Companies
- OncoGenex Pharmaceuticals Inc.
- MerckKGaA
- Introgen TherapeuticsInc
- GSK plc.
- GenVec
- Genelux Corporation
- Elevate BioInc
- CelgeneInc
- Bluebird bio Inc.
- BioCancell Inc.
- Asklepios BioPharmaceutical Inc.
- Altor Bioscience Inc.
- Abeona Therapeutics Inc.
Recent Developments
- In February 2026, Dr. George Coukos aims to transform tumor biology research into personalized, cell-based therapies for difficult cancers through a new lab resulting from collaboration between Weill Cornell Medicine and the Ludwig Institute for Cancer Research. (Source: https://news.weill.cornell.edu )
- In January 2026, MedPark Hospital launched a Genomics Laboratory and genomic medicine services to enhance targeted cancer therapy. This initiative supports personalized treatment by utilizing patients' unique genetic profiles, moving beyond a one-size-fits-all approach, and aiming for precision medicine in cancer care.
(Source: https://www.medparkhospital.com )
Segments Covered in the Report
By Therapy
- Gene Induced Immunotherapy
- Oncolytic Virotherapy
- Gene Transfer
By End-User
- Hospitals
- Biopharmaceutical Companies
- Diagnostic centers
- Research Institutes
- Others
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
