What is the Catheter Stabilization Device Market Size in 2026?
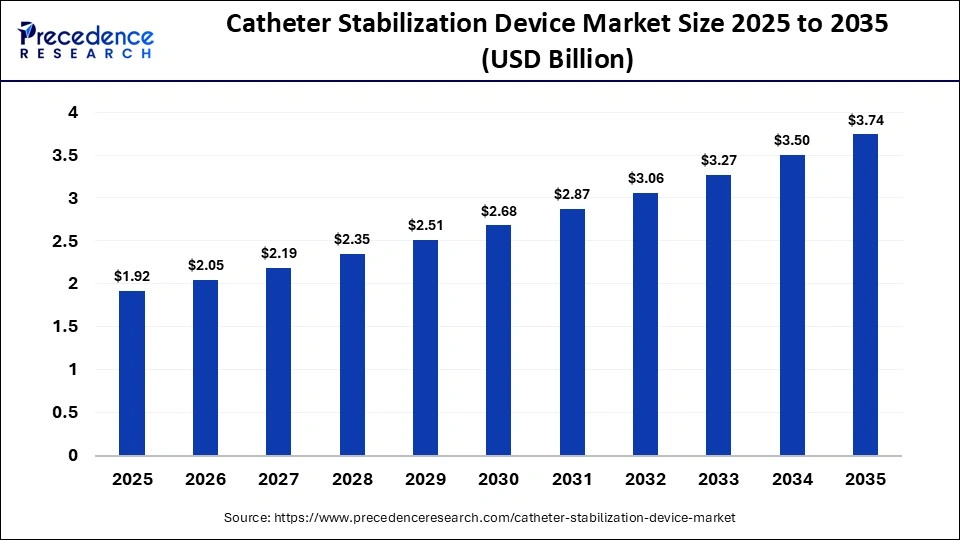
The global catheter stabilization device market size was calculated at USD 1.92 billion in 2025 and is predicted to increase from USD 2.05 billion in 2026 to approximately USD 3.74 billion by 2035, expanding at a CAGR of 6.90% from 2026 to 2035. The market is driven by the rising need to prevent catheter-related complications and infections, growing hospitalizations, and advancements in patient-centric securement technologies.
Key Takeaways
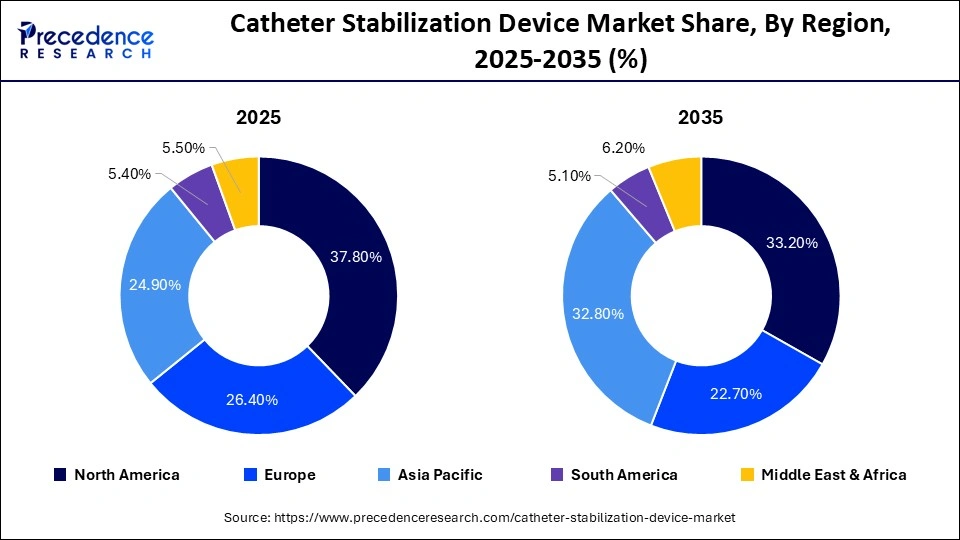
- North America dominated the global catheter stabilization device market with a share of 37.80% in 2025.
- The Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period.
- By product type, the peripheral intravenous catheter stabilization devices segment held a dominant position in the market with a share of 31.80% in 2025.
- By product type, the central venous catheter stabilization devices segment is expected to grow at the fastest CAGR of 8.0% between 2026 and 2035.
- By mechanism type, the adhesive securement devices segment held a 34.70% share of the market in 2025.
- By mechanism type, the locking securement devices segment is expected to grow with the highest CAGR of 9.20% during the studied period.
- By material, the polyurethane-based devices segment held a dominant position in the market, accounting for 37% share of the market in 2025.
- By material, the silicone-based devices segment is expected to grow at the fastest CAGR of 8.50% from 2026 to 2035.
- By end user, the hospitals segment led the global market with the largest share of 68.60% in 2025.
- By end user, the home care settings segment is expected to expand rapidly in the market with a CAGR of 9.50% in the coming years.
Market Overview
The catheter stabilization device market comprises medical securement products used to anchor catheters, tubes, and drainage lines to the skin to reduce migration, accidental dislodgement, catheter-related complications, and infection risk. It includes adhesive, sutureless, locking, strap-based, and anchor-pad systems designed for peripheral, central venous, arterial, urinary, epidural, and chest drainage catheters across hospitals, ambulatory settings, and home care. The escalating rates of chronic illnesses, including cardiovascular diseases, cancer, and kidney diseases, have led to the increased use of catheters. The growing focus on improving patient comfort and safety is also driving the market. Moreover, advances in skin-friendly adhesives, antimicrobial materials, and sutureless fixation devices, which minimize complications like catheter-related bloodstream infections, are contributing to market growth.
How is AI Influencing the Catheter Stabilization Device Market?
The emergence of artificial intelligence is significantly transforming the catheter stabilization device market by enhancing patient care, enabling smart monitoring, and improving device functionality. AI-powered systems can continuously monitor catheter positioning and detect early signs of dislodgement or potential complications, allowing healthcare providers to receive real-time alerts and take timely preventive action. In addition, AI is being leveraged in the development of advanced stabilization devices with improved adhesion, durability, and skin compatibility, ensuring greater patient comfort and reduced risk of device-related issues. As healthcare increasingly shifts toward connected and data-driven ecosystems, AI plays a crucial role in streamlining clinical workflows, reducing complications, and driving innovation in catheter stabilization technologies.
Catheter Stabilization Device Market Trends
- The growing adoption of sutureless catheter stabilization devices by healthcare professionals is driven by the need to reduce needlestick injuries, improve patient comfort, and minimize infection risks. These devices align with strict safety protocols and regulatory standards followed in hospitals and outpatient care settings worldwide.
- Manufacturers are increasingly developing advanced materials, such as antimicrobial coatings and hypoallergenic adhesives, to prevent infections and skin irritation. This is particularly important for patients requiring long-term catheterization, ensuring both safety and compliance with infection control guidelines.
- The rise in home healthcare and ambulatory care services is boosting demand for easy-to-use, reliable catheter stabilization solutions. These devices enable safe long-term catheter management outside hospital settings, helping reduce healthcare costs and hospital readmissions.
- As healthcare systems adopt stricter clinical practices, there is a growing emphasis on advanced catheter securement systems that minimize catheter movement and reduce the risk of catheter-associated complications and bloodstream infections (CABSI). This trend reflects the increasing focus on improving patient outcomes through enhanced medical device innovation.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.92 Billion |
| Market Size in 2026 | USD 2.05 Billion |
| Market Size by 2035 | USD 3.74 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 6.90% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product Type, Mechanism Type, Material, End User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Driver
Increasing Chronic Diseases and Catheterization Procedures
The increasing prevalence of chronic diseases, such as cardiovascular diseases, cancer, and renal diseases, is boosting the need for frequent and prolonged catheterization. The increasing number of surgical procedures and hospital admissions across the world is a major factor driving the market. Healthcare facilities are adopting stringent infection prevention measures and guidelines that minimize healthcare-associated infections, which also boosts the growth in the market. The patient comfort and safety are also enhanced with technological innovations, including sutureless devices and antimicrobial materials.
Restraint
High Costs, Regulatory Barriers, and Skin Sensitivity Issues
High cost associated with stabilization equipment, especially those that contain antimicrobial material or advanced designs, may create a barrier to adoption. Limited awareness and insufficient training among healthcare professionals in developing regions may further contribute to the continued use of traditional security techniques like sutures or tapes. Moreover, new devices are subject to stringent regulatory requirements and must undergo lengthy approval processes, which can ultimately delay product launches and slow down the introduction of innovative solutions into the market.
Opportunity
Expansion Opportunities in Home Care Settings and Integration of Smart Devices
The potential of the catheter stabilization device market is high with respect to innovation and evolving healthcare delivery models. The growing need for home care and remote patient monitoring is being addressed by user-friendly, long-lasting, and easily applicable stabilization devices. Material science, such as hypoallergenic and antimicrobial adhesives, provides an opportunity for better patient outcomes and wider implementation. Digital health technologies and smart monitoring systems with stabilization devices can be incorporated into patient safety processes and allow real-time tracking of catheters.
Segment Insights
Product Type Insights
Why Did the Peripheral Intravenous Catheter Stabilization Devices Segment Dominate the Market?
The peripheral intravenous catheter stabilization devices segment dominated the catheter stabilization device market, holding a major share of 31.80% in 2025. The dominance of the segment is driven by the widespread use of peripheral IV catheters in hospitals, emergency rooms, and outpatient centers that offer short-term intravenous care. There is extensive use of peripheral IV-based catheters in diverse groups of patients, as they are simple to place and versatile, and this leads to high demand for efficient stabilization gadgets. In addition, the growing knowledge of healthcare workers about the catheter-related bloodstream infection and hospital-acquired infection has facilitated the use of such equipment.
Catheter Stabilization Device Market Share, By Product Type, 2025-2035 (%)
| Product Type | 2025 | 2035 |
| Peripheral Intravenous Catheter Stabilization Devices | 31.80% | 27.40% |
| Central Venous Catheter Stabilization Devices | 24.60% | 27.90% |
| Arterial Catheter Stabilization Devices | 11.70% | 10.80% |
| Urinary Catheter Stabilization Devices | 17.80% | 18.30% |
| Chest Drainage Tube Stabilization Devices | 6.40% | 7.00% |
| Epidural Catheter Stabilization Devices | 3.90% | 3.70% |
| All-Site Catheter Stabilization Devices | 3.80% | 4.90% |
The central venous catheter stabilization devices segment is expected to grow at the fastest CAGR of 8.0% in the market during the forecast period due to the increased requirement of long-term vascular access in critical care, cancer, and dialysis patients. CVCs are required in chemotherapy, parenteral nutrition, and high-volume fluid therapy that needs constant stabilization to prevent the migration of the catheter, dislodging, and catheter-associated complications. The biocompatibility and comfort increase, and the clinical application is fostered by the use of materials with technological improvements, such as antimicrobial and skin-friendly adhesives. The increase in the number of intensive care units and the rise in the number of cases of chronic diseases that require long-term catheterization also support segmental growth.
Mechanism Type Insights
Catheter Stabilization Device Market Share, By Mechanism Type, 2025-2035 (%)
| Mechanism Type | 2025 | 2035 |
| Adhesive Securement Devices | 34.70% | 30.10% |
| Sutureless Securement Devices | 29.60% | 32.80% |
| Anchor Pad Securement Devices | 16.40% | 15.10% |
| Locking Securement Devices | 8.80% | 11.90% |
| Strap-Based Securement Devices | 10.50% | 10.10% |
Why Did the Adhesive Securement Devices Segment Lead the Catheter Stabilization Device Market?
The adhesive securement devices segment dominated the market with a share of 34.70% in 2025, driven by the increased popularity of adhesive-based devices as they are easy to use, comfortable to the patient, and useful in safe fixation. They minimize the movement of catheters, their unintended removal, and the development of catheter-associated infections. Moreover, adhesive securing devices can be used across various catheter materials and also have limited training requirements among the healthcare providers, which further enhances usability. The increased awareness of hospital-acquired infections and strict infection prevention measures has contributed to the increased adoption of adhesive stabilization solutions.
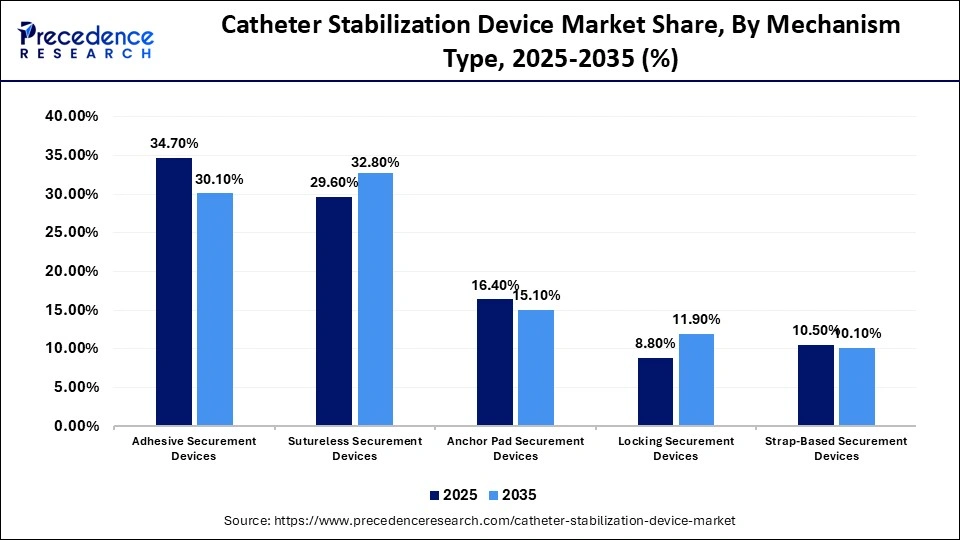
The locking securement devices segment is expected to grow at the highest CAGR of 9.20% between 2026 and 2035, driven by the growing need to stabilize patients with higher levels of safety and fewer catheter-related complications, especially in long-term vascular access of the critical care and oncology patients. Further improvements in clinical outcomes and comfort are enhanced by technological progress in locking mechanisms in combination with the use of antimicrobial and skin-friendly materials. The growth of the segment is also driven by growing awareness of infection prevention, an increase in ICU admissions, and the rising prevalence of chronic disease that necessitates extended catheterization.
Material Insights
Why Did the Polyurethane-Based Devices Segment Dominate the Catheter Stabilization Device Market?
The polyurethane-based devices segment held a dominant position in the market with a share of 37% in 2025. This is mainly due to their superior flexibility, durability, and biocompatibility, which ensure better patient comfort and reduced risk of skin irritation. These devices offer strong adhesion while maintaining the ability to conform to various body contours, making them suitable for long-term catheterization. Additionally, the availability of antimicrobial and hypoallergenic polyurethane options has further strengthened their adoption in both hospital and home-care settings.
Catheter Stabilization Device Market Share, By Material, 2025-2035 (%)
| Material | 2025 | 2035 |
| Silicone-based Devices | 18.70% | 22.40% |
| Foam-based Devices | 16.80% | 15.10% |
| Fabric-based Devices | 14.90% | 13.20% |
| Hydrocolloid-based Devices | 12.60% | 11.40% |
| Polyurethane-based Devices | 37.00% | 37.90% |
The silicone-based devices segment is expected to grow at the fastest CAGR of 8.50% in the market between 2026 and 2035, owing to their excellent biocompatibility and gentle adhesion, which minimize skin irritation and allergic reactions, making it ideal for sensitive patients. Silicone's flexibility and softness enhance patient comfort, particularly during long-term catheterization, supporting wider adoption in home care and hospital settings. Additionally, advancements in silicone formulations, including antimicrobial and durable variants, are driving increased preference for these devices among healthcare providers.
End User Insights
Catheter Stabilization Device Market Share, By End User, 2025-2035 (%)
| End User | 2025 | 2035 |
| Hospitals | 68.60% | 62.70% |
| Ambulatory surgical centers | 12.80% | 13.40% |
| Home care settings | 11.40% | 17.10% |
| Specialty clinics | 7.20% | 6.80% |
Why Did Hospitals Lead the Catheter Stabilization Device Market?
The hospitals segment registered its dominance in the global catheter stabilization device market with a share of 68.90% in 2025. This is because they handle a high volume of patients requiring short-term and long-term catheterization, creating consistent demand for reliable stabilization solutions. Hospitals prioritize patient safety and infection prevention, making advanced and antimicrobial stabilization devices essential in their care protocols. Additionally, hospitals have the resources and trained staff to implement and monitor these devices effectively, further driving their dominance in the market.
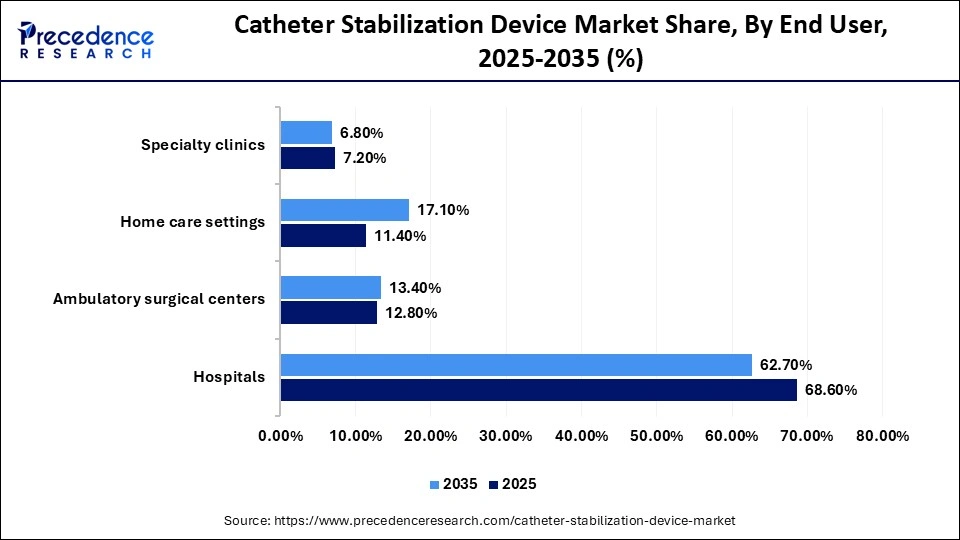
The home care settings segment is expected to grow at the fastest CAGR of 9.50% over the forecast period. The growth of the segment is driven by the rising demand for remote patient monitoring and at-home healthcare services. User-friendly, long-lasting, and easy-to-apply stabilization devices enable patients and caregivers to manage catheter care safely outside hospital settings. Additionally, the increasing focus on patient comfort and reducing hospital visits is driving the adoption of advanced devices in home care environments.
Regional Insights
North America Catheter Stabilization Device Market Size and Growth 2026 to 2035
The North America catheter stabilization device market size is estimated at USD 725.26 million in 2025 and is projected to reach approximately USD 1,432.42 million by 2035, with a 7.04% CAGR from 2026 to 2035.
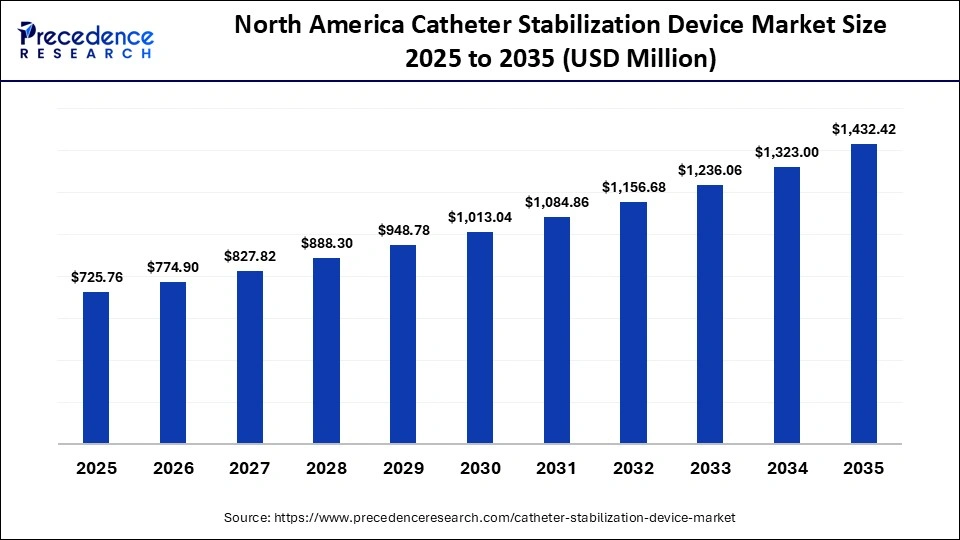
Why Did North America Lead the Global Catheter Stabilization Device Market?
North America led the catheter stabilization device market by holding a major share of 37.80% in 2025. The region's dominance is mainly driven by the availability of modern healthcare systems, a high level of implementation of innovative medical equipment, and stringent regulatory requirements that are focused on the safety of the patients. The adoption is aided by the rising chronic disease rates, including cardiovascular diseases, cancer, and kidney diseases, driving the need to be catheterized regularly and have a reliable method of stabilization. The regular updates in infection prevention measures and growing knowledge of catheter-related complications support the region's dominance in the market. The rising healthcare spending and availability of skilled healthcare workers make it possible to adopt new methods of catheter stabilization at a very early stage, which consolidates the hegemony of North America.
U.S. Catheter Stabilization Device Market Size and Growth 2026 to 2035
The U.S. catheter stabilization device market size is calculated at USD 544.32 million in 2025 and is expected to reach nearly USD 1,081.48 million in 2035, accelerating at a strong CAGR of 7.11% between 2026 and 2035.
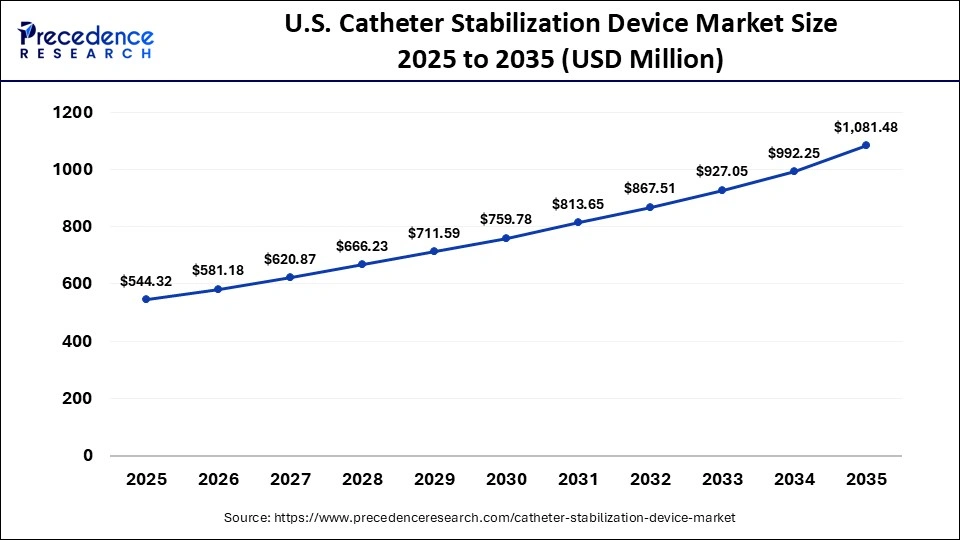
U.S. Catheter Stabilization Device Market Analysis
The U.S. is the major contributor to the Catheter Stabilization Device market within North America due to its substantial investment in AI research, presence of major tech giants, and ongoing advancements in AI models like LLMs, NMT, and others, which are the backbone of the translation services. A vibrant startup ecosystem with specialized translation tools is further supporting the country's position in the market.
Why is Asia Pacific Experiencing the Fastest Growth in the Catheter Stabilization Device Market?
Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period, driven by expanding healthcare infrastructure, increasing hospital admissions, and greater awareness of catheter-related complications and infection prevention guidelines. Rising rates of chronic diseases, increasing surgical procedures, and growing demand for home healthcare and outpatient services are further supporting market growth. Economic development, improved access to healthcare, and investment in advanced medical equipment are encouraging hospitals and clinics to adopt modern catheter stabilization systems, while government programs, medical reimbursement plans, and heightened patient awareness of safety and quality care are accelerating adoption in the region.
Catheter Stabilization Devices Market Value Chain Analysis
- Raw Material Sourcing
This stage involves sourcing essential raw materials such as medical-grade adhesives, polymers, silicone, and nonwoven fabrics, which are critical for ensuring patient safety, comfort, and durability.
Key Players:Dow Inc., DuPont, and 3M
- Component Manufacturing
At this stage, raw materials are processed into components such as adhesive backings, securement bases, locking mechanisms, and skin-friendly interfaces.
Key Players: Avery Dennison Corporation, Scapa Group plc, and Berry Global Inc.
- Clinical Trial and Regulatory Authorizations
Innovations are highly regulated under clinical trials to establish the safety and performance, along with the compatibility of the devices with various kinds of catheters. Regulatory approvals like the FDA and EMA given to the product before its market introduction are valid guarantees that the product conforms to the international standards of quality, safety, and efficacy.
Key Players: B. Braun Medical Inc., Smiths Medical, 3M Company
- Distribution to Hospitals and Pharmacies
Finished equipment is distributed via hospital procurement, medical distributors, and pharmacies. Efficient logistics will ensure timely delivery, maintenance of product integrity, and ease of delivery of the products in the inpatient, outpatient, and home health units.
Key Players: McKesson Corporation, Cardinal Health, Inc., Medline Industries, Inc.
Catheter Stabilization Device Market Companies
- Becton, Dickinson and Company
- 3M
- Baxter International
- B. Braun Melsungen AG
- Cardinal Health
- ConvaTec
- Merit Medical Systems
- Medtronic
- ICU Medical
- Dale Medical Products
- Centurion Medical Products
- TIDI Products
- Teleflex Incorporated
- Vygon
- Avanos Medical
Recent Developments
- In January 2025, the B. Braun medical company came up with a low-profile, soft catheter under the brand name of Clik-FIX Epidural/Peripheral Nerve Block Catheter Securement Device, which is designed to reduce catheter motion and dislocation in case of regional anesthesia. This innovation is expected to enhance patient safety and pain management during operations. (Source: https://www.bbraunusa.com )
- In February 2024, Cook Medical merged with Bedal International to purchase FlexGRIP catheter securement devices as an addition to its percutaneous drainage lines. The collaboration will promote patient care and mobility throughout various treatment procedures (Source: https://www.cookmedical.com )
Segments Covered in the Report
By Product Type
- Peripheral intravenous catheter stabilization devices
- Central venous catheter stabilization devices
- Arterial catheter stabilization devices
- Urinary catheter stabilization devices
- Chest drainage tube stabilization devices
- Epidural catheter stabilization devices
- All-site catheter stabilization devices
By Mechanism Type
- Adhesive securement devices
- Sutureless securement devices
- Anchor pad securement devices
- Locking securement devices
- Strap-based securement devices
By Material
- Silicone-based devices
- Foam-based devices
- Fabric-based devices
- Hydrocolloid-based devices
- Polyurethane-based devices
By End User
- Hospitals
- Ambulatory surgical centers
- Home care settings
- Specialty clinics
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
