What is the Terminal Sterilization Market Size in 2026?
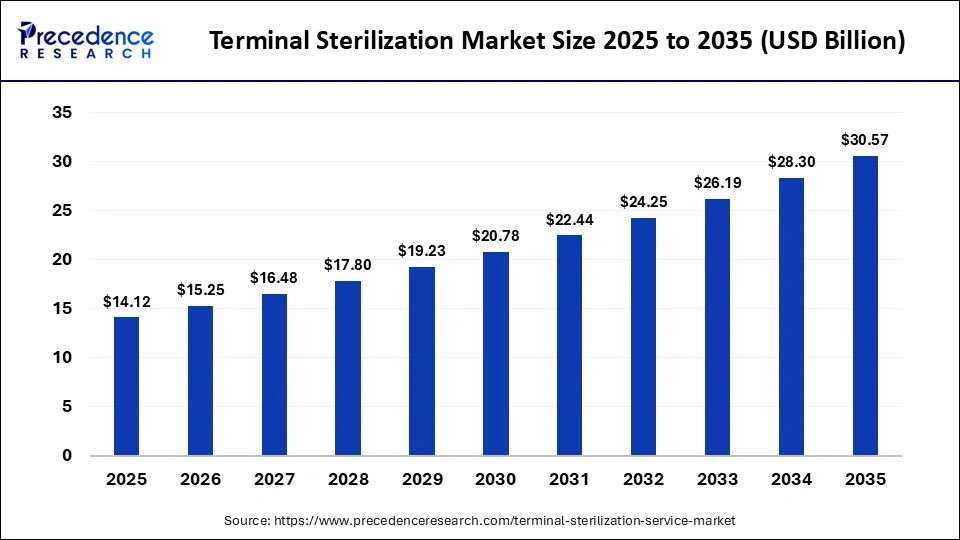
The global terminal sterilization market size was calculated at USD 14.12 billion in 2025 and is predicted to increase from USD 15.25 billion in 2026 to approximately USD 30.57 billion by 2035, expanding at a CAGR of 8.03% from 2026 to 2035.This market is growing due to the rising demand for safe and contamination-free medical products.
Key Takeaways
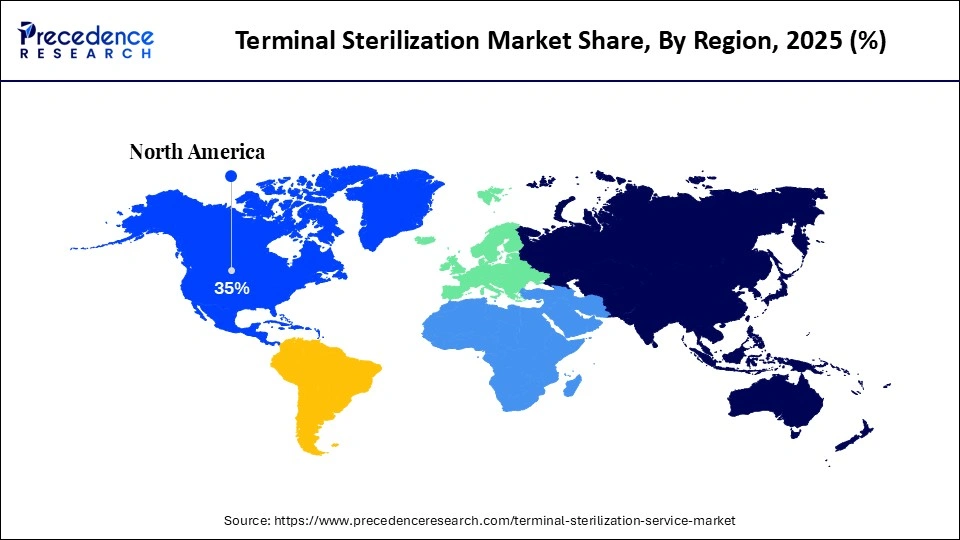
- North America dominated the global market with the largest market share of 35% in 2025.
- The Asia Pacific is expected to grow at the fastest CAGR between 2026 and 2035.
- By product, the equipment segment held the biggest market share in 2025.
- By product, the consumables segment is expected to expand at the fastest CAGR between 2026 and 2035.
- By method, the low-temperature sterilization segment held a major market share in 2025.
- By method, the high temperature segment is expected to expand at the fastest CAGR from 2026 to 2035.
- By end use, the hospitals segment held the largest market share in 2025.
- By end use, the ambulatory surgical centers segment is expected to expand at the fastest CAGR between 2026 and 2035.
Why is the Terminal Sterilization Market Gaining Momentum?
The terminal sterilization market is witnessing significant growth, as sterile pharmaceuticals, biologics, and medical devices are becoming increasingly necessary. Demand is being driven by rising surgical procedures, increased production of injectable drugs, and strict product safety regulations. To guarantee contamination-free products, technologies like steam, ethylene oxide, and radiation sterilization are widely used. Global market expansion is also being supported by the biopharmaceutical industry's expansion and the emphasis on infection prevention in healthcare settings.
Future Market Overview
- Growing Biopharmaceutical Production: The rising pipeline of biologics and cell & gene therapies presents strong opportunities for advanced sterilization solutions.
- Technological Innovation in Low-Temperature Sterilization: Development of novel sterilization techniques for temperature-sensitive products can unlock new market segments.
- Emerging Market Expansion: Countries in Asia-Pacific, Latin America, and the Middle East offer untapped growth potential due to expanding healthcare investments.
- Strategic Partnerships and Outsourcing Models: Collaboration between manufacturers and contract sterilization providers can drive operational efficiency and market penetration.
- Digitalization and Smart Manufacturing Integration: Implementation of AI-based validation, real-time tracking, and Industry 4.0 practices can enhance productivity and compliance.
How is Artificial Intelligence Transforming the Terminal Sterilization Market?
Artificial intelligence is reshaping the terminal sterilization market by improving quality control, process monitoring, and proactive maintenance. AI-powered systems optimize temperature, pressure, and exposure time by analyzing real-time data from sterilization cycles, which lowers errors and boosts productivity. Manufacturers can ensure regulatory compliance, reduce product recalls, and identify deviations early with the aid of advanced analytics. AI also facilitates automation and intelligent validation procedures, allowing for quicker batch releases and increased operational efficiency in the production of pharmaceuticals and medical devices.
Key Trends in the Terminal Sterilization Market
- Rising Adoption of Advanced Sterilization Technologies: Growing use of low-temperature and radiation-based sterilization methods to protect heat-sensitive biologics and complex medical devices.
- Integration of AI and Automation: Smart monitoring systems and automated cycle validation improve efficiency, accuracy, and regulatory compliance.
- Increasing Demand for Injectable and Biologic Drugs: The surge in vaccines, biosimilars, and monoclonal antibodies is boosting the need for reliable terminal sterilization processes.
- Stringent Regulatory Compliance Requirements: Regulatory authorities are enforcing strict sterility assurance levels, pushing manufacturers toward validated terminal sterilization solutions.
- Expansion of Contract Sterilization Services: Pharmaceutical and medical device companies are increasingly outsourcing sterilization to specialized service providers to reduce costs.
- Growth in Single-Use and Disposable Medical Devices: Higher demand for syringes, catheters, and surgical kits is fueling sterilization capacity expansion.
- Focus on Sustainable and Eco-Friendly Methods: Companies are adopting environmentally safer sterilization agents and energy-efficient technologies.
- Increasing Healthcare Infrastructure in Emerging Markets: Rapid hospital expansion and local pharmaceutical manufacturing are creating new demand for sterilization systems.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 14.12 Billion |
| Market Size in 2026 | USD 45.92 Billion |
| Market Size by 2035 | USD 30.57 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 8.03% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Method, End Use ,and region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Product Insights
What Made Equipment the Dominant Segment in the Terminal Sterilization Market?
The equipment segment dominated the market with the largest share in 2025 because of the widespread use of sterilizers like steam radiation systems and ethylene oxide in pharmaceutical and medical device manufacturing facilities. The segment's leadership is attributed to increased capital investments in automated and high-capacity sterilization equipment, as well as strict regulatory requirements for validated sterility assurance. Additionally, the need for sophisticated programmable sterilization systems increased due to the expansion of biologics production and the production of complex devices.
The consumables segment is expected to grow at the fastest CAGR in the coming years due to the ongoing need for biological testing kits, pouches, indicators, and sterilization wraps. Consistent use of these products is driven by rising surgical volumes and standard sterilization monitoring procedures. Growth is also being accelerated by the move toward single-use sterilization accessories to avoid cross-contamination. Furthermore, hospitals and manufacturers are making consistent repeat purchases thanks to stringent compliance requirements for routine sterility testing.
Method Insights
Why Did the Low Temperature Sterilization Segment Dominate the Market?
The low temperature sterilization segment dominated the terminal sterilization market in 2025 because it works well with heat-sensitive goods like biologics, injectables, and cutting-edge medical equipment. The use of techniques like EtO and hydrogen peroxide sterilization has grown due to the increasing production of pharmaceutical formulations that are sensitive to temperature. Additionally, segment growth is aided by the development of minimally invasive surgical instruments that require careful material handling. Its capacity to preserve product integrity while guaranteeing high levels of sterility assurance strengthened its position as the market leader.
The high temperature segment is expected to grow at the fastest CAGR in the coming years because of its widespread use in hospital sterilization units to reduce cycle times and affordability. The need for quick instrument reprocessing and an increase in surgical procedures is driving its future expansion. For durable medical equipment in particular, stream sterilization is still the recommended technique. High-temperature sterilization methods are increasingly adopted by healthcare facilities due to their low cost, operational simplicity, and reliability in ensuring effective sterilization.
End Use Insights
Why Did the Hospitals Segment Dominate the Terminal Sterilization Market?
The hospitals segment dominated the market in 2025 because hospitals perform a lot of inpatient and surgical procedures, necessitating regular sterilization of medical equipment and supplies. Their market dominance is further reinforced by stringent infection control procedures and on-site sterilization infrastructure. Additionally, large tertiary care hospitals run central sterile services departments, which boost the use of equipment. Increased emphasis on lowering hospital-acquired infections has made sterilization procedures in these facilities even more stringent.
The ambulatory surgical centers segment is expected to grow at the fastest CAGR in the coming years because minimally invasive and outpatient procedures are becoming increasingly popular. The need for quick turnaround, effective sterilization solutions is increased by these facilities, which increases the need for small cutting-edge systems. ASCs' operational scale is growing because of procedures being moved from hospitals due to rising healthcare costs. Consequently, ASCs are continuously investing more in contemporary sterilization technologies.
Regional Insights
North America Terminal Sterilization Market Size and Growth 2026 to 2035
The North America terminal sterilization market size is estimated at USD 17.43 billion in 2025 and is projected to reach approximately USD 29.75 billion by 2035, with a 5.49% CAGR from 2026 to 2035.
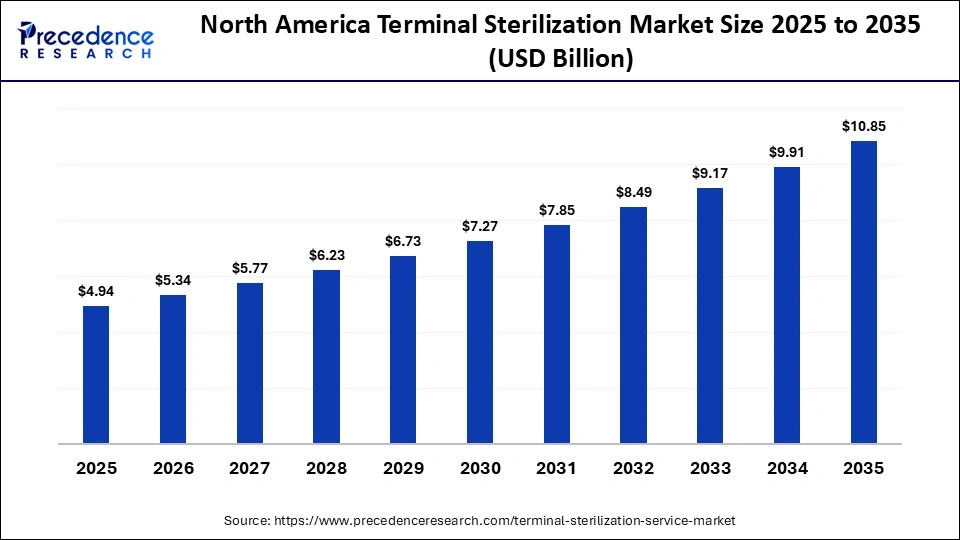
What Made North America the Dominant Region in the Terminal Sterilization Market?
North America dominated the market while holding the largest share in 2025 because of its robust regulatory framework, well-established healthcare system, and widespread use of cutting-edge sterilization technologies. Regional dominance was further reinforced by the existence of important industry players and substantial pharmaceutical manufacturing operations. Sustained demand was influenced by high healthcare costs and rigorous adherence to sterility assurance regulations. Furthermore, early automation adoption and ongoing technological innovation enhanced market leadership.
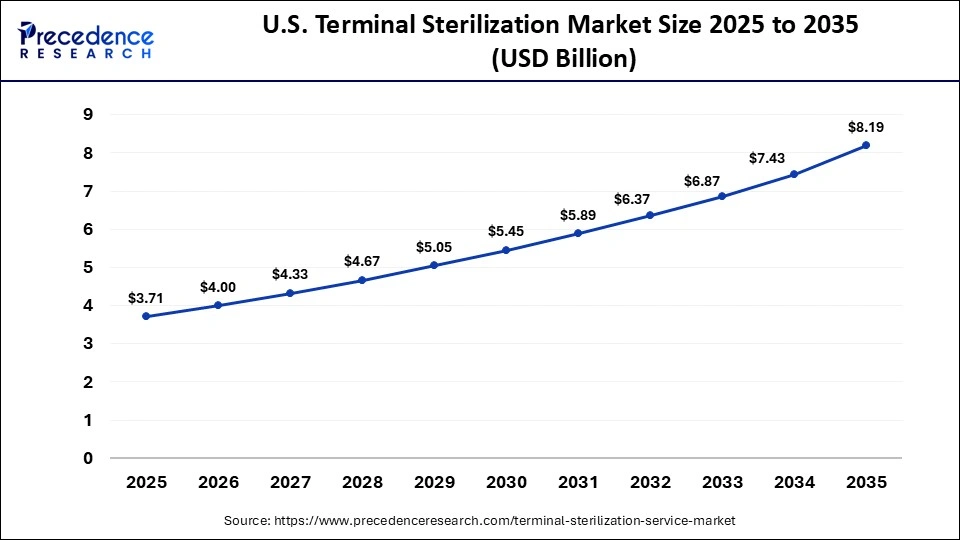
U.S. Terminal Sterilization Market Size and Growth 2026 to 2035
The U.S. terminal sterilization market size is calculated at USD 13.07 billion in 2025 and is expected to reach nearly USD 22.46 billion in 2035, accelerating at a strong CAGR of 5.56% between 2026 and 2035.
U.S. Terminal Sterilization Market Trends
The market in the U.S. is driven by growing outsourcing to contract sterilization, which provides tight sterility regulations and rising biologics production. Sustainable sterilization technologies and AI-enabled monitoring systems are also becoming more popular nationwide. Demand is being further increased by the growth of domestic pharmaceutical and medical device manufacturing. Long term growth is also supported by rising investments in infrastructure for infection prevention.
How is the Opportunistic Rise of Asia Pacific in the Market?
Asia Pacific is expected to grow at the fastest CAGR in the coming years because of growing pharmaceutical production, growing healthcare infrastructure and growing healthcare infrastructure and growing infection control consciousness. Demand is rising even more due to government programs encouraging regional drug manufacturing. Increasing foreign direct investments in life science and healthcare is bolstering regional capacities. Sterilization of equipment adoption is also being fueled by hospital expansion projects and increasing surgical volumes.
India Terminal Sterilization Market Trends
The market in India is expanding due to the rising production of medical devices and the expansion of the pharmaceutical industry. The need for validated sterilization systems is growing as hospital infrastructure is developed, and regulations are enforced more strictly. Capacity expansion is also being encouraged by government initiatives that support Make in India medical devices. Additionally, modernizing sterilization procedures in healthcare facilities is being driven by growing awareness of hospital-acquired infection prevention.
Terminal Sterilization Market Value Chain Analysis

Key Government Initiatives Supporting the Terminal Sterilization Market
| Country/Region | Government | Initiative | Impact on Market |
| United States | U.S. Food and Drug Administration (FDA) | Sterility assurance guidelines under Current Good Manufacturing Practices (cGMP) | Mandates validated terminal sterilization processes for pharmaceuticals and medical devices. |
| United States | Centers for Disease Control and Prevention (CDC) | Infection prevention and control guidelines | Promotes strict sterilization standards in healthcare facilities |
| Europe | European Medicines Agency (EMA) | Good Manufacturing Practice (GMP) regulations | Requires validated sterilization methods for market approval in EU countries |
| Europe | European Commission | Medical Device Regulation (MDR) | Strengthens sterility compliance requirements for device manufacturer. |
| India | Central Drugs Standard Control Organization (CDSCO) | Medical device and drug manufacturing regulations | Enforces sterility validation and quality standards in production |
| India | Ministry of Health and Family Welfare | National infection control programas | Encourages adoption of standardized sterilization practices in hospitals. |
| Global | World Health Organization (WHO) | Global guidelines on sterilization and infection prevention | Supports harmonized sterility standards across developing and developed nations. |
Terminal Sterilization Market Companies
- STERIS plc
- Sotera Health Company
- Getinge AB
- Advanced Sterilization Products
- 3M Company
- Belimed AG
- Steelco S.p.A.
- MMM Group
- Matachana Group
- Fedegari Group
- Tuttnauer
- Cantel Medical
- Olympus Corporation
- E-BEAM Services, Inc.
- BGS Beta-Gamma-Service GmbH
- Medistri SA
- Servizi Italia S.p.A.
- Andersen Sterilizers / Andersen Products
- Noxilizer, Inc.
- TSO3 Inc.
- Consolidated Sterilizer Systems
Recent Developments
- In September 2025, Noxilizer, Inc. secured $30 million in growth capital to expand commercial-scale nitrogen dioxide terminal sterilization capacity, supporting the global transition away from ethylene oxide for sensitive biologics.(Source: https://www.prnewswire.com)
- In June 2025, Solventum introduced the Attest Super Rapid VH2O2 Clear Challenge Pack and FDA-cleared solution providing biological indicator results in 24 minutes to monitor vaporized hydrogen peroxide sterilization.(Source: https://www.pharmaindustrial-india.com)
- In April 2025, De Lama announced being awarded as the top leader for pharmaceutical sterilization solutions in Europe 2025 by Pharmaceutical Outlook, highlighted for its legacy of innovation. The company was recognized for its HyPerPure hydrogen peroxide deep vacuum sterilizer and its commitment to sustainability. This award underscores De Lama's integration of washing and sterilization into a single, efficient, and continuous workflow. (Source: https://www.delama.it)
Segments Covered in the Report
By Product
- Equipment
- Consumables
- Service & Maintenance
- Connectivity Solutions and Software
By Method
- Low Temperature
- Hydrogen Peroxide (H2O2)
- Ethylene Oxide (EtO)
- Others
- High Temperature (Steam Sterilization)
- Systems
- Consumables
By End Use
- Hospitals & Clinic
- Pharma and Nutraceuticals
- Ambulatory Surgical Centres (ASCs)
- Medical Device Manufacturers/Packaging
- Others
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
