What is the Pharmaceutical Analytical Testing Market Size?
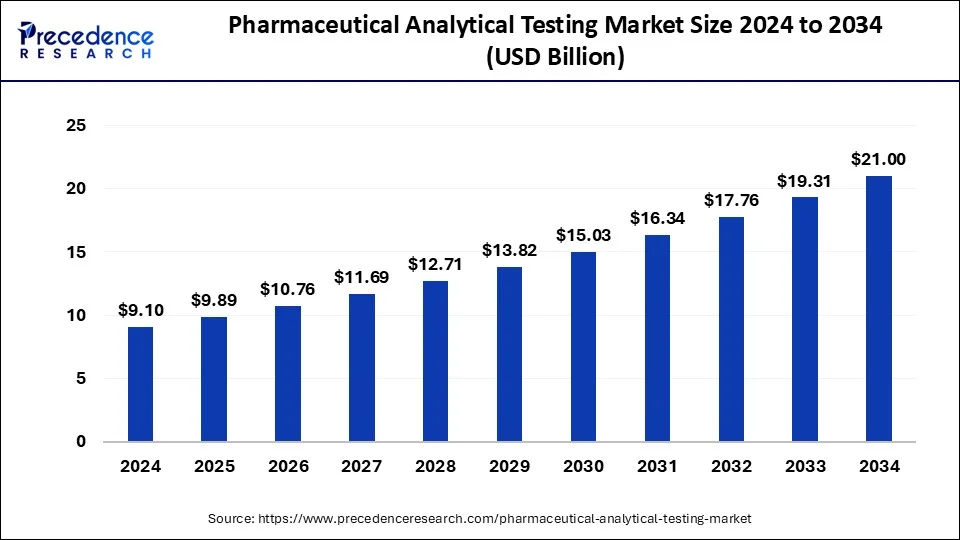
The global pharmaceutical analytical testing market size was estimated at USD 9.89 billion in 2025 and is predicted to increase from USD 10.76 billion in 2026 to approximately USD 22.60 billion by 2035, expanding at a CAGR of 8.62% from 2026 to 2035. The pharmaceutical analytical testing market is driven by the major factors such as rising investments in the pharmaceutical industry and booming government support for the same. Alongside, major players are involved in creating sophisticated and advanced testing materials, these factors along with technological innovations will support the market's growth.
Pharmaceutical Analytical Testing Market Key Takeaways
- The global pharmaceutical analytical testing market was valued at USD 9.89billion in 2025.
- It is projected to reach USD 22.60 billion by 2035.
- The market is expected to grow at a CAGR of 8.62% from 2026 to 2035.
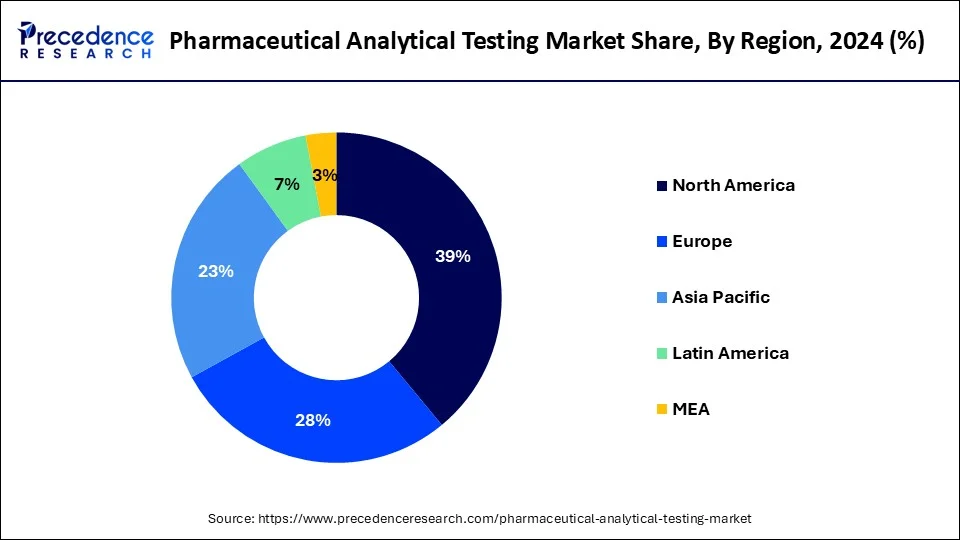
- North America dominated in the pharmaceutical analytical testing market with the largest market share of 39% in 2025.
- Asia Pacific is observed to be the fastest growing in the pharmaceutical analytical testing market during the forecast period.
- By service type, the bioanalytical testing segment contributed more than 41% of market share in 2025.
- By service type, the method development and validation segment show a significant growth in the pharmaceutical analytical testing market during the forecast period.
- By service type, the stability testing segment shows a notable growth in the pharmaceutical analytical testing market during the forecast period.
- By end-user, the biopharmaceutical companies segment recorded the highest market share of 43% in 2025.
Market Overview
In the pharmaceutical industry, ensuring that medications are safe and effective for their intended purpose is the primary goal of analytical testing. Extensive testing aids in locating possible pollutants, impurities, or variances that can jeopardize the safety of pharmaceuticals. Pharmaceutical product quality must be maintained through analytical testing. It aids in ensuring that every batch satisfies the requirements by helping to check the consistency and purity of the products. Advanced analytical testing techniques are crucial to validate novel approaches and assure the viability of newly created medications and therapies, especially in biologics and personalized medicine .
- In June 2024, according to a new analysis from Milliman that PhRMA commissioned, the Inflation Reduction Act (IRA)'s drug pricing provisions may cause 3.5 million Medicare Part D beneficiaries to have higher out-of-pocket costs in 2026, undermining the advantages of the new out-of-pocket limit for millions of seniors and people with disabilities.
How AI Will Help Pharmaceutical Testing Industry?
Testing is expedited by AI, which analyzes hundreds of data points more quickly than the human eye can. As many data analysis jobs in laboratory testing involve repetition and take time, assigning simpler tasks to artificial intelligence (AI) expedites and simplifies processing. This enables lab personnel to concentrate on more delicate testing issues, like assessing anomalous or critical samples of great concern to clients. As a result, they can provide more insightful analysis and recommendations.
Furthermore, AI can learn as it goes along, which is vital for testing services because it allows it to recognize sample patterns more rapidly and accurately over time. Machine learning maintains its interpretive flexibility since it is not rule-based but instead considers correlations and permutations.
Pharmaceutical Analytical Testing Market Growth Factors
- Quality assurance and compliance testing are in high demand due to stricter requirements from organizations such as the FDA, EMA, and others.
- Specific analytical methods are needed to propel market expansion with the growing attention to biologics and biosimilars. This drives the growth of the pharmaceutical analytical testing market.
- Efficiency and accuracy are increasing due to advancements in analytical techniques like mass spectrometry, chromatography, and spectroscopy.
- The demand for analytical testing is driven by the high focus on ensuring pharmaceutical goods' safety and efficacy.
- Advanced analytical techniques are necessary to develop more complicated medications, such as combination products and targeted therapy.
Market Scope
| Report Coverage | Details |
| Market Size by 2035 | USD 22.60 Billion |
| Market Size in 2025 | USD 9.89 Billion |
| Market Size in 2026 | USD 10.76 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 8.62% |
| Largest Market | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Service Type, End-user, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Driver
Growing prevalence of chronic diseases
Adequate medication is in high demand because of the rise of chronic diseases such as diabetes, cancer, cardiovascular disease, and respiratory problems worldwide. The pharmaceutical sector is under pressure to create novel medications and treatments to deal with these illnesses. Medication for chronic diseases is usually needed for the rest of one's life. This calls for ongoing pharmacovigilance programs to monitor drug efficacy and safety. Analytical testing is essential to identify any possible side effects and guarantee the long-term safety of these drugs.
The Drug Supply Chain Security Act (DSCSA) was not followed by Connecticut-based contract testing lab Valisure, according to a recent untitled letter from the US Food and Drug Administration (FDA). The letter also revealed that the company's analytical testing laboratory had "methodological deficiencies."
Restraints
Limited availability of skilled professionals
The need for analytical testing services rises with the expansion of the pharmaceutical industry. However, the scarcity of qualified personnel can lead to bottlenecks, making it difficult for labs to expand to accommodate growing demand. In some areas, a large percentage of the highly qualified staff in pharmaceutical analytical testing is getting close to retirement. If there isn't enough fresh talent entering the market, the skilled professional deficit will probably get worse over time, raising questions about the industry's long-term viability.
Limited access to advanced testing facilities
This equipment demands continuous expenditure for calibration and maintenance after initial setup. High-tech lab running expenses, such as utilities, consumables, and recurring improvements, can be unaffordable. Analytical testing for pharmaceuticals is strictly regulated. Facilities must be accredited by regulatory agencies such as the FDA or EMA and adhere to Good Laboratory Practices (GLP). Companies may find it challenging to achieve these strict requirements if they have limited access to cutting-edge testing facilities, which could cause delays in the licensing of new drugs. This limits the growth of the pharmaceutical analytical testing market.
Opportunity
Growth in generic drug manufacturing
In contrast to small-molecule generics, biosimilars , generic equivalents of biologic drugs, require more intricate and thorough analytical testing. Thus, the expansion in biosimilar manufacturing has increased the demand for specialized analytical testing services.
Analytical testing organizations invest in cutting-edge testing apparatus and methodologies to remain competitive. The growing complexity of generic medications, particularly considering the emergence of biosimilars, calls for the creation of novel testing procedures. This ongoing innovation gives analytical testing companies a competitive edge and creates new growth opportunities. This opens an opportunity for the growth of the pharmaceutical analytical testing market.
Segment Insights
Service Type Insights
The bioanalytical testing segment dominated the pharmaceutical analytical testing market in 2025. Biologics, which includes gene therapies, vaccines, and monoclonal antibodies, has experienced rapid growth. Bioanalytical testing has become crucial since these complex compounds need highly sophisticated analytical techniques to guarantee their safety, efficacy, and quality. More robust and standardized bioanalytical testing procedures are required when clinical trials are carried out worldwide to ensure uniform and trustworthy results in all areas.
- In August 2024, in North America, SGS plans to launch new, specialized bioanalytical testing services. The new services are offered by a Hudson, New Hampshire lab and can be applied in the pharmaceutical and biopharmaceutical industries. In addition to technique transfer, development validation, PD bioanalysis, immunogenicity testing, PK bioanalysis, ELISA and multiplex assays, bioassays, and more, the new services will support discovery, preclinical, and phase 1-3 clinical trials.
The method development and validation segment show a significant growth in the pharmaceutical analytical testing market during the forecast period. The Quality by Design (QbD) approach highlights the importance of thorough method development in pharmaceutical production. To guarantee consistent product quality throughout the lifespan, well-defined and validated analytical procedures are necessary for QbD, which focuses on integrating quality into the product from the outset. The need for method development and validation services is anticipated to increase as QbD concepts are applied more frequently in drug development and production.
The stability testing segment shows a notable growth in the pharmaceutical analytical testing market during the forecast period. Stability testing has gotten more challenging with the introduction of increasingly complex medication formulations, such as biologics, biosimilars, and combination medicines. Specific stability studies are needed to assess the chemical, physical, and microbiological stability of these sophisticated formulations under varied circumstances. The demand for complicated stability testing services has grown due to this complexity, which has fueled the market expansion.
End-user Insights
The biopharmaceutical companies segment dominated the pharmaceutical analytical testing market in 2025. Massive, complicated molecules like biopharmaceuticals often need thorough characterization, which includes stability tests, potency testing, and structural analysis. Due to their intricacy, high-performance liquid chromatography (HPLC), nuclear magnetic resonance (NMR), and mass spectrometry are among the sophisticated analytical methods that must be used. Many biopharmaceutical businesses would rather hire specialized service providers to handle their analytical testing needs. By outsourcing, companies can cut down on the overhead of running internal testing facilities while concentrating on their core skills.
- In March 2023, MS Pharma, a business specializing in chemical and microbiological quality control studies, was purchased by Neopharm Labs Inc., a full-service analytical laboratory catering to the pharmaceutical industry.
Regional Insights
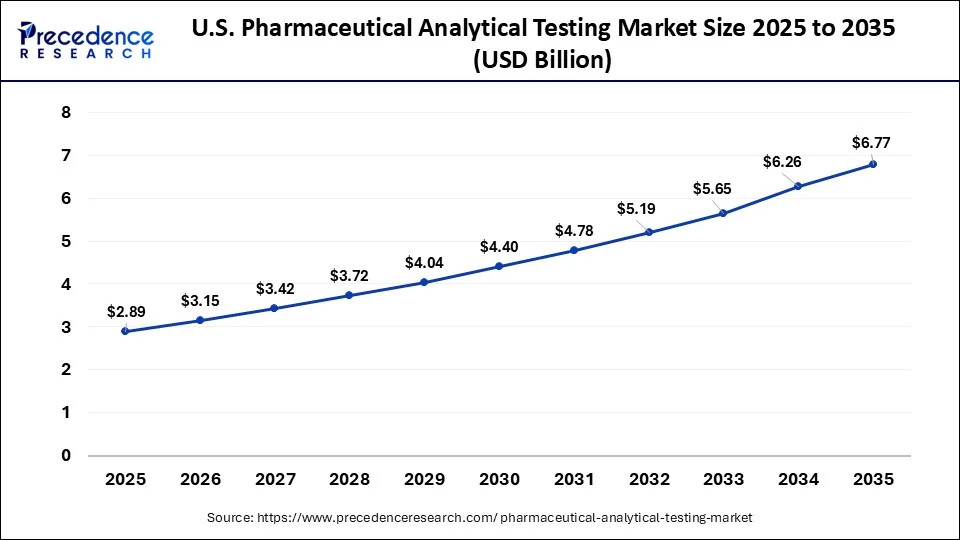
What is the U.S. Pharmaceutical Analytical Testing Market Size?
The U.S. pharmaceutical analytical testing market size was valued at USD 2.89 billion in 2025 and is projected to surpass USD 6.77 billion by 2035 with a CAGR of 8.89% from 2026 to 2035.
North America dominated in the pharmaceutical analytical testing market in 2025. North America, particularly the United States, has some of the world's strictest and most well-defined regulatory environments. Health Canada and the U.S. Food and medication Administration (FDA) set high criteria for medication development, production, and testing. These organizations demand thorough analytical testing at every phase of the pharmaceutical development process, from preclinical research to post-market surveillance. The strict guidelines established by these regulatory agencies fuel the need for superior analytical testing services, hence reinforcing North America's dominant position in this industry.
- In 2022, 52.3% of global pharmaceutical sales were in North America, compared to 22.4% in Europe. As per IQVIA, the US market accounted for 64.4% of the sales of newly introduced pharmaceuticals between 2017 and 2022, while the European market accounted for 16.4% of the total.
U.S. Pharmaceutical Analytical Testing Market Trends
The U.S. market is growing as companies increase R&D and quality control to meet regulatory standards. Firms are outsourcing to accelerate time-to-market and even manage complex drug development. Moreover, the implementation of advanced techniques such as mass spectrometry and next-generation sequencing, along with AI-driven data analytics, is increasing to enhance precision and speed in drug characterization.
Asia-Pacific is observed to be the fastest growing in the pharmaceutical analytical testing market during the forecast period. Global pharmaceutical regulations, such as Good Manufacturing Practices (GMP) and Good Laboratory Practices (GLP), are being adopted throughout the Asia-Pacific region at an increasing rate. The need for these services is driven by thorough analytical testing that conforms with international regulations. The market for analytical testing has also been helped by tightening pharmaceutical quality rules in some countries, which call for more thorough testing and compliance.
- In June 2024, IISc researchers have created a new technique for producing recombinant proteins in large quantities. Modified bacterial, viral, or mammalian cells are grown in considerable bioreactors to develop recombinant proteins in enormous quantities. Examples of these proteins include insulin, monoclonal antibodies, and vaccine antigens.
Bioanalytical and Stability Testing Market in Europe
Europe shows a significant growth during the forecast period. It is driven by the growing incidence of chronic diseases such as cancer and autoimmune diseases, which have fueled the need for complex biologics and, thus, consequently, biosimilar testing to lower costs. Heavy investment in R&D, mainly in Germany, the UK, and France, drives the requirement for rigorous bioanalytical and stability testing to determine shelf-life and safety.
Impact of Serialization and Quality Assurance Standards in Latin America
Latin America shows a notable growth during the forecast period. It is driven by the fact that pharmaceutical forgers are prevalent, and even serialized, tamper-evident packaging offers essential security to verify product authenticity from producer to patient. Implementation requires significant capital for technology, software, and infrastructure, causing challenges, mainly for smaller local manufacturers. The push forces firms to upgrade, driving the adoption of AI and even advanced data processing for better traceability.
Adoption of Automation and Digital Laboratory Solutions in MEA
MEA shows a rapid growth during the forecast period. It is driven by automated systems, like Total Laboratory Automation (TLA), which significantly decrease turnaround times, enabling laboratories to manage increasing workloads without adding staff. Automated systems decrease manual intervention, which in turn decreases errors in pre-analytical, analytical, and post-analytical phases, ensuring more reliable and standardized diagnostic results.
Pharmaceutical Analytical Testing Market Companies
- PPD Inc: PPD provides comprehensive pharmaceutical analytical testing services via its GMP and bioanalytical labs, validation, stability studies, covering method development, and quality control for small molecules, biologics, and vaccines.
- Boston Analytical: Boston Analytical provides comprehensive cGMP-compliant, FDA/DEA registered, and even ISO/IEC-17025:2017 certified analytical testing services to the pharmaceutical, biopharmaceutical, and medical device industries.
- Steris: STERIS offers a comprehensive suite of analytical testing, laboratory services, and validation solutions programmed for the pharmaceutical, biopharmaceutical, and medical device markets. Their offerings aim on quality control, regulatory compliance, and even food safety and contamination control.
Other Major Key Players
- SGS SA
- LabCorp
- Eurofins Scientific
- Laboratory Testing Inc
- Bee Pharmo Labs Pvt Ltd
- Apple Pharmaceutical
- Doctors Analytical Laboratories Pvt Ltd.
Recent Developments
- In February 2024, at its good manufacturing practices (GMP) facility in Middleton, Wisconsin, Thermo Fisher Scientific Inc.'s PPD clinical research business, the global leader in supporting science, is expanding its portfolio of services to include mycoplasma and further biosafety testing capabilities. This new service helps clients deliver safe medications to patients by guaranteeing biopharmaceutical goods are free of contaminants. The analytical testing service of the clinical research business provides it.
- In December 2023, based on its SUREtechnology Platform, KBI Biopharma, Inc., a JSR Life Sciences subsidiary, introduced SUREmAb, a product for optimal, safe, and economical monoclonal antibody (mAb) development and manufacture.
Segments Covered in the Report
By Service Type
- Bioanalytical Testing
- Method Development and Validation
- Stability Testing
- Drug Substances Testing
- Others
By End-user
- Biopharmaceutical Companies
- Contract Research Organizations
- Medical Device Companies
- Others
By Geography
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
