Electronic trial master file (eTMF) systems are transforming clinical research by replacing paper-based documentation with secure, digital solutions. They improve compliance, streamline workflows, and enhance collaboration across global trial teams.
In the rapid-paced world of clinical trials, documentation is not just administrative. It is the heart of the regulatory approval process. If a trial is not accurately documented, the authorized regulatory agencies, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), assume it did not happen. The traditional paper trial master file (TMF) is increasingly becoming obsolete and rapidly replaced by the electronic trial master file (eTMF), a digital, secure, and collaborative solution essential for advanced and efficient research. Now, it is time to harness the power and unlock a new era of efficiency and excellence in trial management.
The Digital Evolution of Clinical Trial Management
During the last decade, the landscape of clinical research has experienced a seismic shift, driven by accelerating technological advancement toward high efficiency and compliance. At the heart of such change is the emergence of eTMF systems, which would revolutionize the way a research team organizes and manages its trial data.
The electronic trial master file (eTMF) is a digital version of the traditional trial master file (TMF) widely used in clinical research. Nowadays, the eTMF systems are the crucial tools that improve data security and enable conducting clinical trials efficiently to align with the regulatory requirements.
However, with the surge in complex trials and the inadequacies of paper-based TMFs and outdated file-sharing methods. eTMF systems centralize trial documentation, making it easier to manage and access the processes involved.
What is an Electronic Trial Master File (eTMF)?
An eTMF refers to the modern software system that acts as a digital repository to capture, manage, share, and store essential documents generated during a clinical trial. It is the electronic counterpart that is replacing paper-based TMF. The eTMF is specifically designed to house all essential documentation that assists sponsors and regulators in evaluating the conduct of a clinical trial and the quality of the data produced. It is the crucial set of documents proving that a given clinical trial was conducted under GCP and other regulations applied. As per the Good Clinical Practice (GCP) guidelines, these documents are required to be accurate, legible, contemporary, original, and attributable (ALCOA principles).
What is the Electronic Trial Master File System Market Size in 2026?
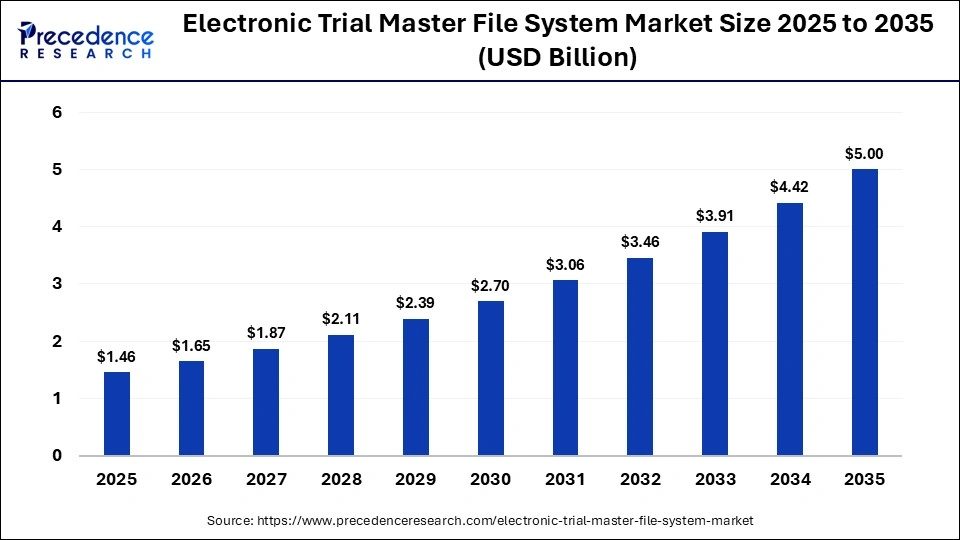
The global electronic trial master file system market was valued at USD 1.46 billion in 2025 and is projected to grow from USD 1.65 billion in 2026 to approximately USD 5.00 billion by 2035, registering a CAGR of 13.10% during the forecast period from 2026 to 2035. The market is witnessing significant growth, driven by the increasing need for secure and centralized data management solutions, a rise in R&D activities, and growing integration with AI and automation.
Major Benefits of Adopting eTMF
- Improved Inspection Readiness: It enables quick retrieval of sensitive documents, which assists in inspection and prevents findings.
- Reduced Costs: The shift away from paper-based, physical file management significantly lowers storage and logistics costs.
- Enhanced Oversight: The real-time visibility empowers CROs and sponsors to monitor TMF quality and site performance.
How are eTMF Systems Streamlining Modern Clinical Trials?
Clinical trials are generally complex. Without a robust system, it often becomes difficult to organize the documents required. This is where the use of eTMF systems arises. Streamline clinical trial documentation with an eTMF solution that ensures compliance, enhances collaboration, and accelerates study timelines, all in one secure digital platform.
- Centralized Document Management: Those days of searching for clinical trial documents in filing cabinets and sifting through multiple email threads are gone. An Electronic Trial Master File centralizes all crucial files in one place and provides real-time access. Sponsors and researchers can collaborate conveniently from anywhere in the world, ensuring that every person has timely access to up-to-date information.
- Automated Workflows: The modern eTMF has the ability to automate the tedious and repetitive work. The use of automated alerts assists team members with a reminder about pending document approvals or near-expire documents. Automated workflows ensure smooth passage through defined, successive stages, reduce delays, and increase compliance.
- Real-Time Reporting: Monitoring the development of a trial requires detailed information on the quality of documentation, distinguishing between documents that are complete and those requiring urgent focus. A modern eTMF system provides dashboards and real-time reporting that give a comprehensive view of the trial's status.
From Paper to Progress: How eTMF Systems Accelerate Trial Timelines?
The shift toward eTMF is driven by the rising need for high efficiency, speed, and compliance. There are various major benefits, including:
- Real-time Inspection Readiness: Paper files often take weeks to organize; the eTMF systems allow for live review of documents, which makes the system perpetually ready for audits.
- Reduced the Time for Audit Preparation: Several studies suggest that the adoption of an eTMF system can significantly reduce the time spent preparing for audits and site visits.
- Enhanced Collaboration: Contract Research Organizations (CROs), sites, and sponsors can efficiently access the same documents simultaneously from anywhere, which facilitates remote monitoring.
- Improved Speed and Efficiency: The digital workflows can automate the routing of documents for review and approval, which accelerates the overall trial lifecycle.
- Superior Security and Backup: eTMF helps in ensuring that documents are backed up, preventing loss, and are typically more secure than physical files.
Core Features of an Effective eTMF System
A robust eTMF system should include the following features:
- Document Management and Indexing: Automatic routing of documents based on a standardized structure, often aligned with the DIA TMF Reference Model. The attachment of unique identifiers (study, site, country, document type) allows for 2D searching and rapid retrieval.
- Quality Control (QC) Workflows: Support for electronic signatures compliant. The ability to shift documents automatically through the review and approval process.
- Reporting and Dashboards: The real-time reporting on document completeness, expected document tracking, and site-specific progress.
- Security and Compliance: The Electronic Trial Master File (eTMF) system is designed to maintain inspection readiness, data integrity, and regulatory compliance. Restricting access to sensitive documents based on user roles such as site monitor, sponsor, and investigator, which prevents unauthorized viewing or editing, and protects blinded information.
Best Practices for eTMF Management
To maximize the core value of an eTMF system, organizations are following these best practices:
- Adopt a Standardized Reference Model: Utilize the Drug Information Association (DIA) TMF Reference Model to ensure a consistent and recognizable structure for all trials.
- Implement Risk-Based Oversight: The increasing focus on quality reviews of high-risk, critical documents to ensure trial integrity.
- Ensure Continuous QC: The regular and ongoing QC ensures that missing or inaccurate documents are detected quickly.
- Train Team Members: Ensure all staff, including Contract Research Organizations (CROs) partners, are well-trained on the specific eTMF system being used.
- Secure Access: Regularly audit user permissions to ensure that only authorized personnel have access to highly sensitive documents.
The Future of eTMF: AI and Automation
The eTMF is rapidly evolving beyond a simple document repository, moving toward intelligent automation. AI-driven document processing now helps automatically categorize documents and identify missing items, improving efficiency and compliance. Additionally, tighter integration with clinical trial management systems (CTMS) and electronic data capture (EDC) systems ensures seamless data flow, enhancing overall trial management and accelerating decision-making.
Simplifying Compliance with eTMF Systems
Compliance is a critical aspect of clinical research, as any deviation from regulatory requirements can compromise the validity of a trial. eTMF systems are specifically designed to streamline compliance, offering integrated tools to help researchers adhere to ICH, GCP, and other stringent regulations. Managing multiple document versions can be challenging, but an electronic trial master file ensures users always reference the most current version while archiving older versions for reference. All clinical trial documents must be accurate, complete, and, when required, properly signed. Automated completeness checks in eTMF systems detect missing information, reducing the risk of non-compliance. During audits, files must be well-organized and readily accessible; eTMF systems enable trial teams to retrieve documents quickly and provide audit trails that demonstrate compliance efficiently.
Driving Trial Success through Centralized Document Management
Electronic trial master file (eTMF) systems have become an indispensable digital tool in modern clinical research. By replacing traditional paper-based methods, eTMF systems provide a secure, centralized, and real-time environment that ensures regulatory compliance, enhances global collaboration, and accelerates drug development timelines. The rising adoption of eTMF systems is not only a technological upgrade; it is a strategic data-driven decision that allows clinical research teams to work more smartly and securely.
The implementation of these systems streamlines operations, enhances data security, and simplifies compliance, ultimately paving the way for more efficient and successful clinical trials. Whether a CRO, sponsor, or investigator, embracing the power of the Electronic Trial Master File is no longer an option; it is a necessity in today’s fast-paced clinical research environment. The outstanding ability of eTMF systems to streamline, secure, and simplify trials makes them truly the backbone of modern clinical research. Their impact is undeniable, and their potential is limitless.
Expert Opinion
According to our industry experts, eTMF systems have become indispensable in the clinical research industry, offering both operational efficiency and regulatory assurance. As clinical trials grow in complexity and scale, managing vast amounts of documentation manually can be time-consuming, error-prone, and costly. eTMF systems streamline this process by centralizing trial documents, automating version control, and enabling real-time access for global teams, significantly reducing administrative overhead.
Integration with clinical trial management systems (CTMS) and electronic data capture (EDC) platforms enhances workflow efficiency, allowing seamless data flow across different trial functions. Advanced features such as AI-driven document classification, completeness checks, and audit trail generation not only improve compliance with ICH, GCP, and other stringent regulations but also mitigate the risk of regulatory delays or fines.
From a business standpoint, these efficiencies translate into faster trial timelines, reduced operational costs, and improved data integrity. Companies that invest in robust eTMF systems gain a competitive advantage by accelerating drug development cycles, enhancing collaboration across decentralized teams, and ensuring audit readiness, ultimately driving higher returns on clinical research investments.
About the Authors

Aditi Shivarkar
Aditi, Vice President at Precedence Research, brings over 15 years of expertise at the intersection of technology, innovation, and strategic market intelligence. A visionary leader, she excels in transforming complex data into actionable insights that empower businesses to thrive in dynamic markets. Her leadership combines analytical precision with forward-thinking strategy, driving measurable growth, competitive advantage, and lasting impact across industries.

Aman Singh
Aman Singh with over 13 years of progressive expertise at the intersection of technology, innovation, and strategic market intelligence, Aman Singh stands as a leading authority in global research and consulting. Renowned for his ability to decode complex technological transformations, he provides forward-looking insights that drive strategic decision-making. At Precedence Research, Aman leads a global team of analysts, fostering a culture of research excellence, analytical precision, and visionary thinking.

Piyush Pawar
Piyush Pawar brings over a decade of experience as Senior Manager, Sales & Business Growth, acting as the essential liaison between clients and our research authors. He translates sophisticated insights into practical strategies, ensuring client objectives are met with precision. Piyush’s expertise in market dynamics, relationship management, and strategic execution enables organizations to leverage intelligence effectively, achieving operational excellence, innovation, and sustained growth.





 Request Consultation
Request Consultation

