What is the Dermatology CRO Market Size?
The global dermatology CRO market size is valued at USD 4.75 billion in 2025 and is predicted to increase from USD 5.04 billion in 2026 to approximately USD 8.55 billion by 2035, expanding at a CAGR of 6.05% from 2026 to 2035.
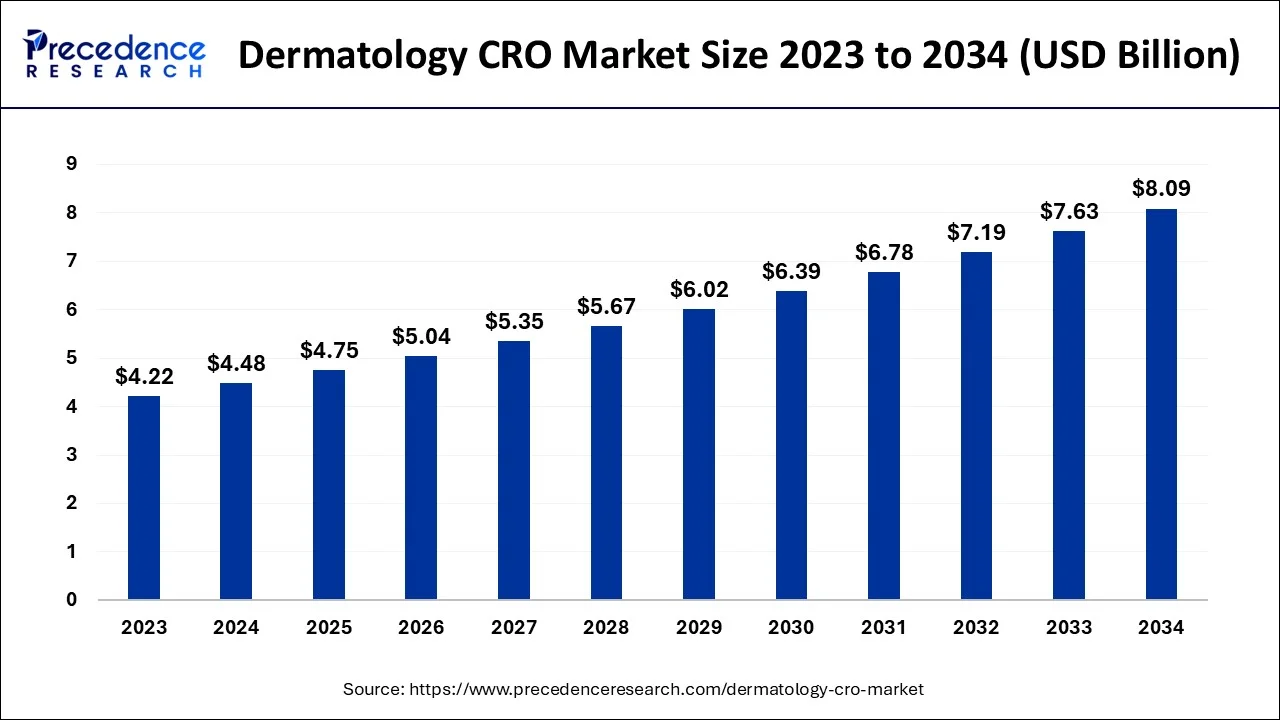
Dermatology CRO Market Key Takeaways
- In 2025, The Asia Pacific region led the market with more than 40.60% of the revenue share.
- Dermatology clinical research has been contracted out to CROs during the past ten years.
- In 2025, By type, the clinical segments captured more than 75.30% of the revenue share.
- By type, the preclinical segment is projected to grow at the highest CAGR of 8.5% from 2026 to 2035.
- In 2025, By service, the clinical monitoring segment had the greatest revenue share of more than 20.60%.
- In 2025, By phase type, the Phase III segments are the most expensive and include the most participants, therefore in 2025, they led the market with a revenue share of over more than 50.40%.
Market Overview
Institutions that help pharmaceutical or biotechnology corporations with their contracted research activities are known as dermatology contract research organizations. Both pharmaceuticals and medical equipment are produced by CROs. CROs provide all of the essential research for pharma and biotech businesses, including project planning and clinical trial administration
Dermatology CRO Market Growth Factors
One of the main factors propelling the market is the need for topical dermatological medications including anti-infective, anti-inflammatory, emollients, local anesthetics, and cleansers to treat acne. The dermatology contract research organization (CRO) industry is also being driven ahead by the increased public awareness of skin illnesses, the critical need for quick detection, and the rising incidence of skin cancer and other skin conditions.
The COVID-19 outbreak had a negative impact on the global economy in 2020 and is still having an impact now. The market for dermatological CRO was largely spared by the effects of the pandemic because of the development of online clinical trials and government initiatives to preserve faulty healthcare supply chains. Additionally, due to ongoing immunization campaigns and the repeal of the shelter-in-place rules, the backlog of clinical studies is decreasing. The market, therefore, seems to have a promising future.
Over the projection period, it is predicted that the prevalence of skin diseases would increase globally. Up to 50 million North Americans have acne each year, making it the most prevalent skin condition in the country, according to the American Academy of Dermatology Association. One in ten people will experience atopic dermatitis at some time in their lives. In the United States, 7.5 million individuals suffer from psoriasis. In the United States, rosacea is a common skin disorder that affects 16 million individuals. In the United States, more than 9,500 people receive a skin cancer diagnosis each day.
- A rise in the occurrence of skin cancer and other skin conditions as well as increased awareness of skin illnesses
- Expanding R&D expenditures, rising pharmaceutical industry outsourcing, and rising collaborative efforts will all contribute to the Contract Research Organization Services industry's expansion.
- Dermatological drugs are required due to the rising prevalence of skin cancer and other skin conditions. This will probably support market expansion.
- The elimination of adverse laws and the surge in vaccination demand are expected to accelerate market expansion.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 4.75 Billion |
| Market Size by 2035 | USD 8.55 Billion |
| Growth Rate from 2026 to 2035 | CAGR of 6.05% |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered |
|
| Regions Covered |
|
Market Dynamics
Key Market Drivers
An increasing number of clinical trials
- Worldwide, clinical trials are crucial to the process of developing skin medications. It aids in the development of innovative methods for detecting, diagnosing, and avoiding the onset of disease as well as a unique therapy for skin conditions. Clinical trials provide the scientific basis for diagnosing and treating patients as well as testing new drugs and medical devices. Even while researchers might not achieve the desired outcomes, clinical trial data assist point researchers on the proper path.
- Comparatively fewer clinical trials are undertaken in the United States than in other countries throughout the world. Since it is frequently simpler and less expensive to conduct clinical trials outside of the United States and the European Union. The success rates of dermatology clinical trials vary depending on the stage of the study and the therapies or products under development. Only 9% of drugs make it through phase 1 and are approved.
- The quantity of clinical studies that have been filed has dramatically increased recently. Dermatology clinical trials are becoming more complex, yet they are still important to research and create cutting-edge skin treatments and technology. The expansion of the dermatological CRO market is anticipated to be driven by this rise in clinical trials, which is anticipated to accelerate the creation of novel skin care medications.
Key Market Challenges
Several issues associated with dermatology clinical research
- CROs are still dealing with a number of problems, including commoditization, competitive pressure, and novel patient engagement models. Smaller CROs are under pressure from market segmentation as larger companies take a larger share of the market. In addition, there is a dearth of people with knowledge of and competence in the most recent regulatory requirements in many contract research businesses. There is a severe scarcity of clinical research associates and clinical trial associates, according to the Association of Clinical Research organizations. The dermatological CRO market's revenue growth is anticipated to be constrained by intense competition and a shortage of experienced specialists for clinical trials.
Key Market Opportunities
The growing investment by biopharmaceutical, pharmaceutical, and medical device companies
- The majority of biopharmaceutical, pharmaceutical, and medical device companies continue to invest a substantial amount of money in creating novel treatments and technology for illnesses connected to the skin. To get high-end, cutting-edge skin care products on the market, the pharmaceutical industry, in particular, is making significant investments in research and development. Sponsors typically contract with independent service providers to handle the development of medicinal and other products, which enables them to employ a more flexible pricing structure and do away with the need to retain redundant development capabilities around the globe.
- The expansion of the global market is being fueled by increasing expenditures made by many important firms in clinical and non-clinical research activities as well as outsourcing to various dermatological contract research organizations services that assist give cost-effective development solutions. The pattern indicates that the major pharmaceutical corporations are increasing the effectiveness of their research and development efforts and working together on new R & D.
- The pharmaceutical research and development industry is evolving as businesses strive to increase remote employee participation and put more emphasis on creating trial outcomes that are responsive to patient needs. In addition to significantly contributing to the market boom in the projected term, these new growth trends are anticipated to accelerate the development of new technologies and pharmaceuticals.
Dermatology CRO Market Segment Insights
[[segment_insights]]
Dermatology CRO Market Regional Insights
[[regional_insights]]
Top Companies in the Market & Their Offerings
- Bioskin: Offers specialized dermatology clinical trial services, including patient recruitment, skin assessments, protocol management, and data handling for drugs and devices targeting various skin conditions.
- TFS HealthScience: Provides end-to-end dermatology CRO services, including study design, patient enrollment, safety monitoring, and regulatory support for clinical trials across multiple skin disorders.
- Javara: Focuses on decentralized and site-based dermatology trials, offering patient recruitment, remote monitoring, and data management solutions for both rare and common skin conditions.
- ProInnovera: Delivers customized dermatology clinical research services, covering trial planning, endpoint assessments, safety oversight, and regulatory compliance for innovative skin therapies.
Dermatology CRO Market Companies
- IQVIA HOLDINGS INC.
- Covance Inc.
- Pharmaceutical Product Development, LLC (PPD)
- Parexel International Corporation
- Charles River Laboratories International, Inc.
- Icon, Plc
- Medidata Solutions, Inc.
- Syneos Health
- Pharmaron
- GVK Biosciences Private Limited
- Wuxi AppTec
- MEDPACE HOLDINGS, INC.
- PRA Health Sciences
- CTI Clinical Trial & Consulting
- Bioskin
- Proinnovera
- Biorasi
- Javara
- TFS
Recent Developments
- Icon plc and PRA Health Sciences signed a legally binding agreement in February 2021 to buy each other for $12 billion. Customers will benefit from the merged company's expanded geographic, functional, and therapeutic scope.
- A final deal was reached in 2021 for Thermo Fisher to pay USD 17.4 billion for PPD. Thermo Fisher will considerably increase its market share in the clinical research services sector as a result of this purchase, particularly in the early phase clinical trials sector.
Dermatology CRO Market Segment Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
