Ischemia-reperfusion injury challenges modern medicine by paradoxically harming cells when blood flow is restored. Advanced interventions include pre- and post-conditioning, pharmacological antioxidants, and regenerative therapies. Emerging approaches in nanomedicine, mitochondrial-targeted drugs, and stem cell therapy aim to minimize tissue damage, reduce arrhythmias, and optimize recovery, offering new hope for patients with myocardial infarction, stroke, or organ transplant complications.
Abstract
The damage of ischemia-reperfusion injury (IRI) is one of the most complicated paradoxes of contemporary medicine. The recovery of blood circulation in the tissue, which lacks oxygen, may paradoxically raise cellular damage. This effect is more specifically applicable in cardiovascular disease, stroke, organ transplant, and traumatic vascular cases. On the molecular level, ischemia causes disturbance in cellular energy metabolism, ion homeostasis, and mitochondrial activity, and reperfusion is accompanied by the development of oxidative stress, inflammatory cascades, and calcium overload.
Imagination Unleashed: A Journey Through Creativity
Ischemia results in a decrease or even an absence of blood supplying tissues, which leads to oxygen deficit and imbalance of metabolic processes. Once circulation is reestablished or reperfusion, as it is sometimes called, the onslaught of oxygen and nutrients can cause biochemical activities that destroy the already damaged cells. This twofold mechanism is referred to as IRI and is very important in the conditions that encompass myocardial infarction, stroke, kidney injury, and organ transplantation.
The pathophysiology of IRI involves many interconnected processes, including oxidative stress, mitochondrial dysfunction, inflammation, and calcium overload. All these mechanisms disrupt cellular homeostasis and lead to tissue damage. Conversely, current treatment strategies focus on alleviating these processes to preserve cell integrity and promote recovery.
Primeval Era to the Industrial Revolution: The History of Ischemia Research Development
Medical knowledge of circulatory disturbances and tissue injury has its basis in ancient medicine. Ancient doctors were able to identify the correlation between blood circulation and vitality despite the lack of understanding of the underlying molecular mechanisms of ischemic trauma. The practice involving anesthesia emerged gradually in early medical history, and this gave the physicians the opportunity to carry out surgical operations that exposed the effects of limited blood supply to body tissues.
Anatomists and physiologists had to record the significance of circulation in supporting life during the Renaissance and early scientific revolution. The industrial revolution also stimulated further advancement in the field of medicine, with the emergence of experimental physiology and better surgical methods. Scientists started to notice that the tissues with a lack of blood contained cell degeneration and necrosis.
In the twentieth century, scientists realized that the electrolytes bringing blood back to ischemic tissue may be at times counterproductive to injury, which resulted in the so-called IRI. Future developments in the field of molecular biology and biochemistry, and cardiovascular biology, made clear the mechanisms of oxidative stress, mitochondrial dysfunction, and inflammatory signaling pathways related to reperfusion injury.
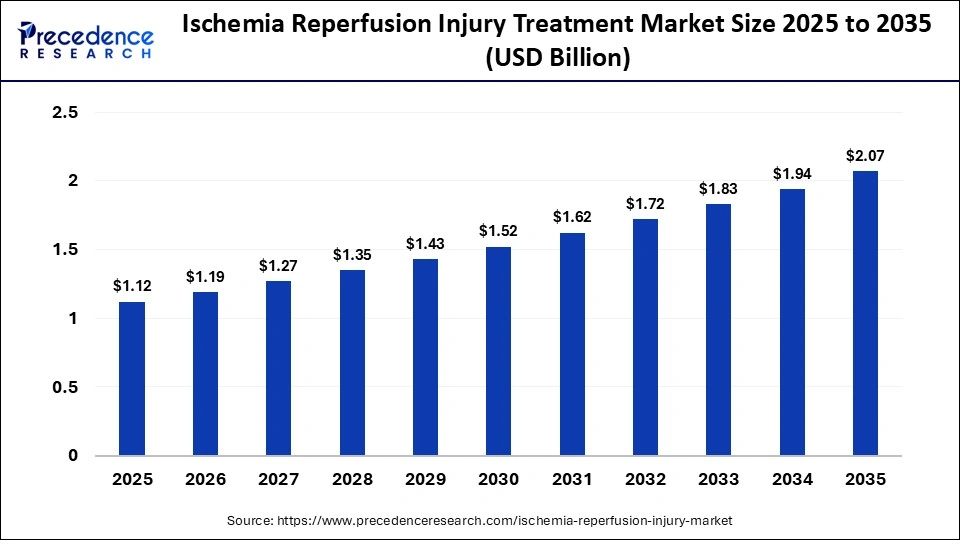
What is the Ischemia Reperfusion Injury Treatment Market Size in 2026?
The global ischemia reperfusion injury treatment market size accounted for USD 1.12 billion in 2025 and is predicted to increase from USD 1.19 billion in 2026 to approximately USD 2.07 billion by 2035, expanding at a CAGR of 6.32% from 2026 to 2035
Types of Cardiac Ischemia
Cardiac ischemia is a challenging condition where the coronary arteries struggle to supply enough blood, leading to insufficient oxygen delivery to the heart muscle. For some individuals, this can occur silently, meaning they do not experience noticeable symptoms, yet it still contributes to the gradual weakening of blood flow to the heart tissues. On the other hand, stable angina can be an unsettling experience, as it often manifests as predictable chest pain triggered by physical activity or stress, resulting from partial blockages in the arteries. It is important to recognize the impact of these conditions on individuals and to ensure they receive the care and support they deserve.
- Histopathology of CI/RI: Serious structural alterations at both cellular and subcellular levels can be observed on the histopathological analysis of the ischemic tissues. Ischemia leads to swelling, destruction of mitochondria, and the cell membrane in the cells. The deficiency of oxygen causes the accumulation of metabolic waste and loss of ATP. When it is exacerbated, further damage results because reactive oxygen species form quickly. These molecules attack cellular lipids, proteins, and DNA, resulting in further degeneration of tissues. The inflammatory cell infiltration, microvascular obstruction, and endothelial dysfunction are also among the special visible features under reperfusion injury.
- Energy metabolism in CI: Oxidative metabolism is vital to the heart to produce ATP that is used to sustain contraction. In a case of ischemia, lack of oxygen halts the mitochondrial oxidative phosphorylation, thus communities are compelled to use energy through anaerobic glycolysis. The metabolic change causes a reduction in ATP production and lactic acid accumulation in the cells. Lowered ATP levels damage ion pumps like the sodium-potassium ATPase, resulting in ion imbalance and swelling of the cells. The dysfunction of mitochondria also worsens cellular metabolism and makes it a contributing factor to oxidative stress during reperfusion.
- Therapeutic Strategies for the Restoration of Energy Metabolism: Therapeutic strategies for the restoration of energy metabolism aim to optimize cellular function and enhance overall health. These approaches often involve dietary interventions, targeted supplementation, and exercise programs that stimulate mitochondrial biogenesis and improve ATP production. By addressing underlying metabolic dysfunctions, these strategies can contribute to the prevention and management of various chronic diseases.
- Ca2+ Homeostasis: Calcium ions are important in the regulation of muscular contraction in the heart and cellular signalling. Isometric deprivation of ATP leads to the inhibition of calcium transport systems in cells. Consequently, the intracellular calcium concentration goes up tremendously. Calcium influx is further aggravated when reperfusion occurs, leading to the acute resumption of oxygen and metabolic activities. This calcium overload activates cell-destructive enzymes, including proteases and phospholipases, hence causing cellular damage.
Cardioprotective Strategies for Acute Myocardial Infarction
Cardioprotective strategies aim to reduce myocardial damage during and after ischemic events. Early reperfusion therapies such as thrombolysis and percutaneous coronary intervention remain the cornerstone of treatment. However, adjunct therapies targeting reperfusion injury are increasingly being developed to enhance patient outcomes.
Pharmacological agents, including antioxidants, anti-inflammatory drugs, and mitochondrial stabilizers, are being explored to reduce cellular damage during reperfusion. Additionally, ischemic preconditioning techniques and metabolic modulators have shown promise in improving myocardial resilience. Advances in regenerative medicine, including stem cell therapy and tissue engineering, also hold potential for restoring damaged cardiac tissue. These evolving strategies collectively represent the future of cardio protection in ischemia-related cardiovascular diseases.
Ischemic Pre-conditioning
Ischemic preconditioning is a form of the most impressive intrinsic heart defense process. This phenomenon was first noted in trials in the scientific literature of cardiology, and it is observed that short periods of non-lethal ischemic exposure of the myocardial tissue in experimental studies produce an enhancement of the myocardial tissue to the effects of subsequent extended ischemic attacks. These periods of transient ischemia trigger complicated intracellular signal transduction mechanisms that sensitize cells to oxygen deprivation.
Protective mediators that preconditioning activates on the molecular level include protein kinase C, mitochondrial ATP-sensitive potassium channels, and nitric oxide signaling. All these pathways ultimately maintain mitochondrial integrity and lessen the death of cells by apoptosis in the case of ischemic stress. Ischemic preconditioning enables cardiomyocytes to be better adapted to survive metabolic stress conditions, and thus ischemic preconditioning is an essential step in reducing the size of the infarct and enhancing myocardial survival.
Ischaemic Post-conditioning
One of the cardioprotective strategies is ischaemic post-conditioning, which occurs at the time of the reestablishment of blood flow in case of ischemia. The technique produces momentary death of the blood flow rather than permitting the application of a full reperfusion period to limit intermittent interruptions of blood flow during the initial stages of reperfusion.
These regulated cycles help reduce the sudden biochemical surge that tends to be part of reperfusion. Post-conditioning reduces oxidative stress, calcium overload, and mitochondrial membrane stabilization. It also regulates inflammatory signalling pathways that would otherwise increase myocardial damage. This approach thus assists in maintaining the cardiac contractile activity and reducing structural injury after acute myocardial infarction (AMI).
Remote Ischemic Conditioning
Remote ischemic conditioning is one of the most interesting systems of systemic adaptation where delayed ischemia of one of the far-off organs - the limb in most cases - causes the heart to protect itself. This is normally done by inflation of a blood pressure cuff, in a brief constraint of blood flow in an arm or leg. Signals protecting generated in peripheral tissues are conveyed by neural and humoral cytokines and eventually, the cardio-protectant systems of the myocardium are stimulated. These nymphids make mitochondria more resistant, decrease the oxidative stress, and tune down inflammatory processes. Remote conditioning is especially attractive in clinical practice since it is non-invasive and cost-effective.
Clinical Management of Myocardial Infarction Event
Clinical care of AMI involves the quick reestablishment of coronary artery blood circulation to save viable myocardium. Timely intervention can be implemented through early disease identification by performing electrocardiography and detecting cardiac biomarkers. Percutaneous coronary intervention and thrombolytic agents are still considered to be the mainstays of treatment as reperfusion therapies.
Coronary circulation is stabilized using adjunct pharmacological drugs such as antiplatelet agents, anticoagulants, beta-blockers, and statins with an anti-myocardial workload reduction effect. Early identification of complications, including arrhythmias and heart failure, is ensured due to intensive monitoring in special cardiac care units. Management of the long-term type entails lifestyle change, pharmacotherapy, and rehabilitation interventions aimed at preventing a repeat of the ischemic events.
Spectrum of Reperfusion Arrhythmia
Reperfusion arrhythmias are one of the electrical complications that commonly occur after termination of myocardial ischemia and the restoration of blood flow in these areas. These arrhythmias are caused by high metabolic changes, ionic changes, and short-term changes in cardiac conduction pathways. Reperfusion arrhythmias range benignly with pre-ventricular contractions with occurrence to severe forms of ventricular tachycardia and ventricular fibrillation. Even though most of the reperfusion arrhythmias are self-limiting and short-lasting, there are severe cases that may necessitate urgent use of pharmacological intervention or electrical intervention. It is necessary to know the electrophysiological processes of these disturbances can optimize post-reperfusion cardiac care.
Electrical Storm in Acute Coronary Syndromes
Acute coronary syndromes (ACS) are one of the most dramatic and life-threatening emergencies of cardiovascular disease. When the blood flow to the myocardium decreases, electrical stability in the heart may stop, and ACS can cause malignant arrhythmias and complex biochemical cascades. The electrical storm is one of the most dreaded complications, a condition that is manifested by recurrent cases of ventricular tachycardia or ventricular fibrillation within a brief period. Interpretation of electrophysiological alterations and molecular pathways related to ischemia provides better answers on how to manage emergencies alongside how to design a response approach in the next generation of therapies.
Some Common Therapeutic Regimens for IRI
- Completion RevascularizationImmediate revascularization is a highly important treatment goal among those with ACS. Clinicians are trying to salvage ischemic myocardium by reinstating resting blood flow through occluded coronary arteries to help prevent the additional electrical instability. Common procedures that are used to realize revascularization are percutaneous coronary intervention and coronary artery bypass graft. Effective reperfusion cannot just decrease the size of the foci of infarction, but it also normalizes the electrophysiological state of the heart, which minimizes the possibility of repeated arrhythmia. Rapid and extensive restoration in a coronary circulation is considered one of the key principles of contemporary cardiology.
- Death-Associated Protein Kinase (DAPK)Death-associated protein kinase (DAPK) is important in the regulation of apoptosis and programmed cell death of ischemic tissues. The activation of DAPK signalling pathways under calcium overload and oxidative stress also takes place during ischemic injury. It is a mitochondrion-based kinase that affects the mitochondrial integrity and enhances cascades of apoptotic signalling. Studies have indicated that DAPK inhibition has neuroprotective effects, decreasing the death of cells in the heart and brain. Therefore, the DAPK has become one of the promising molecular targets for the development of therapies to alleviate cellular damage caused by ischemia.
The Promising Future of the Industry
Biotechnology, precision medicine, and advanced cardiovascular therapeutic approaches are predicted to be crucial at the intersection of ischemia and reperfusion injury management in the future. New studies are underway to reveal specific molecular therapies that can regulate oxidative stress, mitochondrial dysfunctions, and inflammatory signalling. Regenerative medicine advances, such as stem cell therapy and tissue engineering, have the potential to cure damaged tissues of the myocardium and neural tissues. Clinical research is also incorporating AI in identifying new therapeutic targets and estimating treatment approaches. Furthermore, innovations in drug delivery systems, nanomedicine, and biomarker-based therapy should revolutionize the process of handling ischemic diseases. With further progress in science, the creation of extremely specific and custom-made treatment options is most likely to make the world of cardiovascular and neurological medicine a new one.
Conclusion
In conclusion, IRI represents a complex and multifaceted challenge within modern medicine, particularly in the contexts of cardiovascular disease, stroke, and organ transplantation. The paradoxical nature of reperfusion therapy, while essential for restoring blood flow, can inadvertently exacerbate cellular damage through mechanisms such as oxidative stress, inflammation, and calcium overload. As our understanding of the underlying molecular and cellular dynamics continues to evolve, it becomes increasingly important to develop targeted therapeutic strategies that mitigate these harmful processes.
By focusing on innovative pharmacological agents, mitochondrial protection, and metabolic regulation, we can strive to minimize reperfusion injury and improve clinical outcomes. The historical journey of ischemia research highlights the progress made over the centuries and underscores the need for ongoing investigation and interdisciplinary collaboration to enhance patient care and treatment efficacy in the face of these intricate pathophysiological challenges.
Expert Advise
Our research concludes that with the rising burden of severe health illnesses across the globe among people of all age groups, the risk of IRI increases significantly. Novel treatment regimens are underway, including drug delivery systems, medical devices, and surgical procedures. Some emerging therapies include cell therapy, gene therapy, mitochondrial-targeted drugs, and nitric oxide donors. Governments are making constant efforts to build a robust healthcare infrastructure and develop favorable reimbursement policies to enhance the accessibility and affordability of complex IRI treatment in the near future.
About the Authors

Aditi Shivarkar
Aditi, Vice President at Precedence Research, brings over 15 years of expertise at the intersection of technology, innovation, and strategic market intelligence. A visionary leader, she excels in transforming complex data into actionable insights that empower businesses to thrive in dynamic markets. Her leadership combines analytical precision with forward-thinking strategy, driving measurable growth, competitive advantage, and lasting impact across industries.

Aman Singh
Aman Singh with over 13 years of progressive expertise at the intersection of technology, innovation, and strategic market intelligence, Aman Singh stands as a leading authority in global research and consulting. Renowned for his ability to decode complex technological transformations, he provides forward-looking insights that drive strategic decision-making. At Precedence Research, Aman leads a global team of analysts, fostering a culture of research excellence, analytical precision, and visionary thinking.

Piyush Pawar
Piyush Pawar brings over a decade of experience as Senior Manager, Sales & Business Growth, acting as the essential liaison between clients and our research authors. He translates sophisticated insights into practical strategies, ensuring client objectives are met with precision. Piyush’s expertise in market dynamics, relationship management, and strategic execution enables organizations to leverage intelligence effectively, achieving operational excellence, innovation, and sustained growth.





 Request Consultation
Request Consultation

