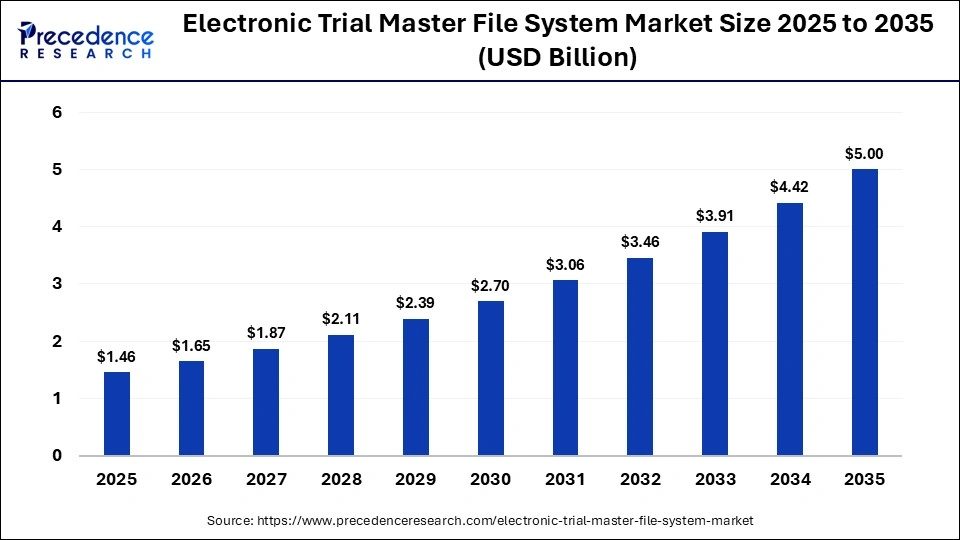
What is the Electronic Trial Master File System Market Size in 2026?
The global electronic trial master file system market size was calculated at USD 1.46 billion in 2025 and is predicted to increase from USD 1.65 billion in 2026 to approximately USD 5.00 billion by 2035, expanding at a CAGR of 13.10% from 2026 to 2035. The market is experiencing unprecedented growth, driven by the growing need for secure and centralized data management solutions, a surge in the number of R&D activities, and rising integration with AI and automation.
Key Takeaways
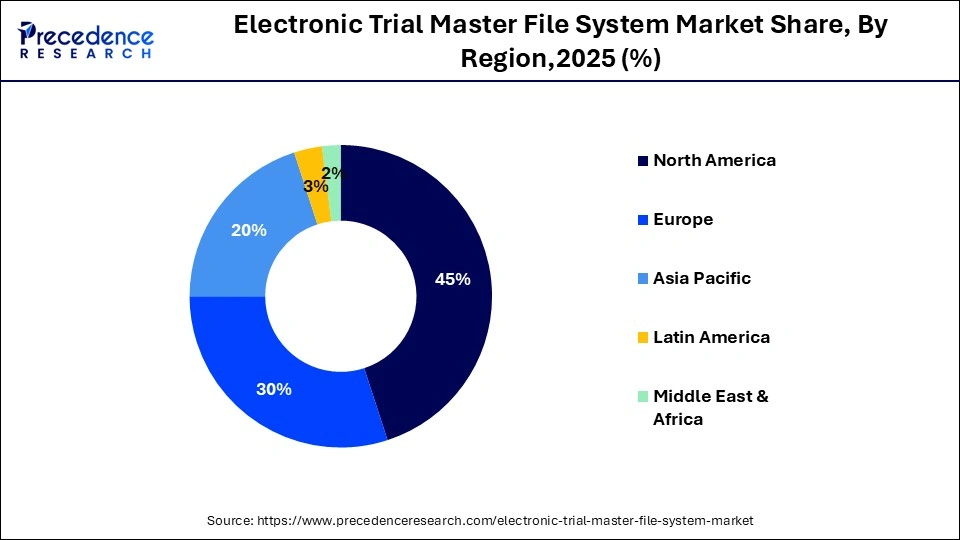
- North America held the largest market share of 45% in 2025.
- Asia Pacific is expected to expand at the fastest CAGR of 13.5% between 2026 and 2035.
- By component, the software segment held the largest market share of 70% in 2025.
- By component, the services segment is expected to grow at the fastest CAGR of 11.2% between 2026 and 2035.
- By deployment mode, the cloud-based segment contributed the biggest revenue share of 65% in the market in 2025.
- By deployment mode, the on-premises segment is expected to expand at the fastest CAGR of 11.4% between 2026 and 2035.
- By application, the clinical trial management segment held a dominant share of 50% in 2025.
- By application, the collaboration & workflow management segment is expected to grow at the highest CAGR of 12.1% from 2026 to 2035.
- By end user, the pharmaceutical & biotechnology companies segment held a dominant share of 55% in 2025.
- By end user, the contract research organizations (CROs) segment is expected to grow at the highest CAGR of 11.7% from 2026 to 2035.
Market Overview
The electronic trial master file system market refers to the global industry focused on digital platforms designed to collect, manage, store, and track clinical trial documents in a compliant and secure manner. eTMF systems improve efficiency, reduce regulatory risks, and enhance collaboration among clinical research teams by replacing paper-based trial master files. They are widely used by pharmaceutical companies , biotechnology firms, contract research organizations (CROs), and academic research institutions to streamline clinical trial documentation and ensure adherence to regulatory standards like the FDA, EMA, and ICH-GCP.
How is AI Impacting the Electronic Trial Master File System Market?
In the rapidly evolving technological landscape, Artificial Intelligence (AI) integration emerges as a game-changer in accelerating the growth of the electronic trial master file system market by transforming traditional repositories into intelligent and digital platforms, significantly reducing document processing time. The integration of AI enhances efficiency through automated indexing, quality control, and risk-based management, which reduces the risk of human error and ensures regulatory compliance. Leveraging the power of artificial intelligence (AI) and machine learning (ML) can automate the classification, metadata extraction, and quality control of clinical trial documents in real-time.
AI algorithms, which are highly trained on massive datasets, can accurately analyze document content to automatically store files in the correct folders and populate metadata, reducing the need for manual entry. AI facilitates real-time auditing and anomaly detection to identify missing and incorrect records to enhance regulatory compliance. The eTMF system uses AI to prioritize high-risk documents critical to inspections, allowing study teams to focus resources on critical and sensitive trial data.
What are the Emerging Trends in the Electronic Trial Master File System Market?
- The rising chronic disease prevalence, along with the rising volume of clinical trials by biotech and pharmaceutical companies, is expected to accelerate the growth of the market during the forecast period. The increase in global clinical trials necessitates standardized and efficient document management systems.
- The rapid shift from paper-based and physical file management to digital platforms is expected to contribute to the overall growth of the market. Shifting from manual paper-based TMFs significantly reduces turnaround times, operating costs, and risk of human error or data loss.
- The stringent regulatory guidelines are anticipated to boost the market's expansion in the coming years. The regulations implemented by the regulatory bodies, like the EMA and FDA, necessitate better data integrity and transparency. The adoption of eTMF systems assists in improving data security and enables conducting clinical trials efficiently to align with strict regulatory requirements.
- The rising shift toward cloud-based solutions is anticipated to promote the market's growth during the forecast period. The rapid adoption of cloud computing enables real-time access and collaboration between Contract Research Organizations (CROs) and sponsors across different locations.
- The rising outsourcing of clinical trials to Contract Research Organizations (CROs) is expected to drive the growth of the market during the forecast period. As biotech and pharmaceutical companies externalize trials, the prominent eTMF systems provide centralized, digital security , real-time collaboration, and regulatory compliance across global sites.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.46 Billion |
| Market Size in 2026 | USD 1.65 Billion |
| Market Size by 2035 | USD 5.00 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 13.10% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Component, Deployment Mode, End User, Application, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Driver
Adoption of Digital Solutions
The increasing adoption of digital solutions to streamline clinical trial documentation and compliance is driving the market. eTMF systems enable faster, more accurate management of trial records, reducing the risk of regulatory non-compliance and audit findings. Additionally, the shift toward decentralized and remote clinical trials has further accelerated demand for digital platforms that allow secure, real-time access to trial documents for sponsors, investigators, and regulatory authorities.
Restraint
High Cost
One significant restraint of the electronic trial master file system market is the high implementation and maintenance costs associated with these digital platforms. Many small and mid-sized clinical research organizations face challenges in allocating sufficient budget and technical resources for system deployment, training, and ongoing support. Additionally, integrating eTMF systems with existing IT infrastructure and ensuring data security and regulatory compliance can be complex, which may slow adoption in cost-sensitive organizations.
Opportunity
Trend of Decentralized and Remote Clinical Trials
One key opportunity for the market lies in the growing trend of decentralized and remote clinical trials. As clinical research increasingly moves toward virtual and hybrid models, there is a rising need for secure, cloud-based platforms that enable real-time access to trial documents from multiple locations. This shift allows sponsors and investigators to improve collaboration, accelerate trial timelines, and enhance regulatory compliance, creating significant growth potential for eTMF solutions.
Segment Insights
Component Insights
Electronic Trial Master File System Market Share, By Component, 2025 (%)
| Component | 2025 |
| Software | 70% |
| Services | 30% |
What Made Software the Dominant Segment in the Market?
The software segment dominated the electronic trial master file system market with the largest share of 70% in 2025, owing to the rapid digital transformation, widespread AI integration, and the increasing shift toward the cloud-based Software-as-a-Service (SaaS) models. Software has become the heart of organizational strategy, rapidly transitioning towards becoming an autonomous and AI-powered system. Modern software integrates AI to cut down on manual labor and speed up document retrieval times. In addition, the growing need to improve data security and the surge in decentralized clinical trials (DCTs) are anticipated to drive the segment's growth during the forecast period.
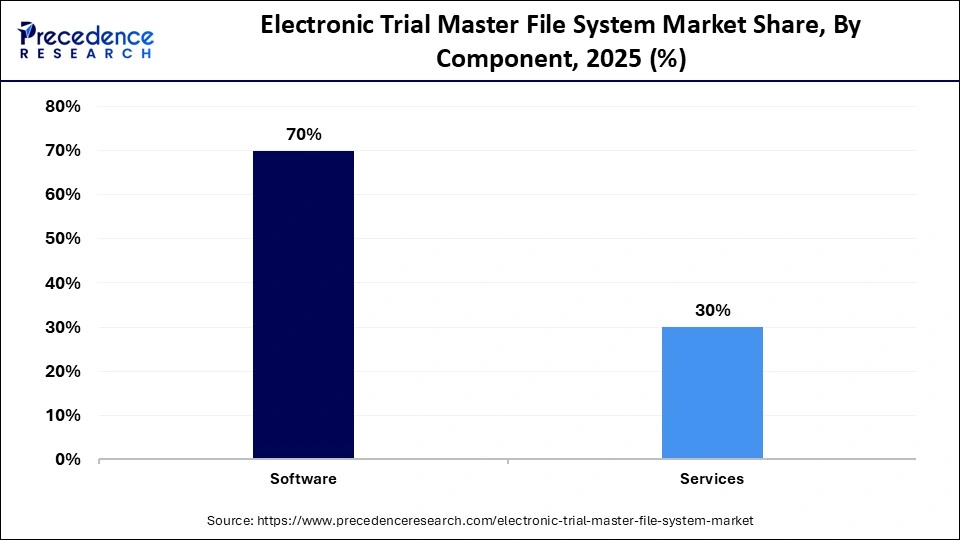
The services segment is expected to grow at a remarkable CAGR of 11.2% between 2026 and 2035. The segment's fastest growth is driven by the growing need for expert consulting, implementation, training, and regulatory validation services. With the rise in clinical trial complexity, organizations are increasingly relying on these services to ensure regulatory compliance, efficiently manage data migration, and optimize system usage for audit readiness. Companies require specialized support to configure and customize the eTMF system to fit the specific and required clinical trial needs.
Deployment Mode Insights
Electronic Trial Master File System Market Share, By Deployment Mode, 2025 (%)
| Deployment Mode | 2025 |
| Cloud-Based | 65% |
| On-Premises | 35% |
Why Did the Cloud-Based Segment Dominate the Market?
The cloud-based segment dominated the electronic trial master file system market with a 65% share in 2025. This is because cloud-based solutions offer numerous benefits such as cost-effectiveness, scalability, easier access, and eliminate the need for high upfront investment in IT infrastructure, allowing organizations of all sizes to adopt tools based on subscription. Cloud-based eTMF systems provide secure and real-time remote access to trial documents, essential for modern decentralized trials. Cloud-based systems empower decentralized teams, CROs, and sponsors to access documents in real-time from any location, which in turn improves efficiency. In addition, the rising shift toward digital work environments and the enhanced collaboration between stakeholders are driving the adoption of cloud-based tools.
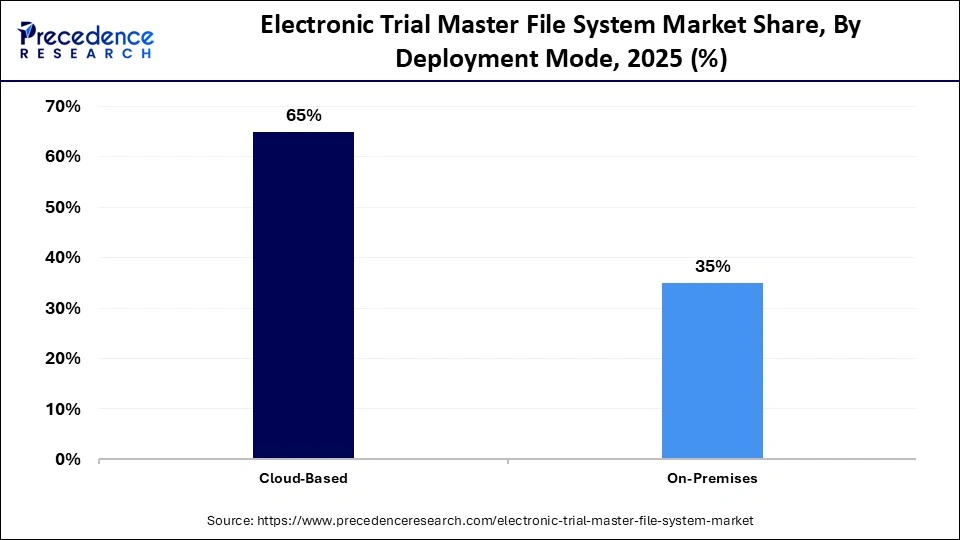
The on-premises segment is expected to expand at the fastest CAGR of 11.4% during the forecast period, owing to the rising need for direct control, increasing requirement for deep customization by large pharmaceutical and biotechnology companies, the rising need to eliminate unauthorized access or third-party interference, and stringent data privacy laws. Organizations with stringent data policies often use on-premise solutions to prevent breaches, maintaining physical control over their servers. The ability to customize systems to meet stringent regulatory requirements is driving the segment's growth during the forecast period.
Application Insights
Electronic Trial Master File System Market Share, By Application, 2025 (%)
| Application | 2025 |
| Clinical Trial Management | 50% |
| Regulatory Compliance | 20% |
| Document Management | 15% |
What Made Clinical Trial Management the Dominant Segment in the Market?
The clinical trial management segment held the largest share of 50% in the electronic trial master file system market in 2025, owing to the rising need to manage complex trials, speed up document submission, and ensure compliance with strict regulatory norms. Clinical trial management manages complex trial operations, such as subject tracking, protocol planning, and financial management, crucial for managing trial workflows. Clinical trial management increases efficiency and helps shorten time-to-market for new therapies.
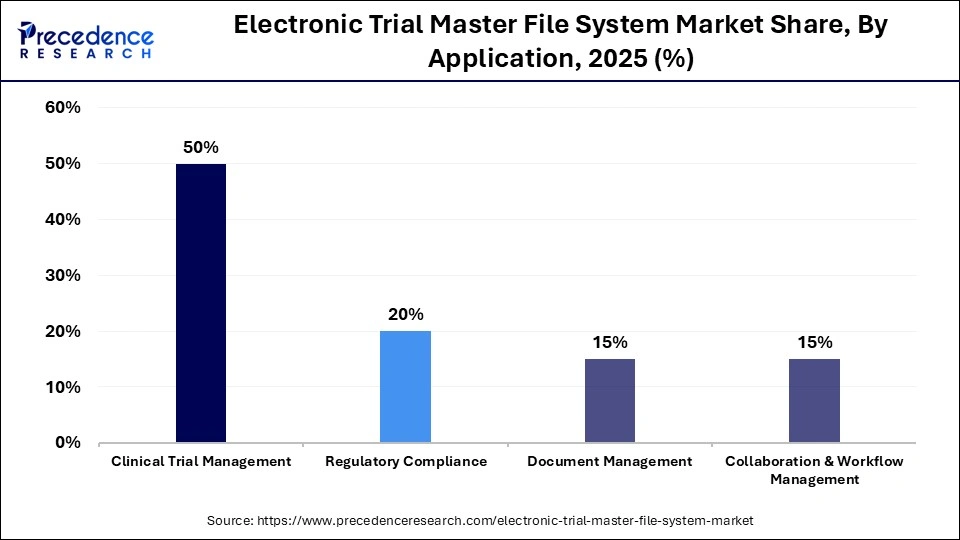
The collaboration & workflow management segment is expected to expand at the highest CAGR of 12.1% over the forecast period because it addresses the critical need for streamlined communication and task coordination among multiple stakeholders in clinical trials. As clinical trials involve more remote and international locations, the eTMF system provides centralized and instant collaboration to address the limitations of traditional paper-based systems. Workflow management tools automate tasks like document tracking, approvals, and notifications, significantly enhancing efficiency and reducing human error.
End User Insights
Which End User Dominated the Market in 2025?
The pharmaceutical & biotechnology companies segment dominated the electronic trial master file system market with a major share of 55% in 2025. The segment dominance is mainly driven by the rising need to manage complex clinical trials, increasing focus on meeting strict regulatory compliance standards, and high R&D expenditures to accelerate drug development. Pharmaceutical and biotech companies often face intense scrutiny to maintain comprehensive and audit-ready documentation. eTMF systems provide a secure and compliant document management platform.
Electronic Trial Master File System Market Share, By End User, 2025 (%)
| End User | 2025 |
| Pharmaceutical & Biotechnology Companies | 55% |
| Contract Research Organizations (CROs) | 25% |
| Academic & Research Institutes | 15% |
| Others | 5% |
The contract research organizations (CROs) segment is expected to grow at the fastest CAGR of 11.7% from 2026 to 2035, owing to the surge in outsourcing of clinical trials and the rising requirement for real-time compliance and collaboration across global sites. Pharmaceutical and biotechnology companies are increasingly outsourcing trials to CROs to speed up drug development and reduce costs. CROs handle multiple trials simultaneously, which increases the need for eTMF systems to manage documents across various sponsors and ensure data consistency. With the rise in stricter compliance regulations, CROs prefer eTMF solutions for audit readiness and secure document handling.
Regional Insights
North America Electronic Trial Master File System Market Size and Growth 2026 to 2035
The North America electronic trial master file system market size is estimated at USD 657.00 million in 2025 and is projected to reach approximately USD 2,275.00 million by 2035, with a 13.22% CAGR from 2026 to 2035.
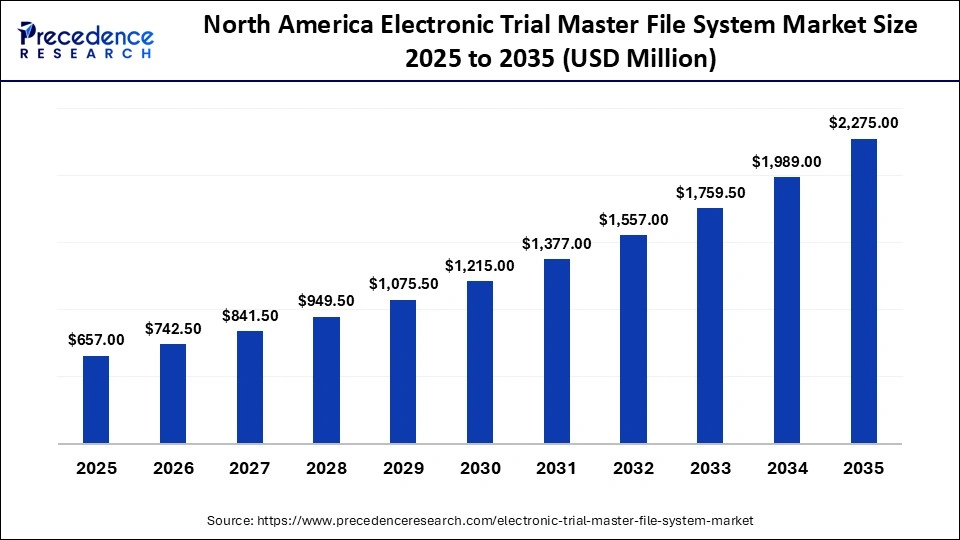
What Made North America the Dominant Region in the Electronic Trial Master File System Market?
North America dominated the global electronic trial master file system market, holding a major share of 45% in 2025. The region's dominance in the market is driven by the high concentration of pharmaceutical and biotechnology companies and CROs, creating strong demand for digital systems to manage clinical trial data. Stringent requirements from the U.S. FDA necessitate comprehensive and secure documentation, accelerating the adoption of advanced eTMF systems in the region. The rapid shift toward cloud-based digital solutions, a high volume of clinical trial data, and increasing investment in R&D activities ensure the leading position of the region in the market.
U.S. Electronic Trial Master File System Market Size and Growth 2026 to 2035
The U.S. electronic trial master file system market size is calculated at USD 492.75 million in 2025 and is expected to reach nearly USD 1,717.63 million in 2035, accelerating at a strong CAGR of 13.30% between 2026 and 2035.
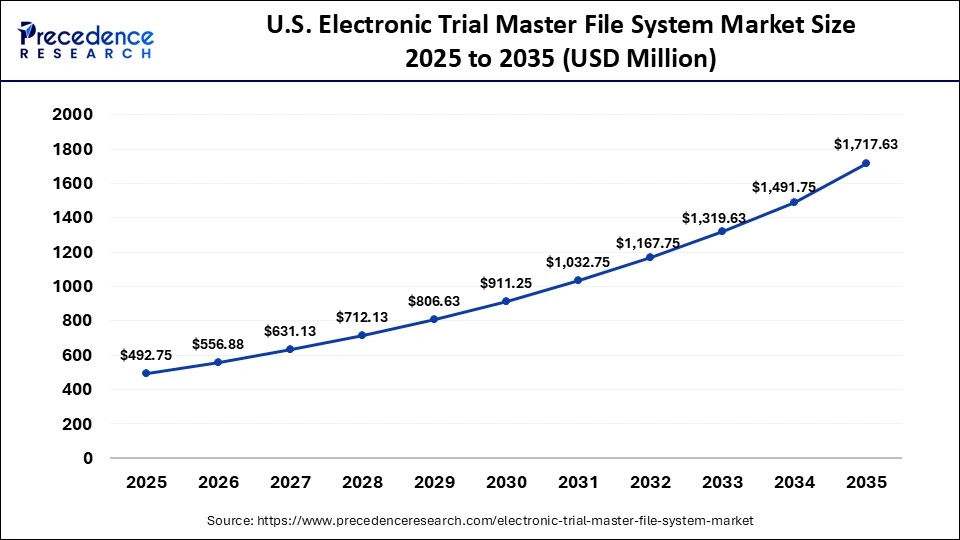
U.S. Electronic Trial Master File System Market Analysis
The U.S. holds a substantial share of the market and is the major contributor to the market. The country has a well-developed healthcare IT infrastructure. Significant investment in research and development provides the backbone for rapid technological advancements. The country is home to leading market players such as Veeva Systems, MasterControl Inc., Oracle Corporation, ArisGlobal, Parexel International, Merative, Xybion Corporation, ClinOne (Clindex), EXTEDO GmbH, and others. These companies are recognized for providing secure, validated, and compliant document management tools. Additionally, the increasing integration of Artificial Intelligence (AI) and Machine Learning (ML) assists in automating workflows and enhancing document quality control, which is expected to fuel the market's growth in the country.
How is the Opportunistic Rise of Asia Pacific in the Electronic Trial Master File System Market?
Asia Pacific is expected to grow at the fastest CAGR of 13.5% in the upcoming period. The rapid digitalization in the region has substantially increased the reliance on decentralized and hybrid clinical trials, combined with the increasing need to optimize costs and efficiency, supporting the move from paper-based to electronic systems. The high R&D investments, particularly in China, Japan, and India, along with the increasing volume of clinical trials, drive the need for eTMF systems to manage massive documentation for trials. Moreover, the rapid technological innovation and adoption of AI, machine learning, and cloud-based systems enable faster and more secure document management.
China Electronic Trial Master File System Market Analysis
The market in China is expanding, as the country is a hub for international and local clinical studies, leading to the spurring of the demand for advanced and compliant document management solutions. The country's growth is also supported by a strong and rapidly expanding pharmaceutical and biotechnology sector, which drives the demand for sophisticated and compliant document management systems. The rapid digitization, significant investment in modernizing clinical trial infrastructure, strict regulatory compliance, rise in the volume of clinical trials, rising integration of Artificial Intelligence (AI), and rapid shift to cloud-based solutions also drive the market in China.
Contract Research Organizations (CROs) in the country are increasingly adopting cloud-based eTMF solutions to handle multi-site trials, ensure compliance, and enhance collaboration across teams. In addition, the rising focus on compliance with ICH-GCP standards, which has compelled Chinese clinical trials to adopt advanced eTMF systems to manage documents and audit readiness, is bolstering the country's market growth during the forecast period.
Electronic Trial Master File System Market Companies
- Veeva Systems
- MasterControl Inc .
- Oracle Corporation
- Medidata Solutions (Dassault Systèmes)
- Phlexglobal
- Sparta Systems (Honeywell)
- ArisGlobal
- Parexel International
- IBM Watson Health
- BioClinica
- Complion
- eTMF Solutions
- Xybion Corporation
- ClinOne (Clindex)
- EXTEDO GmbH
Recent Developments
- In January 2026, Enlighten Clinical Solutions announced the official release of its integrated electronic Trial Master File (eTMF) module, expanding the company's clinical trial platform to support compliant, inspection-ready trial execution within a modern and unified system. The eTMF module is part of Enlighten Clinical Solutions' integrated clinical trial platform that is specifically designed to support inspection-ready trial execution by bringing trial documentation management and Electronic Data Capture (EDC) into a single and cohesive system.(Source: https://www.greenbaypressgazette.com )
- In September 2025, Arkivum announced a partnership with PHARMASEAL, the provider of the Engility Trial Management Platform, which consists of an Electronic Trial Master File (eTMF) and Clinical Trial Management System (CTMS). With the integration of Arkivum's GxP-validated digital archiving and preservation technology, PHARMASEAL's unified trial management platform offers an end-to-end trial technology solution. This new streamlined process for archiving data will encourage sponsors to consider retention requirements as a key part of their planning process at the beginning of the trial rather than after study closeout.(Source: https://www.contractpharma.com )
Segments Covered in the Report:
By Component
- Software
- Services
By Deployment Mode
- Cloud-Based
- On-Premises
By End User
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations (CROs)
- Academic & Research Institutes
- Others
By Application
- Clinical Trial Management
- Regulatory Compliance
- Document Management
- Collaboration & Workflow Management
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
