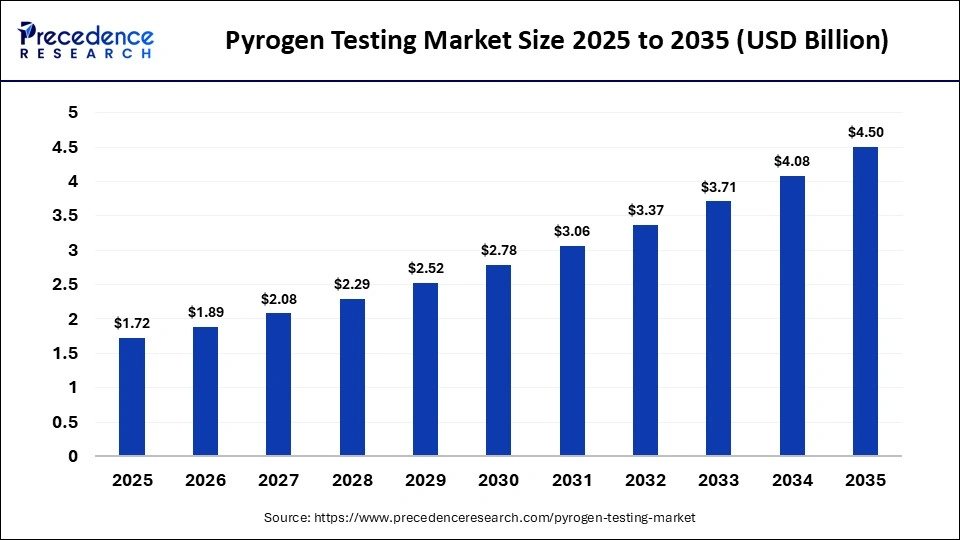
What is the Pyrogen Testing Market Size in 2026?
The global pyrogen testing market size was calculated at USD 1.72 billion in 2025 and is predicted to increase from USD 1.89 billion in 2026 to approximately USD 4.50 billion by 2035, expanding at a CAGR of 10.10% from 2026 to 2035. The market is a key indicator of safety for pharmaceutical products, medical devices, and biologics through the identification of fever-inducing contaminants. With regulatory scrutiny being stricter and biologics production growing, pyrogen testing has become the foundation of quality assurance in the life science sector.
Key Takeaways
- North America dominated the global pyrogen testing market, holding a share of 38.60% in 2025.
- Europe is expected to grow at the fastest CAGR in the market during the forecast period.
- By test type, the limulus amoebocyte lysate testing segment held a dominant position in the market by holding a share of 67.80% in 2025.
- By test type, the monocyte activation test segment is expected to grow at the fastest CAGR of 10.80% in the market between 2026 and 2035.
- By offering type, the reagents and kits segment led the global market by holding a share of 51.70% in 2025.
- By offering type, the services segment is expected to grow with the highest CAGR of 9.60% in the market during the studied years.
- By application, the pharmaceutical products testing segment held the largest pyrogen testing market share of 41.60% in 2025.
- By application, the cell and gene therapy products testing segment is expected to expand rapidly in the market with a CAGR of 12.40% in the coming years.
- By end user, the pharmaceutical companies segment registered its dominance over the global market with a share of 46.80% in 2025.
- By end user, the contract research organizations segment is expected to witness the fastest growth in the market with a CAGR of 10.20% over the forecast period.
- By technology, the gel clot LAL method segment contributed the biggest revenue share of 37.20% in the market in 2025.
- By technology, the recombinant factor C assay segment is expected to gain the highest market share with a CAGR of 12.60% between 2026 and 2035.
- By sample type, the injectable drugs segment accounted for the highest revenue share of 38.40% in the market in 2025.
- By sample type, the vaccines segment is expected to show the fastest growth with a CAGR of 11.70% over the forecast period.
Which Factors Drive the Pyrogen Testing Market?
The pyrogen testing industry is currently experiencing robust growth because of the complexity of drug formulations and the globalization of pharmaceutical manufacturing . The Limulus Amebocyte Lysate (LAL) test and the recombinant factor C (rFC) assays represent more sensitive and ethically acceptable methods to replace traditional methods, such as the Rabbit Pyrogen Test. Market players are concentrating on innovation, collaboration, and capacity building to satisfy the increasing demand in the market in the pharmaceutical and biotechnology sectors.
How is AI Influencing the Pyrogen Testing?
Artificial intelligence (AI) continues to find its way into the workflow of pyrogen testing to improve the precision, efficiency, and predictability of test outcomes. AI-based data analytics could discover the pattern of contamination, optimize testing procedures, and decrease false positives or negatives in assay outcomes. Machine learning models are also being applied to simplify quality control measures by predicting the risk of endotoxin contamination based on prior data. The automation of manufacturing with the use of AI is facilitating live monitoring of production settings and the decision-making process. In addition, AI facilitates digital validation and regulatory compliance by ensuring consistent documentation and traceability of testing processes.
Pyrogen Testing Market Trends
- Shift to Animal-Free Testing: The industry is experiencing a high rate of replacement of animal-based testing, like rabbit pyrogen testing, with in vitro testing. The use of recombinant and synthetic testing solutions is being encouraged by regulatory authorities and ethics.
- Second Wave of Recombinant Technology: Recombinant factor C (rFC) tests are increasingly becoming a viable and dependable substitute for conventional LAL tests. These technologies do not rely on horseshoe crab blood, which guarantees stability in supply and protection of the environment.
- Robotization and Technology Modifications: Pyrogen testing laboratories are automating and decreasing human error, increasing throughput, and enhancing reproducibility. High-tech tools and built-in computer programs are simplifying tasks.
- Expanding Biologics Market: The shifting trend towards biologics , vaccines , and cell and gene therapies is causing the demand for sensitive pyrogen detection techniques to rise substantially because of their intricacy.
- Outsourcing to CROs: Pharmaceutical firms are also outsourcing pyrogen testing services to contract research organizations (CROs) as a cost-saving strategy, specialized expertise, and improved time-to-market strategy.
- Increased R&D Investment: Increased investment in drug discovery and development is driving the need to develop more sophisticated quality control techniques, such as pyrogen testing, in all periods of the pharmaceutical pipeline.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.72 Billion |
| Market Size in 2026 | USD 1.89 Billion |
| Market Size by 2035 | USD 4.50 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 10.10% |
| Dominating Region | North America |
| Fastest Growing Region | Europe |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Test Type, Offering Type, Application, End User, Technology, Sample Type, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Need for Pharmaceutical Product Safety
The increasing focus on pharmaceutical product safety is a significant driver for the market. Regulatory bodies worldwide, such as the FDA and EMA, are enforcing stringent guidelines on endotoxin testing to ensure the safety of medical devices and biopharmaceuticals. This growing demand for reliable testing methods is propelling the adoption of advanced pyrogen testing technologies across the industry.
Restraint
High Costs
Despite the growth outlook, the high costs associated with pyrogen testing methodologies can be a considerable restraint for smaller pharmaceutical companies and laboratories. The complexity and need for specialized equipment can deter these organizations from implementing rigorous testing protocols. Additionally, the lack of trained personnel skilled in advanced testing methods further adds to the challenges within this market.
Opportunity
Development of Biologics and Biosimilars
The rise of biologics and biosimilars presents significant growth opportunities for the market. As more companies venture into this sector, the demand for comprehensive testing solutions will likely increase, creating a favorable environment for innovation and development of state-of-the-art testing technologies. Furthermore, advancements in rapid testing methods can enhance the efficiency of detecting pyrogens, making it attractive for market players to invest in such innovations.
Segmental Insights
Test Type Insights
Pyrogen Testing Market Share, By Test Type, 2025-2035 (%)
| Test Type | 2025 | 2035 |
| Limulus Amebocyte Lysate Testing | 67.80% | 54.20% |
| Monocyte Activation Test | 18.60% | 32.70% |
| Rabbit Pyrogen Test | 13.60% | 13.10% |
Why the Limulus Amebocyte Lysate Testing Segment Dominated the Pyrogen Testing Market?
The limulus amebocyte lysate testing segment dominated the market by holding the largest share of 67.80% in 2025, as it is regulatory approved and is a reliable test in the detection of endotoxins. It is a popular tool in the pharmaceutical and medical device sectors due to its extreme sensitivity and standard procedures. The fact that the method is compatible with different types of samples also contributes to its usage in the process of quality control. Also, the fact that it is a part of the major pharmacopeia recommendations is still a great way of consolidating its position in the market.
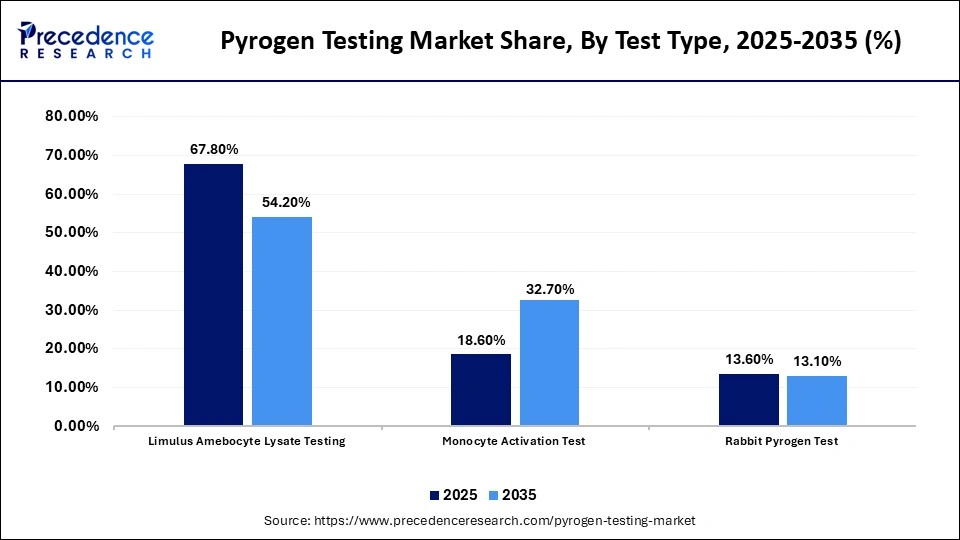
The monocyte activation test segment is expected to show the fastest growth with a CAGR of 10.80% in the forecasted period, due to the rise in the use of animal-free and human-relevant tests. Compared to traditional assays, MAT has the advantage of identifying both endotoxin and non-endotoxin pyrogens. It is being adopted faster due to regulatory backing of alternative ways and ethical care. Moreover, its future potential is improved by the fact that it can be used in more complex biologics and formulations.
Offering Type Insights
Pyrogen Testing Market Share, By Offering Type, 2025-2035 (%)
| Offering Type | 2025 | 2035 |
| Reagents and Kits | 51.70% | 46.60% |
| Instruments and Readers | 18.30% | 16.80% |
| Consumables | 14.60% | 13.70% |
| Services | 15.40% | 22.90% |
How the Reagents and Kits Segment Dominated the Pyrogen Testing Market?
The reagents and kits segment held a dominant market share of 51.70% in 2025, as they are important consumables needed to conduct routine pyrogen tests throughout all laboratories. Their frequent use guarantees them a consistent demand, particularly in large throughput pharmaceutical processing facilities. To make efficient and minimize waste, manufacturers are emphasizing the stability of reagents and their shelf life. The ease of use of kits that are ready-to-use also makes the testing processes easier, and this makes them very popular.
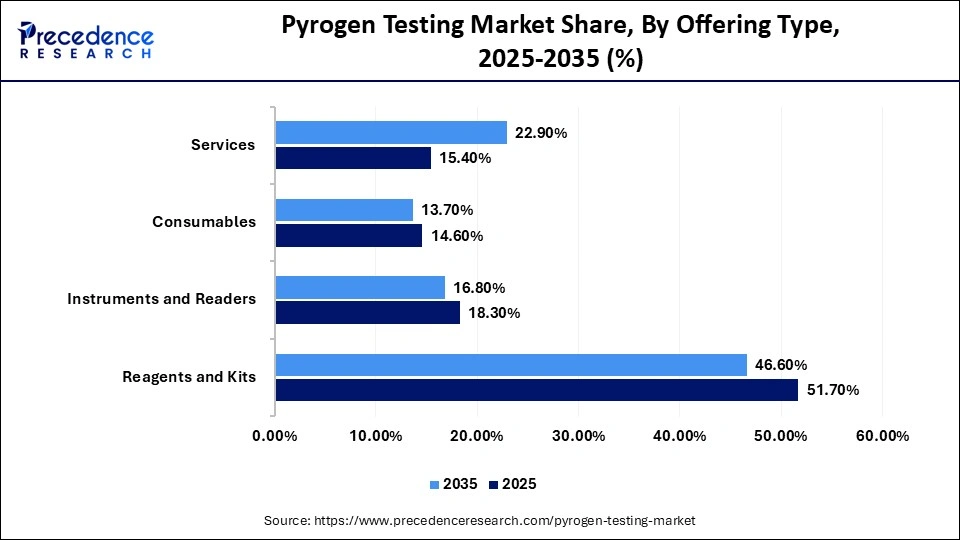
The services segment is expected to gain the highest market share with a CAGR of 9.60% between 2026 and 2035, driven by the rising outsourcing of tests to specific laboratories and CROs. Pharmaceutical companies favor the external service providers in order to lower the infrastructure expenses and to gain technical skills. Service providers provide all-around solutions, such as testing of the method, compliance with the regulations, and fast testing. The increasing complexity of drug products is also providing a reason that companies should be encouraged to use specialized testing services.
Application Insights
Which Application Segment Led the Pyrogen Testing Market?
The pharmaceutical products testing segment led the market with a share of 41.60% in 2025, because of the mandatory pyrogen testing related to injectable drugs and sterile formulations. Strict regulatory systems require strict endotoxin testing prior to approval and release of products in batches. The prevalence of this segment is facilitated by the large number of pharmaceutical production facilities spread across the world. Also, the rising need for safe and effective therapeutics is still increasing to meet demand.
Pyrogen Testing Market Share, By Application, 2025-2035 (%)
| Application | 2025 | 2035 |
| Pharmaceutical Products Testing | 41.60% | 35.20% |
| Biologics and Biotechnology Products Testing | 24.70% | 23.60% |
| Medical Devices Testing | 16.40% | 15.30% |
| Cell And Gene Therapy Products Testing | 9.80% | 16.70% |
| Vaccine Testing | 7.50% | 9.20% |
The cell and gene therapy products testing segment is expected to witness the fastest growth in the market with a CAGR of 12.40% over the forecast period, due to the blistering development of higher-order therapies. Special and sensitive pyrogen testing is needed in such therapies because they are complex in biology. The conventional technique has, in most cases, failed, and more advanced techniques such as MAT and recombinant technologies are therefore being embraced. Increased rate of clinical trials and approvals of gene therapies is also increasing the demand in this segment.
End User Insights
Why Did the Pharmaceutical Companies Segment Dominate the Pyrogen Testing Market?
The pharmaceutical companies segment contributed the biggest market share of 46.80% in 2025, as they perform comprehensive tests of quality control during drug development and manufacture. Better timeline and regulatory compliance can be conducted in-house. This will provide the pharmaceutical industry with sustained demand due to the constant expansion of the industry.
Pyrogen Testing Market Share, By End User, 2025-2035 (%)
| End User | 2025 | 2035 |
| Pharmaceutical Companies | 46.80% | 41.20% |
| Biotechnology Companies | 21.70% | 20.40% |
| Contract Research Organizations | 18.60% | 24.10% |
| Academic And Research Institutes | 7.40% | 7.30% |
| Medical Device Manufacturers | 5.50% | 7.00% |
The contract research organizations segment is expected to expand rapidly in the market with a CAGR of 10.20% in the coming years, because of the escalating outsourcing tendencies. Services offered by CROs are specialized testing, which allows pharmaceutical companies to devote resources to competitive activities. They are also desirable partners because they provide scalability, high technologies, and regulatory know-how. Clinical trials and drug development activities are also growing at a rapid pace due to the increasing globalization of these processes.
Technology Insights
How the Gel Clot LAL Method Segment Dominated the Pyrogen Testing Market?
The gel clot LAL method segment accounted for the highest market share of 37.20% in 2025, due to its ease of use, affordability, and compliance with regulatory authorities. It has many applications in the detection of endotoxins in the normal operations of pharmaceutical quality control laboratories. The procedure does not involve complicated instrumentation and is, therefore, available to most facilities. Its reliability, along with adherence to the standards of pharmacopeia, still helps it to occupy the first position.
Pyrogen Testing Market Share, By Technology, 2025-2035 (%)
| Technology | 2025 | 2035 |
| Gel Clot LAL Method | 37.20% | 27.60% |
| Turbidimetric LAL Method | 26.40% | 23.30% |
| Chromogenic LAL Method | 22.80% | 21.10% |
| Recombinant Factor C Assay | 13.60% | 28.00% |
The recombinant factor C assay segment is expected to grow with the highest CAGR of 12.60% in the market during the studied years, as this assay is sustainability and innovation-driven. It does not require hose-shoed crab-derived reagents; this has been handled regarding environmental and supply issues. The assay is very sensitive and consistent, and thus is applicable to the present-day pharmaceutical practice. It is being rapidly increased by regulatory acceptance and adoption by the industry.
Sample Type Insights
Which Sample Type Segment Dominated the Pyrogen Testing Market?
The injectable drugs segment registered its dominance over the market with a share of 38.40%, because the endotoxin testing is strictly required for parenteral formulations. These drugs enter the blood, and the utmost safety levels are required. As injectable drugs directly enter the bloodstream, monitoring of the presence of pyrogens is of utmost importance. Ongoing production of injectable therapies is another source of growth in the demand for pyrogen testing.
Pyrogen Testing Market Share, By Sample Type, 2025-2035 (%)
| Sample Type | 2025 | 2035 |
| Injectable Drugs | 38.40% | 33.60% |
| Implantable Medical Devices | 20.70% | 18.60% |
| Parenteral Nutrition Products | 15.40% | 13.70% |
| Dialysis Products | 11.30% | 9.80% |
| Vaccines | 14.20% | 24.30% |
The vaccines segment is expected to grow at the fastest CAGR of 11.70% in the market between 2026 and 2035, due to the growing number of immunization programs around the world and pandemic preparedness efforts. The rush in the development and production of vaccines has greatly amplified the demand for strict pyrogen testing. Highly sensitive testing methods are needed with advanced vaccine platforms such as mRNA and recombinant vaccines. This segment is still growing with the global health initiatives and government funding.
Regional Insights
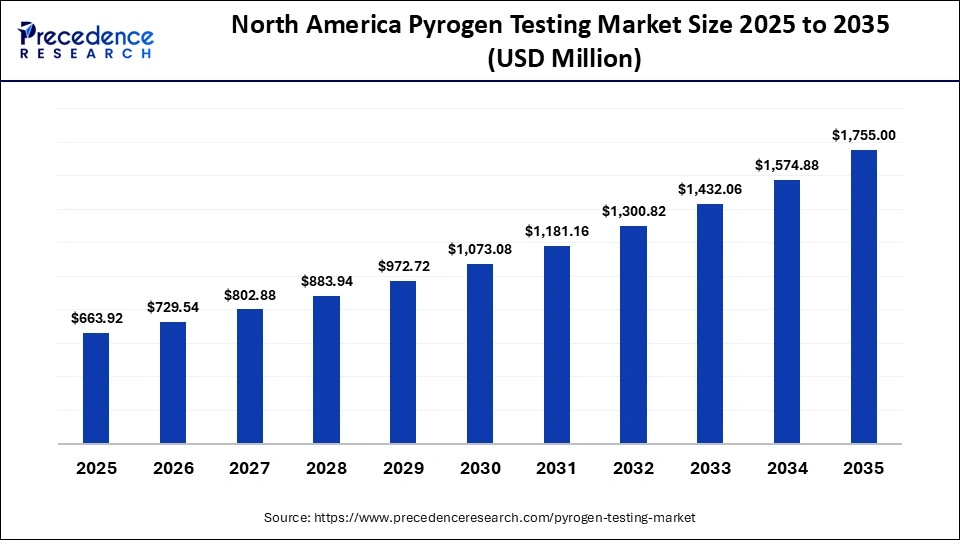
North America Pyrogen Testing Market Size and Growth 2026 to 2035
The North America pyrogen testing market size is estimated at USD 663.92 million in 2025 and is projected to reach approximately USD 1,755.00 million by 2035, with a 10.21% CAGR from 2026 to 2035.
Why North America Dominated the Pyrogen Testing Market?
North America held a major market share of 38.60% in 2025, driven by stringent regulatory measures, which require strict endotoxin and pyrogen testing of all injectable and sterile products. High concentration of top biopharmaceutical companies and high-end manufacturing plants also acts as an advantage to the demand for testing solutions on a regular basis.
In addition, the region has high spending on R&D and the introduction of innovative technologies, such as recombinant assays and automation, which reinforce market leadership. The increasing number of biologics, biosimilars, and novel therapies can all add to the testing requirements to a significant level. Furthermore, the growing trend towards patient safety and quality is expected to strengthen the dominance of North America.
U.S. Pyrogen Testing Market Size and Growth 2026 to 2035
The U.S. pyrogen testing market size is calculated at USD 497.94 million in 2025 and is expected to reach nearly USD 1,325.03 million in 2035, accelerating at a strong CAGR of 10.28% between 2026 and 2035.
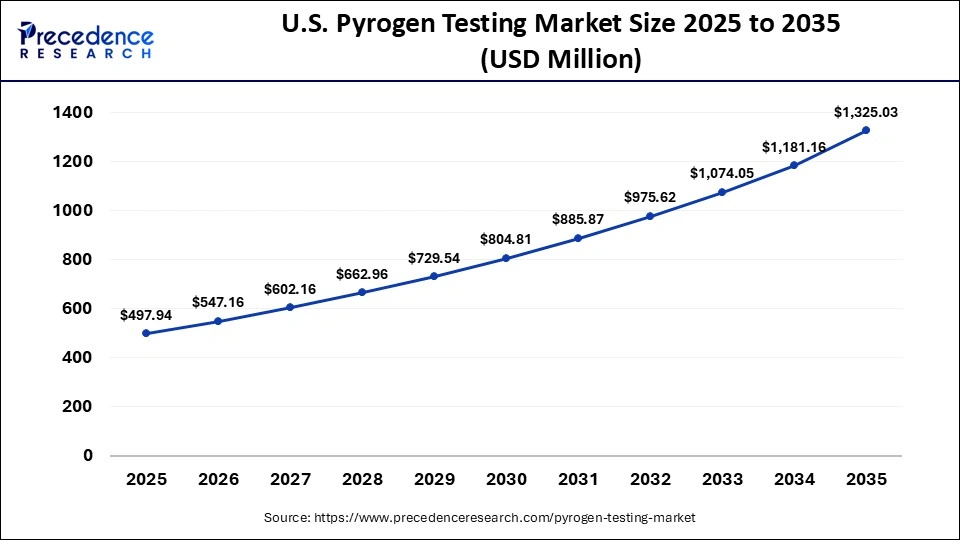
Country-Level Analysis
The U.S. has the highest percentage at the country level, due to a strong presence of pharmaceutical companies and a high level of regulatory control by the relevant agencies, like the FDA. The U.S. is a global hub for biologics development, and this creates a demand for improved methods of pyrogen testing. Canada is also making its share, and this has been made possible by increasing the biotech industry and a friendly government service that encourages research on life sciences .
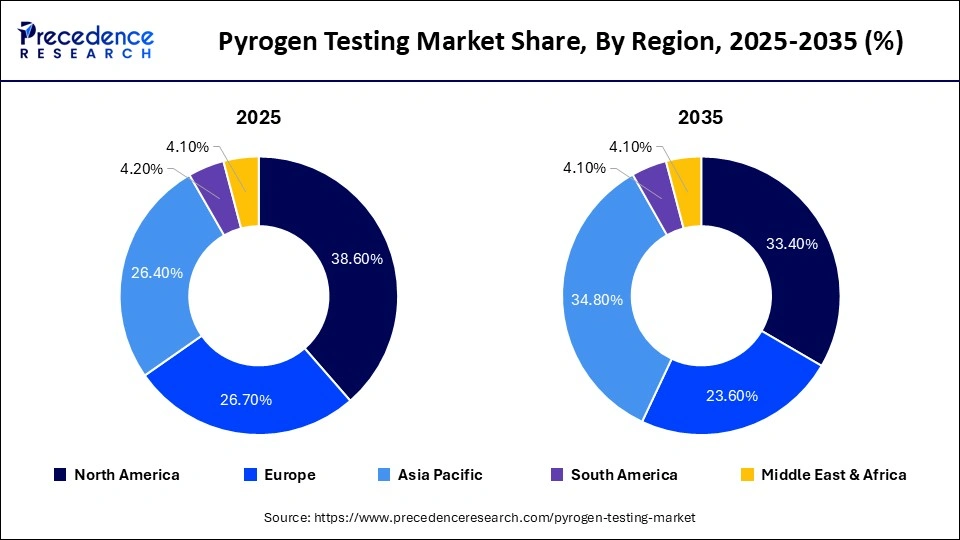
Why is Europe the Fastest-Growing Region in the Pyrogen Testing Market?
Europe is expected to experience the fastest growth during the predicted timeframe, due to growing regulatory convergence and the great focus on ethical testing practices. The area has been on the frontline in encouraging the use of alternatives to animal testing, which has enhanced the implementation of other methods like MAT and recombinant assays. An increase in the production of biologics and the production of vaccines is also playing a major role in the rise in demand for pyrogen testing. More so, the market is growing by boosting investments in the pharmaceutical research and development, and in improving clinical trial activities.
Country-Level Analysis
Germany dominates the European market at the country level because of its good base of pharmaceutical manufacturing and orientation on high-quality standards. One of the main players is the United Kingdom, which is helped by the significant clinical research and the growing use of advanced testing technologies. France and Switzerland also largely have great roles, due to the location of big pharmaceutical companies and innovation centers.
Pyrogen Testing Market Value Chain Analysis

Pyrogen Testing Market Companies
- Charles River Laboratories
- Lonza Group
- Thermo Fisher Scientific
- Merck KGaA
- BioMérieux
- Associates of Cape Cod
- Wako Chemicals USA
- GenScript
- Creative Bioarray
- Hyglos GmbH
- Microcoat Biotechnologie
- Ellab A/S
- Seikagaku Corporation
- Sanquin Reagents
- Fujifilm Wako Chemicals
Recent Developments
- In February 2026, the EDQM and EPAA hosted the Pyrogen Testing 2.0 symposium, focusing on ethical, rapid, and sustainable non-animal pyrogen testing methods. The event was hosted to promote state-of-the-art approaches for safe pyrogen detection globally, reflecting evolving practices in the industry.(Source: https://www.edqm.eu ).
- In January 2026, Betmat launched high-performance, sustainable endotoxin testing services to address the evolving needs of the industry. This initiative focuses on delivering reliable and efficient testing solutions while emphasizing environmental sustainability, positioning Betmat as a leader in quality assurance for pharmaceuticals and biologics.
Segment Covered in the Report
By Test Type
- Limulus Amebocyte Lysate testing
- Monocyte Activation Test
- Rabbit Pyrogen Test
By Offering Type
- Reagents and Kits
- Instruments and Readers
- Consumables
- Services
By Application
- Pharmaceutical Products Testing
- Biologics and Biotechnology Products Testing
- Medical Devices Testing
- Cell and Gene Therapy Products Testing
- Vaccine Testing
By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Contract Research Organizations
- Academic and Research Institutes
- Medical Device Manufacturers
By Technology
- Gel Clot LAL Method
- Turbidimetric LAL Method
- Chromogenic LAL Method
- Recombinant Factor C Assay
By Sample Type
- Injectable Drugs
- Implantable Medical Devices
- Parenteral Nutrition Products
- Dialysis Products
- Vaccines
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
