Pyrogen testing is essential for detecting fever-causing contaminants in pharmaceuticals and medical devices. The field is rapidly evolving toward advanced, ethical, and technology-driven methods that improve accuracy, safety, and regulatory compliance.
Abstract
Pyrogen testing is an important protection mechanism in pharmaceutical and biomedical fields that must guarantee the absence of contaminants of drugs and other medical gadgets injected into the body that cause fever. It has developed from primitive animal observations to highly advanced in vitro and molecular methods. This article discusses the development of pyrogen testing throughout history, its scientific practices, and the newer inventions that are developing the future of pyrogen testing. It also emphasizes the increasing shift to the ethical, animal-free, and technology-based solutions.
Executive Summary
Pyrogen tests have evolved beyond the use of rabbits to the use of a modern biochemical and cell-based test like the bacterial endotoxin test (BET), monocyte activation test (MAT), etc. The growing sophistication of biologics and gene therapies has increased the demand for very sensitive and sound detection systems. Answering the question of the measurement of the pyrogenic response on a molecular scale, emerging technologies such as RNA sequencing and flow cytometry are reshaping the concepts of this approach. The transition to recombinant and animal-free systems is hastened by regulatory pressure and issues of sustainability. Automation, AI, and human-relevant models are the future of pyrogen testing.
Guardians against the Invisible: Why Pyrogen Testing Matters More Than Ever
Although not visible, pyrogens can cause drastic immune reactions if they are introduced into the human body with the help of contaminated pharmaceuticals. They are not only a regulatory requirement but also a serious provider of patient safety and therapeutic success. The significance of accurate, timely, and ethical pyrogen testing has been increasing alongside the development of modern medicine into the area of complex biologics and personalized therapy.
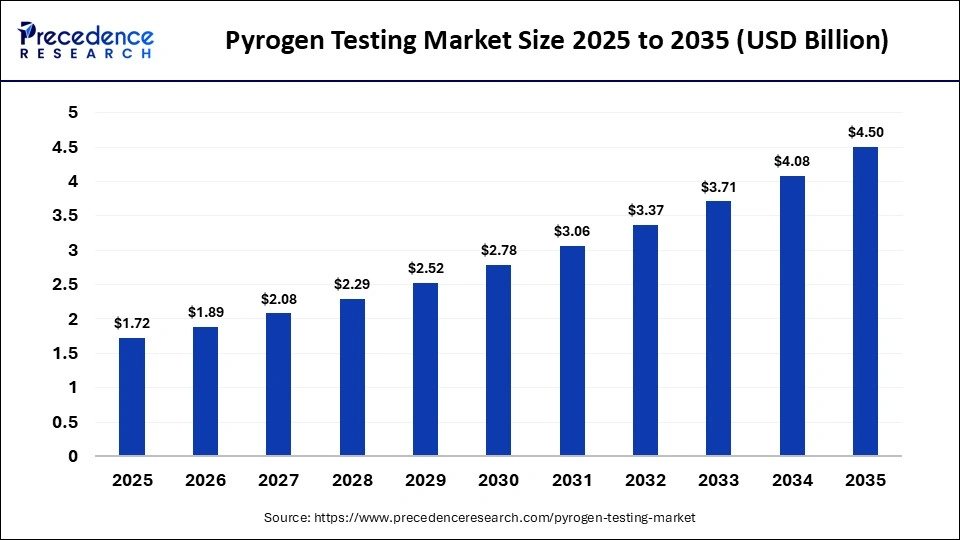
What is the Pyrogen Testing Market Size in 2026?
The global pyrogen testing market was valued at USD 1.72 billion in 2025 and is projected to grow from USD 1.89 billion in 2026 to approximately USD 4.50 billion by 2035, registering a CAGR of 10.10% during the forecast period from 2026 to 2035. The market serves as a critical indicator of safety for pharmaceutical products, medical devices, and biologics by identifying fever-inducing contaminants. With increasing regulatory scrutiny and the growth of biologics production, pyrogen testing has become a cornerstone of quality assurance in the life sciences sector.
History of the Pyrogen Test: Prehistoric Period until the Industrial Revolution
The history of pyrogen testing can be traced to the era of medical practice, when the response of fever was associated with inoculations that were contaminated. During the pre-industrial period, there was an informative state of contamination that was poorly informed and based on symptomatic results and not on scientific validation. During the industrial revolution and the development of injectable medications, the necessity of standardized safety testing was realized.
Rabbit Pyrogen Test (RPT) became one of the first formalized techniques in which physiological reactions in animals are utilized to identify pyrogens. In the course of time, microbiology and biochemistry made improvements, with the discovery of endotoxins and the development of the Limulus Amebocyte Lysate (LAL) test, which was a step towards more accurate and reproducible methods of testing. This development formed the basis of contemporary in vitro and recombinant technology.
Types of Pyrogen Testing
Most pyrogen testing techniques can be categorized into animal-based tests, in vitro biochemical tests, and sophisticated cell-based or molecular tests. All methods are different in terms of sensitivity, specificity, ethical factors, and acceptability by regulations. Animal testing was historically based on controlled experimental tests with rabbits or other species.
These procedures involved close observation of physiological conditions such as body temperature. Although these hold historical significance, varying outcomes and moral issues regarding their use have led to a gradual discontinuity in their use. Conventional assays, such as RPT, are based on physiological reactions, and the BET is based on biological reagents to detect endotoxins. Newer methodologies, like MAT and recombinant assays, are meant to stimulate human immune responses in a better manner.
Rabbit Pyrogen Test (RPT): RPT is a test that is conducted by injecting a sample into rabbits and recording how their bodies react to the sample's temperature. Although it has been the gold standard over the decades, it has some limitations, such as the ethics, poor specificity, and inability to detect certain pyrogens.
Bacterial Endotoxin Test (BET) and Monocyte Activation Test (MAT): BET is based on the LAL reagent that is produced by horseshoe crab blood, which is highly sensitive to the detection of endotoxins. Its use in the pharmaceutical quality control is universal. MAT, in turn, employs human immune cells to identify an expanded spectrum of pyrogens, and as such, it can be considered a more global and ethical solution.
Pyrogen Testing: Plan of Action
- RNA Sequencing: RNA sequencing has become an effective instrument to elucidate the molecular reaction to pyrogens. It allows revealing the changes in gene expression induced by endotoxins or other pyrogenic materials. This method has a full picture of immune activation pathways with more information. It is applicable especially in the research field and sophisticated biologics testing.
- Sequencing and Preparation of Libraries: Preparation of the library RNA sequencing is an important process, and it requires the transformation of RNA into complementary DNA (cDNA) and later amplification. Sequencing platforms then produce bulk data that are signatures of gene expression. This is because proper library preparation is used to ensure that subtle changes in immune-related genes are detected reliably, which are an indication of pyrogenic activity.
- Data Processing and Data Analysis: The sequencing data produced is massive and thus needs strong bioinformatics tools to analyze the data. Data processing refers to the quality control of the processed data, its alignment to the reference genomes, and the measurement of the gene expression.
Regulatory Compliance
Compliance with regulatory guidelines is paramount in the biopharma and plasma industry. Organizations such as the FDA and EMA provide specific frameworks for pyrogen testing methodologies. Manufacturers must demonstrate that their testing procedures are validated and consistently yield reliable results.
Implications for Product Safety
The implications of rigorous pyrogen testing extend beyond regulatory compliance; they play a vital role in patient safety. Failure to adequately assess and manage pyrogen risks can lead to severe adverse events, including febrile reactions and legal repercussions for manufacturers.
| Testing Method | Description | Advantages | Disadvantages |
| RPT | Involves injecting the product into rabbits and monitoring temperature changes. | Long-standing regulatory approval. | Ethical concerns; time-consuming. |
| LAL | Uses horseshoe crab blood to detect endotoxins through clotting. | Highly sensitive; rapid results. | Dependence on animal by-product |
| MAT | Assesses human monocyte response to pyrogens directly. | No animal testing; human-relevant results. |
May require complex validation steps; potentially higher costs. |
The LAL or the BET tests are widely used to test pyrogenicity in the world, with both making up about 80-90% of all pyrogenicity tests. This has been influenced by its sensitivity, acceptance by regulatory bodies, and its applicability to normal quality control in pharmaceutical manufacturing, hence its wide adoption. The procedure is widely applied in large areas such as North America, Europe, and Asia, where tests are carried out by millions every year. Nevertheless, even with its dominance, the LAL method is slowly under attack by some of the emerging recombinant substitutes because of sustainability issues that surround horsehoe crab-derived reagents.
RPT, which is regarded as the gold standard occupy a much lesser portion of approximately 10-15% of the world and is steadily on the decline. It is estimated that about 400,000 rabbits continue to be used in a year to test pyrogens, but numbers have reduced in some areas, such as Europe, due to compliance with a phase-out of regulation and associated ethical reasons. RPT is still applied in some situations, especially where non-endotoxin pyrogens need to be identified, and alternative tests, which have been validated, are yet to be enacted. Its drawbacks, such as variability, ethical issues, and reduced specificity, are, however, hastening the rate of its replacement.
Although it represents a minority of 1-5% of the overall usage now, the MAT is the fastest-rising category of pyrogen testing. The fact that it can detect not only endotoxin but also non-endotoxin pyrogens through human immune responses is very relevant to modern biologics and advanced therapies. Europe shows a great rate of adoption, especially in situations where regulatory authorities encourage the use of animal-free testing techniques. With the increased regulatory acceptance throughout the world and the growing focus of pharmaceutical firms on ethical and full-spectrum testing methods, MAT will have a strong momentum and change the future of pyrogen testing.
Future of the industry
The pyrogen testing industry is on the brink of significant transformation, driven by advancements in technology, regulatory changes, and an increasing emphasis on ethical considerations. As the demand for safer pharmaceuticals and biomedical devices rises, the future will likely see a greater reliance on innovative, animal-free testing methods. The shift from traditional animal-based tests towards in vitro and recombinant technologies will be pivotal in enhancing accuracy and reliability in pyrogen detection.
One of the most promising developments is the integration of molecular techniques, such as RNA sequencing and advanced biosensors, which enable precise monitoring of immune responses at a genetic level. These molecular methods not only enhance sensitivity but also provide a wealth of data that can lead to a deeper understanding of pyrogenic responses. Automation and AI will play a crucial role in processing large datasets and optimizing testing protocols, potentially reducing the time and cost associated with pyrogen testing.
Moreover, the regulatory landscape is evolving, with authorities increasingly prioritizing ethical testing practices. This change is likely to accelerate the adoption of human-relevant models, which simulate human immune responses more accurately than traditional methods. As sustainability becomes a critical concern across industries, the pyrogen testing field will need to adopt greener practices, minimizing waste and resource usage.
Collaboration between academia and industry will also shape the future of pyrogen testing. Partnerships can facilitate the sharing of knowledge and resources, driving innovations that push the boundaries of current testing methodologies. Additionally, the emergence of personalized medicine will necessitate the development of tailored pyrogen testing strategies that account for individual patient responses.
Conclusion
Pyrogen testing is in a groundbreaking change with the abandonment of traditional methods of testing, which relied on animals, to the innovative, human-relevant, and technology-driven tests. Although the LAL test remains dominant through its regulatory approval and familiarity in its working theory, it faces the threat of being replaced by sustainability issues and the emergence of recombinant substitutes in the long term. Meanwhile, the RPT is gradually becoming increasingly irrelevant, with the phase-out of the test hastened by both the ethical aspects and regulatory factors.
The future of the industry is defined by the high rate of adoption of MAT, recombinant factor C assays, and integrated automated platforms with greater sensitivity, reproducibility, and efficiency. The increase in complexity of biologics, cell and gene therapies, and personalized medicine further supports this change as both require more complex and comprehensive strategies to detect pyrogens.
Simultaneously, with the growth of regulatory endorsement of animal-free testing, AI growth, data analytics, and molecular technologies, quality management structures are being reorganized throughout pharmaceutical manufacturing. Businesses that invest in innovation, sustainability, and compliance will be in better positions to be on top of this changing situation. There is a shift in pyrogen testing towards a future where precision, ethics, and the combination of technology will be united, which can guarantee safer therapeutics without going against international sustainability and regulations.
Expert Advise
According to our analysis, the need for pyrogen testing is increasing at a rapid pace, with advancements in testing methods, stringent regulatory guidelines, and evolving molecular techniques. Stakeholders benefit from pyrogen testing through enhanced safety and reduced risk for patients, ability to offer high-sensitivity and consistent testing. The shifting trend towards in vitro testing methods and the reduction in the use of animal models potentiate the demand for pyrogen testing.
About the Authors

Aditi Shivarkar
Aditi, Vice President at Precedence Research, brings over 15 years of expertise at the intersection of technology, innovation, and strategic market intelligence. A visionary leader, she excels in transforming complex data into actionable insights that empower businesses to thrive in dynamic markets. Her leadership combines analytical precision with forward-thinking strategy, driving measurable growth, competitive advantage, and lasting impact across industries.

Aman Singh
Aman Singh with over 13 years of progressive expertise at the intersection of technology, innovation, and strategic market intelligence, Aman Singh stands as a leading authority in global research and consulting. Renowned for his ability to decode complex technological transformations, he provides forward-looking insights that drive strategic decision-making. At Precedence Research, Aman leads a global team of analysts, fostering a culture of research excellence, analytical precision, and visionary thinking.

Piyush Pawar
Piyush Pawar brings over a decade of experience as Senior Manager, Sales & Business Growth, acting as the essential liaison between clients and our research authors. He translates sophisticated insights into practical strategies, ensuring client objectives are met with precision. Piyush’s expertise in market dynamics, relationship management, and strategic execution enables organizations to leverage intelligence effectively, achieving operational excellence, innovation, and sustained growth.





 Request Consultation
Request Consultation

