What is Endoscope Reprocessing Chemicals Market Size in 2026?
The global endoscope reprocessing chemicals market size was calculated at USD 1.09 billion in 2025 and is predicted to increase from USD 1.16 billion in 2026 to approximately USD 2.06 billion by 2035, expanding at a CAGR of 6.55% from 2026 to 2035. The market is driven by rising infection control requirements and increasing minimally invasive procedures.
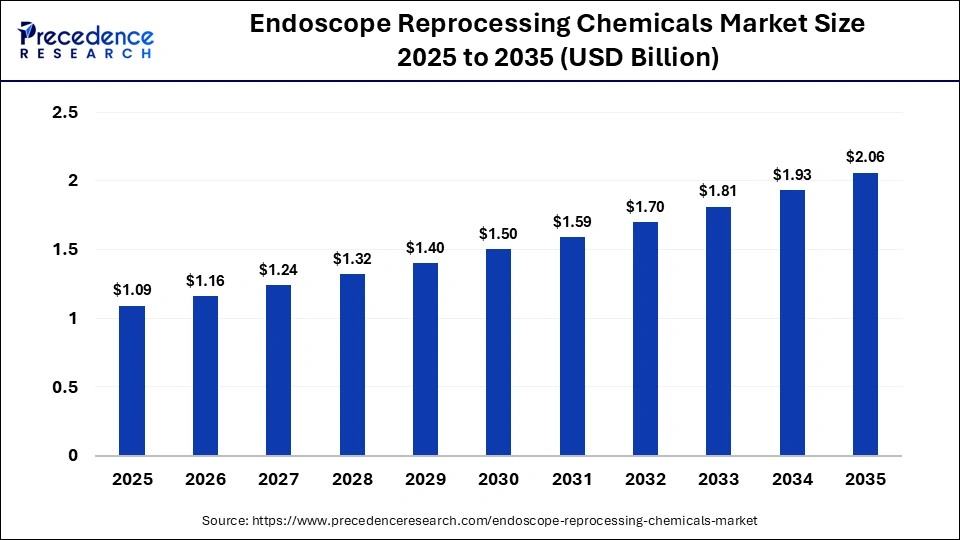
Key Takeaways
- By region, North America dominated the market in 2025.
- By region, Asia Pacific is expected to experience the fastest growth over the forecast period.
- By chemistry type, the high-level disinfectants segment held a significant share of the market in 2025.
- By chemistry type, the enzymatic cleaners segment is expected to grow at a significant rate in the market over the forecast period.
- By disinfectant type, the peracetic acid segment held a significant share of the market in 2025.
- By disinfectant type, the ortho-phthalaldehyde segment is expected to expand at a considerable rate in the market over the forecast period.
- By endoscope type, the GI endoscopes segment held a major market share in 2025.
- By endoscope type, the urology endoscopes segment is expected to grow at a significant CAGR in the market over the forecast period.
- By reprocessing platform, the automated endoscope reprocessors segment held a significant share in 2025.
- By reprocessing platform, the manual reprocessing segment is expected to grow at a considerable rate in the market over the forecast period.
Market Overview
The endoscope reprocessing chemicals market deals with developing and offering special cleansers, detergents, disinfectants, and high-level sterilization solutions to disinfect flexible and rigid endoscopes after medical practice. Endoscopes are widely used in gastrointestinal, pulmonary, urological, and minimal surgery, and to ensure patient safety and prevent cross-contamination, endoscopes require proper reprocessing. Their narrow lumens and complex channels make it impossible to clean the endoscopes because they are complicated in design. The industry is experiencing stable growth, as it has been observed that the number of minimally invasive procedures is increasing across the world, knowledge on hospital-acquired infections is increasing, and the regulatory policies that govern the practice of infection prevention are stringent.
How is AI Influencing the Endoscope Reprocessing Chemicals Market?
The endoscope reprocessing chemicals market is being revolutionized due to the Artificial Intelligence integration in the healthcare facility, driving changes in infection control, compliance checking, and efficiency of operations. Endoscope reprocessors are gradually being fitted with artificial intelligence to optimize the supply of chemicals, contact time, and accurate control of temperature and concentration levels in the process of high-level disinfection. The irregularities in the reprocessing parameters can be observed by AI-driven sensors and analytics platforms in real time, decreasing the need for humans as well as minimizing the chance of failure to sterilize the situation. Moreover, AI-based inventory control applications assist hospitals in predicting the use of chemicals, preventing shortages, and minimizing waste by forecasting demand models.
Endoscope Reprocessing Chemicals Market Trends
- The growing adoption of automated endoscope reprocessors is increasing demand for specialized, compatible chemical formulations that ensure high-level disinfection, reduce manual handling errors, maximize workflow efficiency, and meet global sterilization standards in hospitals.
- Heightened regulatory focus and evolving infection control standards are driving the development of novel, lower-toxicity, and environmentally friendly disinfectants that maintain efficacy against resilient pathogens while minimizing occupational exposure risks for healthcare workers.
- The increasing prevalence of hospital-acquired infections (HAIs) is accelerating the use of high-level disinfectants, such as peracetic acid and hydrogen peroxide-based formulations, which offer faster turnaround times and broader antimicrobial coverage.
- Expansion of outpatient surgical centers and ambulatory care facilities is boosting the need for affordable, easy-to-use reprocessing chemicals that provide consistent sterilization and high instrument turnover, even in smaller healthcare settings.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.09 Billion |
| Market Size in 2026 | USD 1.16 Billion |
| Market Size by 2035 | USD 2.06 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 6.55% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Chemistry Type, Disinfectant Type, Endoscope Type, Reprocessing Platform and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Chemistry Type Insights
Why Does the High-Level Disinfectants Segment Lead the Endoscope Reprocessing Chemicals Market?
The high-level disinfectants segment led the market with the largest revenue share in 2025. This is because of their proven effectiveness in providing all-around microbial removal, such as bacteria, viruses, fungi, and even some of the hard-to-kill pathogenic bacteria. Endoscopes belong to the group of semi-critical medical equipment, which contacts the mucous membranes, which is why high-level disinfection is obligatory according to the international infection control recommendations. The growing focus on healthcare-associated infections prevention and the strict regulatory requirements have also made validated HLD solutions even more acute.
The enzymatic cleaners segment is expected to grow at a significant CAGR over the forecast period, driven by the increasing awareness of the necessity of extensive pre-cleaning and high-level disinfection. Enzyme-based cleaners are important in the degradation of organic materials, e.g., blood, tissue, mucus, and biofilm, which accumulate in endoscope channels during the processes. Healthcare establishments are realizing that poor cleaning undermines the performance of disinfection, and as such, they are willing to invest more in high-quality enzymatic detergents. The demand for enzymatic cleaners is rising, as the standards of infection prevention tighten.
Disinfectant Type Insights
Why Did the Peracetic Acid Segment Dominate the Endoscope Reprocessing Chemicals Market?
The peracetic acid segment dominated the market while holding a major revenue share in 2025 and is expected to sustain its position throughout the projection period. This is mainly due to its broad-spectrum activity against bacteria, viruses, fungi, and spores, making it highly effective for high-level disinfection of flexible and rigid endoscopes. Peracetic acid's fast-acting nature and efficacy at low concentrations, even in the presence of organic matter, make it reliable in complex clinical environments. Adoption is further supported by regulatory encouragement for safer, effective sterilization solutions and growing concerns over hospital-acquired infections. Additionally, the global increase in minimally invasive surgeries has boosted endoscope usage, sustaining demand for high-performance disinfectants like peracetic acid.
The ortho-phthalaldehyde (OPA) segment is expected to grow at a significant CAGR in the coming years due to the fact that it has better material compatibility, reduced odor, and a better safety profile than the conventional disinfectant. OPA offers high-level disinfection that has a reduced exposure duration and does not involve any form of activation or complicated steps of handling, which healthcare workers find convenient. In addition, increasing regulatory attention to the matters of occupational safety and chemical exposure is motivating healthcare facilities to transition to safer options such as OPA. The use of reprocessing chemistry based on OPA is expected to increase significantly as the number of endoscopies in the world increases and healthcare institutions focus on employee safety and workflow optimization.
Endoscope Type Insights
What Made GI Endoscopes the Leading Segment in the Endoscope Reprocessing Chemicals Market?
The GI endoscopes segment led the market with the largest revenue share in 2025. This is mainly due to a significant rise in the number of GI endoscopic procedures, fueled by awareness campaigns emphasizing early disease detection and the routine use of colonoscopies. Reprocessing chemicals are in high demand for GI endoscopes because these procedures require high-level disinfection due to contamination risks. Technological advancements in flexible GI endoscopes have expanded their use in curative interventions, further increasing the frequency of reprocessing. As a result, GI endoscopes remain the largest consumer of disinfectant chemicals, reflecting the healthcare sector's continued focus on infection prevention and patient safety.
The urology segment is expected to grow at a significant CAGR over the forecast period due to the rising cases of urological ailments like kidney stones, bladder cancer, urinary tract infection, and benign prostatic hyperplasia. Demand for reprocessing chemicals is increasing due to the growing use of reusable urology endoscopes in minimally invasive procedures like cystoscopy and ureteroscopy. Segment growth is further supported by greater awareness of early diagnosis, improved healthcare access in developing economies, and advancements in flexible and digital urology scopes.
Reprocessing Platform Insights
Why Did the Automated Endoscope Reprocessors Segment Lead the Endoscope Reprocessing Chemicals Market?
The automated endoscope reprocessors segment led the market in 2025 and is expected to maintain its growth trajectory throughout the forecast period, driven by their capability to standardize and automatize cleaning and high-level disinfection process. Automated endoscope reprocessor (AER) systems significantly reduce human error, improving turnaround times and ensuring strict adherence to infection control protocols. These systems reduce the risk of cross-contamination and increase the efficiency of the entire hospital and ambulatory surgery center by automating the washing, disinfection, rinsing, and drying cycles. The increased attention to patient safety, adherence to the regulations, and the optimization of the workflow have stimulated the implementation of AER systems worldwide.
The manual reprocessing segment is expected to grow at a significant rate in the coming years, driven by strong demand in resource-limited and cost-conscious healthcare facilities. With lower start-up costs compared to automated systems, smaller hospitals, clinics, and facilities in emerging economies often rely on manual reprocessing. While labor-intensive, its safety and effectiveness are increasingly enhanced through training programs and improved standard operating procedures, supporting continued adoption in budget-constrained settings.
Regional Analysis
Why Did North America Lead the Global Endoscope Reprocessing Chemicals Market?
North America held the dominant share of the endoscope reprocessing chemicals market in 2025, driven by its well-established healthcare infrastructure, high procedural volumes, and strict infection prevention regulations. The region's advanced network of hospitals, ambulatory surgical centers, and specialty clinics performs numerous minimally invasive surgeries, creating strong demand for high-level disinfectants and sterilization chemicals. Stringent regulatory requirements from authorities like the U.S. FDA and CDC, along with increased awareness of hospital-acquired infections and rising medico-legal responsibilities, have further spurred investments in automated reprocessing systems and high-quality chemical formulations.
U.S. Market Analysis
The endoscope reprocessing chemicals market in the U.S. is growing due to high healthcare standards, substantial public and private investment, and strong clinical awareness of patient safety. The rising prevalence of chronic conditions, aging population, and increased use of medical devices is driving demand for effective reprocessing and sterilization solutions. Additionally, the presence of leading wound care and disinfectant product manufacturers and ongoing R&D initiatives are accelerating market expansion.
Why is Asia Pacific Experiencing the Fastest Growth in the Endoscope Reprocessing Chemicals Market?
Asia Pacific is expected to grow at the fastest CAGR during the forecast period, driven by the expansion of healthcare infrastructure and rising procedure volumes. Rapid urbanization, increased healthcare investments, and greater access to diagnostic and surgical services are boosting the adoption of minimally invasive procedures, which require frequent endoscope use. Additionally, government initiatives to strengthen infection control policies and hospital accreditation standards, coupled with the rising prevalence of gastrointestinal, respiratory, and urological diseases, are fueling demand for effective endoscope reprocessing chemicals.
Why is the European Endoscope Reprocessing Chemicals Market Growing?
The market in Europe is expected to grow at a notable rate in the upcoming period, driven by stringent regulatory standards and a strong emphasis on patient safety. Healthcare providers must comply with European regulations and health organization guidelines that mandate the use of high-quality reprocessing chemicals for medical device sterilization. Additionally, the rising prevalence of chronic diseases requiring endoscopic procedures, an aging population, and growing concerns over cross-contamination and product recalls are boosting demand for validated, high-performance disinfectants, supporting sustained market growth in the region.
Endoscope Reprocessing Chemicals Marke Value Chain Analysis
- Raw Material Suppliers:
Companies supply active ingredients like peracetic acid, hydrogen peroxide, and quaternary ammonium compounds, forming the backbone of disinfectants.
Key players: BASF, Dow Chemicals, and Solvay. - Chemical Formulation & Manufacturing:
This stage involves converting raw materials into ready-to-use high-level disinfectants with regulatory compliance for medical applications.
Key players: Johnson & Johnson (Ethicon), Cantel Medical (STERIS), and Advanced Sterilization Products (ASP). - Distribution & Logistics:
Efficient supply chain and cold-chain logistics ensure timely delivery to hospitals and clinics.
Key players: McKesson Corporation, Medline Industries, and Cardinal Health.
Endoscope Reprocessing Chemicals Market Companies
- Advanced Sterilization Products (ASP)
- STERIS
- Getinge
- Ecolab
- Olympus (chemical partners)
- Cantel (STERIS)
- Metrex
- Schülke & Mayr
- Paul Hartmann
- Johnson & Johnson (infection prevention)
Recent Developments
- In May 2025, Olympus Corporation introduced the ScopeLocker Air endoscope drying cabinet to improve the workflow concerning drying efficiency and make it easier to store such equipment in the healthcare facility. The system facilitates infection prevention activities and complies with the changing reprocessing policies to enhance the level of patient safety and compliance. (Source: https://www.olympusamerica.com)
- In March 2025, Nanosonics received FDA De Novo clearance for its CORIS system, which is a milestone in the innovation of endoscope reprocessing. The CORIS platform has been identified as the first automated cleaning device that was designed with the special purpose of cleaning the inner tube of the flexible endoscopes and enhancing the validation of cleaning and the minimization of chances of contamination. (Source: https://trial.medpath.com)
Segments Covered in the Report
By Chemistry Type
- High-Level Disinfectants (HLD)
- Enzymatic Cleaners
- Detergents for AER Systems
- Other Chemicals
By Disinfectant Type
- Peracetic Acid
- Ortho-Phthalaldehyde (OPA)
- Hydrogen Peroxide Systems
- Other Actives9
By Endoscope Type
- GI Endoscopes
- Bronchoscopes
- Urology Endoscopes
- Other Scopes
By Reprocessing Platform
- Manual Reprocessing
- AER (Automated Endoscope Reprocessors)
- Other Platforms
By Region
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
