What is Superdisintegrants Market Size?
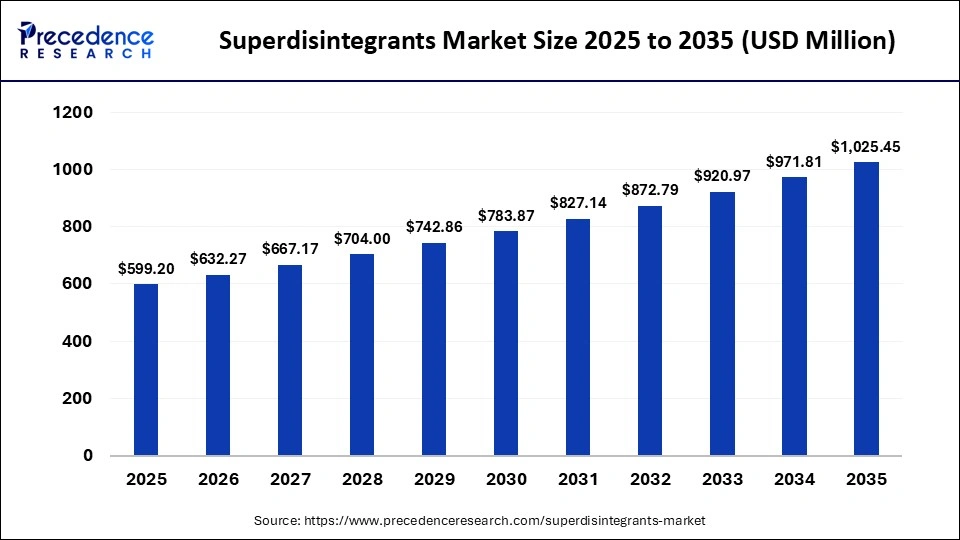
The global superdisintegrants market size was calculated at USD 599.20 million in 2025 and is predicted to increase from USD 632.27 million in 2026 to approximately USD 1,025.45 million by 2035, expanding at a CAGR of 5.52% from 2026 to 2035. The market growth is attributed to the rising demand for rapid disintegrating oral dosage forms and the increasing adoption of advanced excipient technologies.
Key Takeaways
- North America dominated the global market in 2025.
- Asia-Pacific is expected to grow at the fastest CAGR between 2026 and 2035.
- By product, the crospovidone segment contributed the highest market share in 2025.
- By product, the ion exchange resin segment is expected to grow at a strong CAGR between 2026 and 2035.
- By form, the tablets and capsules segment held a major market share in 2025.
- By form, the mouth-dissolving films (MDFs) segment is expected to expand at a rapid CAGR from 2026 to 2035.
Which Factors Drive the Superdisintegrants Industry?
The superdisintegrants market is being driven by the rapid global increase in demand for oral solid dosage forms. Tablets and capsules are still the most used drug forms, with 82.5% of immediate-release oral products being solid dosage forms, 64.5% in the form of tablets, and 22.7% in the form of capsules, according to the 2023 NIH report. Superdisintegrants are substances added to tablets and capsules to allow them to disintegrate rapidly in the gastrointestinal fluids. This enhances the dissolution and bioavailability of drugs. Furthermore, the oral medication demand remains a key market growth for the superdisintegrants sector.
Impact of Artificial Intelligence on the Superdisintegrants Market
Artificial Intelligence is reshaping how pharmaceutical formulation scientists design and optimize excipient systems. This enables rapid prediction of ideal excipient combinations and reduces trial-and-error experimentation. Machine learning and neural networks are types of AI models that predict important parameters of the formulation, including solubility, dissolution behavior, and excipient-API compatibility, accelerating dosage form design with fewer laboratory experiments than traditional dosage form design methods. Moreover, researchers are now adding AI to high-throughput experimentation systems to enable automated labs to screen hundreds of formulation permutations per day and update models with real-time feedback.
Superdisintegrants Market Trends
- Personalized Medicine Formulation Customization
Pharmaceutical companies are shifting towards customized oral solid dosage forms to meet specific subgroups of patients, including pediatrics and geriatrics. This fuels demand for superdisintegrants with tunable performance profiles enabling faster disintegration or sustained action as needed. The release profile is now differentiated in clinical pipelines by personalization. - Rise of Biodegradable and Eco Certified Excipients
Sustainability certifications and biodegradability labeling are becoming influential in procurement decisions for pharmaceutical excipients. There is increased acceptance of disintegrants that are environmentally friendly among formulation teams globally. The trend is in line with the larger-scale pharmaceutical commitment to green chemistry and circular supply chains in the coming years. - Localized Manufacturing and Regional Specification Alignment
The manufacture of pharmaceuticals is increasingly regionalized in order to achieve the country-specific regulatory demands and minimize supply chain risk. Local manufacturing centers are now defining local geographic preferred excipient grades that are consistent with local quality and disintegration demands. These factors drive diversified superdisintegrant portfolios tailored by geography and formulation need.
Superdisintegrants Market Growth Factors
- Rising Demand for Fast-Dissolving Tablets: Growing preference for patient-friendly dosage forms is propelling the adoption of efficient superdisintegrants.
- Expansion of Generic Drug Manufacturing: Boosting generic drug production worldwide fuels higher consumption of high-performance superdisintegrants.
- Advancements in Natural Excipient Alternatives: Growing focus on natural and biodegradable disintegrants is propelling sustainable formulation practices.
- Collaborative Industry Partnerships and Licensing: Strategic collaborations between excipient manufacturers and pharmaceutical companies are propelling innovation and faster market access.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 599.20 Million |
| Market Size in 2026 | USD 632.27 Million |
| Market Size by 2035 | USD 1,025.45 Million |
| Market Growth Rate from 2026 to 2035 | CAGR of 5.52% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Form, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Key Emerging Regulations for the Superdisintegrants Market
|
Regulation Areas |
Key Emerging Regulations |
Focus Areas |
|
Sustainability |
Mandates for eco-friendly, biodegradable, and non-toxic materials, restricting synthetic alternatives. |
Green excipients and biodegradable sourcing. |
|
Quality Control |
Strict quality disclosure for excipients on labels and enhanced EU-GMP certification for global exporters. |
Safety, transparency, and labeling. |
|
Process Control |
Prior Intimation systems required for changes in excipients or manufacturing processes post-approval. |
Supply chain stability & drug efficacy. |
Strategic Expansion, Innovation, and Global Supply Strengthening the Superdisintegrants Market
- In 2025, BASF SE retained its position as one of the largest exporters of crospovidone and croscarmellose sodium globally, leveraging its established polymer chemistry infrastructure across Europe, Asia, and North America to support formulation partners in over 50 countries.
JRS Pharma entered the Indian manufacturing landscape in July 2024 by signing an agreement to build a dedicated cotton-based croscarmellose sodium production facility in Mehsana, aimed at reducing imports and supporting local pharmaceutical OEMs with domestically produced superdisintegrants. - During 2025, Roquette showcased expanded formulation capabilities at AAPS 2025 PharmSci 360, highlighting advanced solutions relevant to superdisintegrants and other excipient innovations, underlining its role as a major global player in pharma excipient technology.
- In 2024–2025, pharma excipient R&D and production facility enhancements were reported at major global events such as CPhI 2025 Frankfurt, where leading producers demonstrated expanded portfolios and technical expertise, indicating ongoing investment in formulation and production capabilities.
- In April 2025 IFF Pharma Solutions showcased next-generation excipient innovations, including technology critical to orally disintegrating tablets that rely on superdisintegrants, at CPHI Japan 2025, reflecting ongoing investment in formulation and production capabilities
- In 2024, the European Union's exports of medicinal and pharmaceutical products grew by 13.5% on year, reaching a total value of €313.4 billion, a record high, underscoring Europe's role as a major production and export source for pharmaceuticals and related formulation ingredients.
Segmental Insights
Product Insights
Why Did the Crospovidone Segment Dominate the Superdisintegrants Market?
The crospovidone segment held the largest revenue share of the market in 2025, due to its ability to provide rapid water wicking and mechanical tablet disintegration. This improves dissolution without the formation of a viscous gel layer, which is an important consideration in immediate-release products. Regulatory frameworks of the FDA SUPAC IR guidance focus on the definition of excipient roles in the change of formulation and promotion of the use of the same performers, like crospovidone.
Crospovidone also has a predictable performance in direct compression, as well as wet and dry granulation methods, enabling pharmaceutical innovators fewer reformulations and quicker scale-up. Furthermore, the crosprovidones' consistent action with poorly water-soluble APIs further boosts segment growth.
The ion exchange resin segment is expected to grow at the fastest CAGR in the coming years, owing to its multifunctional role in tablet formulations, including use as disintegrants, taste masking agents, and controlled release facilitators in oral dosage forms. These synthetic polymers are claimed to have high ionic exchange and structural stability.
They are being used to enhance drug liberation and tablet breakup in orodispersible and other oral dosage forms. Furthermore, researchers have also noted that ion exchange resins can convert drugs into more soluble forms via drug resin complexes, which enhances dissolution profiles in certain oral formulations, further supporting their growth potential.
Form Insights
How the Tablets and Capsules Segment Dominated the Superdisintegrants Market?
The tablets and capsules segment accounted for the highest revenue share of the market in 2025, due to their established role in immediate-release and controlled-release oral therapy regimens widely prescribed across global healthcare systems.
These dosage forms frequently incorporate superdisintegrants to ensure consistent disintegration and rapid drug availability in conventional and generics portfolios. Moreover, the use of tablets and capsules orally in chronic and acute therapeutic settings is expected to facilitate the adoption of tested and proven dosages of tablets and capsules in the coming years.
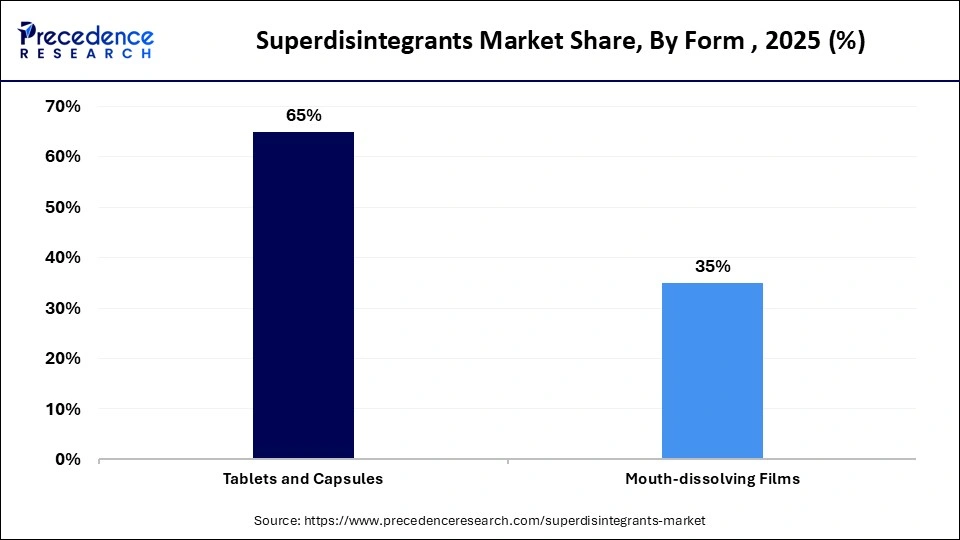
The mouth-dissolving films (MDFs) segment is expected to grow with the highest CAGR in the market during the studied years, owing to the increasing patient preference for convenient and swallow-friendly dosage forms, particularly among elderly and pediatric populations with swallowing impairments. The development of hot melt extrusion and solvent casting processes helps produce MDFs with high efficiency in terms of uniform thickness and the ability to disperse.
New combinations of excipients enabling flexible loading of active moieties into film matrices were the focus of industry conferences and symposia. Furthermore, the growing demand for patient-centric therapies and expanded R&D focus on sensory acceptable, quick action formats, potentiating the demand for MDFs.
Regional Insights
Why North America Dominated the Superdisintegrants Market?
North America led the market in 2025, as it has the most developed pharma manufacturing systems and a long-term focus on the development of high-quality solid oral dosage, owing to the elaborate quality standards enforced by the U.S. FDA and pharmacy standards in accordance with the USP quality standards. The formulators focus on consistent excipient performance of tablets and capsules.
The FDA launched initiatives in 2025 to boost domestic drug production, including a pilot program and the FDA PreCheck scheme to expedite review and construction of manufacturing facilities, strengthening local production capabilities and encouraging investments in formulation science. Moreover, the leadership in regulatory compliance and advanced formulation practices continues to secure North America's position as a core regional base for excipient utilization.
U.S. – Leading North America's Pharmaceutical Strength
The U.S. leads the market in North America due to its advanced regulatory ecosystem and significant drug production scope. In 2026, regional programs such as the FDA PreCheck and the EO 14293 on onshoring pharmaceuticals support the growth of local production and supply chain resiliency. This makes it easier to integrate technologies in excipient tablet production. Furthermore, continued investments in research and development infrastructure and formulation science have ensured that U.S. drug manufacturers remain in the lead to create complex oral solid dosage forms.
Why is Asia-Pacific Projected to Grow Fastest in the Superdisintegrants Market?
Asia-Pacific is expected to witness the fastest growth during the predicted timeframe, owing to the expanding pharmaceutical production capacity, regulatory improvements, and increasing integration into global supply chains across China, India, South Korea, and Southeast Asia. In 2025, the National Medical Products Administration (NMPA) of China introduced specific GMP annexes of pharmaceutical excipients and packaging materials.
This improves quality control and aligns the local needs with the global practice, which is expected to further raise the level of excipient manufacturing. Oxford Economics projects continued percentage gains in overall production, which supports broader formulation and excipient demand. Additionally, government incentives and policies that support pharmaceutical exports and quality improvements are likely to fuel the market in this region.
India – Driving Growth in Asia Pacific's Pharmaceutical Landscape
China is leading the charge in the Asia-Pacific market, supported by expansive generic drug manufacturing and deep global integration. Pharma exports in India increased to USD 30.5 billion in FY25 as a result of the increasing international demand. This further supports other related excipient segments applied in solid formulations. Furthermore, India's strategic position in the global pharmaceutical value chain is projected to sustain strong momentum for oral dosage technologies and underpin regional growth.
How will Europe be considered a Notable Region in the Superdisintegrants Market?
Europe is a notably growing region in the global market. This growth is driven by high demand for pharmaceutical innovation, an aging population, and a strong manufacturing base. There is a strong preference for convenient, fast-dissolving tablets and oral dosage forms, which require high-performance superdisintegrants like cross-linked sodium carboxymethyl cellulose and crospovidone. The growing geriatric population and the increasing prevalence of chronic diseases drive the need for easily swallowable, fast-acting medications.
Germany Market Trends
Germany is pioneering sustainable, high-quality superdisintegrants, driven by major eco-friendly initiatives from leaders like Evonik and JRS Pharma, as Europe's largest pharmaceutical market and R&D leader. Amid 2026 cost-control regulations, the sector is accelerating innovation in advanced, eco-friendly excipients through strategic partnerships like the new Pharma and MedTech Dialogue.
How is Latin America Growing in the Superdisintegrants Market?
Latin America is a significantly growing region in the market. This growth is driven by expanding pharmaceutical manufacturing, particularly in Brazil and Mexico, and increasing consumer demand for faster-acting medication. Local manufacturing capacities are increasing to reduce reliance on imports, particularly for generic and biosimilar drugs. Brazil is a major hub, with other markets like Argentina and Colombia also expanding their footprints. As patents for brand-name drugs expire, the market for generic alternatives is surging, essential for these fast-acting, cost-effective formulations.
Brazil Market Trends
Brazil is transforming into a high-growth hub by aggressively localizing API and excipient production as Latin America's largest pharmaceutical market. With regulatory, fast-track approvals for new formulations aligning with Mexico by 2026 and major strides in precision oncology via partnerships like CellAction, Brazil is rapidly accelerating its shift toward self-sufficient, high-tech manufacturing.
How will the Middle East and Africa Emerge in the Superdisintegrants Market?
The Middle East and Africa are emerging as a significant growth region in the global market. This growth is driven by rapid pharmaceutical industrialization, high import dependency, and a surge in chronic diseases requiring advanced, fast-acting oral dosages. High rates of diabetes, cancer, and cardiovascular diseases in emerging MEA economies demand better drug delivery systems and superior disintegration for rapid bioavailability. The region is pivoting from importing to local manufacturing to reduce costs, leading to increased demand for cost-effective, high-performance excipients.
The UAE Market Trends
UAE is rapidly becoming a leading pharmaceutical hub, moving from importer to high-tech manufacturer by launching new products through Globalpharma and implementing a secure, blockchain-verified supply chain. Backed by the EDE reform enabling competitive, multi-distributor access and accelerated production of insulin analogs, the UAE is cementing its role as an innovative, sustainable, and tech-driven regional hub.
Superdisintegrants Market Value Chain Analysis
- Raw Material Sourcing
Procurement of cellulose, starches, and polymer precursors forms the foundation of superdisintegrant production.
Key Players: Wood pulp suppliers, cellulose producers, starch manufacturers - Chemical Synthesis & Intermediate Production
Raw materials are chemically modified to produce intermediates with excipient functionality.
Key Players: Polymer chemical manufacturers, excipient intermediates processors - Superdisintegrant Manufacturing
Intermediates are processed into finished superdisintegrants with controlled particle size and purity.
Key Players: BASF SE, Ashland Global Holdings Inc., DuPont de Nemours Inc., DFE Pharma GmbH & Co. KG - Distribution & Supply Chain Logistics
Finished excipients are distributed globally to pharmaceutical manufacturers under GMP/GDP standards.
Key Players: Global excipient distributors, specialty chemical logistics firms - Pharmaceutical Formulation & End Use
Pharmaceutical companies incorporate superdisintegrants into tablets and capsules to enhance disintegration.
Key Players: Global pharmaceutical manufacturers, CROs/CMOs
Superdisintegrants Market Companies
- Asahi Kasei Corporation
- Ashland
- BASF SE
- Corel Pharma Chem.
- DFE Pharma
- DuPont.
- JRS PHARMA
- Merck KGaA
- NIPPON SODA CO., LTD.
- Roquette Frères.
Recent Developments in the Superdisintegrants Market
- In February 2026, DFE Pharma introduced its Continuous Manufacturing (CM) platform to support pharmaceutical companies in formulation development, optimization, and lifecycle management of CM processes. Developed in collaboration with Gericke, the platform combines CM-ready excipients, an evaluation environment, and multidisciplinary expertise, enabling data-driven decisions from early development through long-term CM operations.(Source: https://www.expresspharma.in)
- In February 2026, Lupin Limited signed a licensing and supply agreement with Spektus Pharma to commercialize the novel antidepressant DeslaFlex in Canada. Utilizing Spektus's proprietary Flexitab oral drug-delivery platform, the partnership combines Lupin's strong Canadian commercial footprint with Spektus's expertise in differentiated formulations.(Source: https://www.lupin.com)
- In October 2025, Asahi Kasei announced the launch of two new specialty grades in its Sonanos excipient portfolio. These next-generation excipients, designed to improve injectable drug formulations, are manufactured under GMP standards, with commercial availability expected in 2027. (Source: https://www.contractpharma.com)
- In September 2025, Eli Lilly and Company announced plans to construct a USD 6.5 billion next-generation manufacturing facility at Generation Park in Houston, Texas. This facility, the second of four new U.S. sites planned for 2025, will focus on producing small-molecule APIs across therapeutic areas such as cardiometabolic health, oncology, immunology, and neuroscience. (Source: https://www.prnewswire.com)
Segments Covered in the Report
By Product
- Croscarmellose sodium
- Crospovidone
- Ion Exchange Resin
- Sodium Starch Glycolate
- Others
By Form
- Tablets and Capsules
- Mouth-dissolving Films
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
