What is the Antimycotics Market Size?
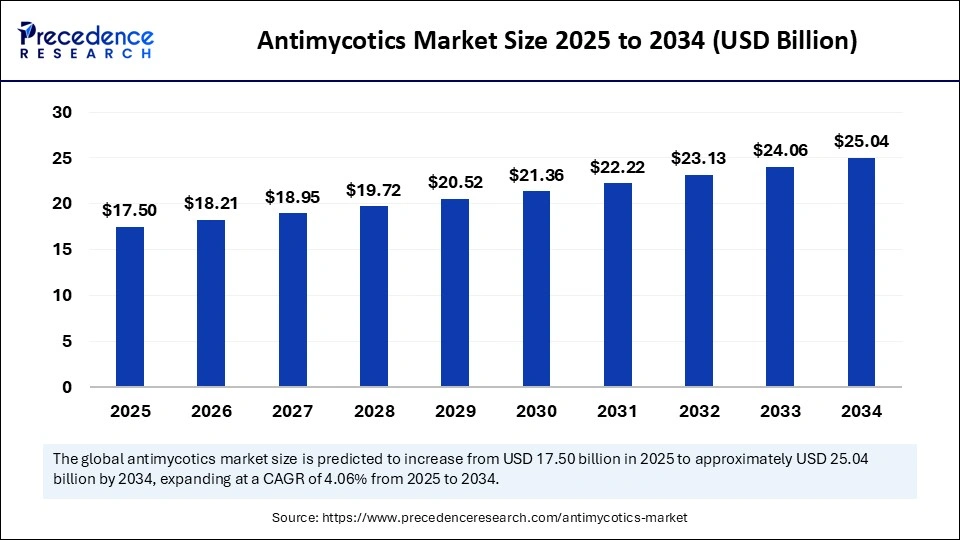
The global antimycotics market size accounted for USD 17.50 billion in 2025 and is predicted to increase from USD 18.21 billion in 2026 to approximately USD 25.04 billion by 2034, expanding at a CAGR of 4.06% from 2025 to 2034. This market is growing due to the rising prevalence of fungal infections worldwide and increasing demand for effective antifungal therapies.
Market Highlights
- North America dominated the market, holding the largest market share of 43.80% in 2024.
- Europe is expected to grow at a notable CAGR of 4.3% from 2025 to 2034.
- By drug class, the azoles segment held the largest share of the antimycotics market, accounting for 40.5% in 2024.
- By drug class, the polyenes segment is expected to grow at the fastest CAGR of 4.2% from 2025 to 2034
- By infection type, the systemic infections segment contributed the largest market share of 65.5% in 2024.
- By infection type, the superficial infections segment is expected to grow at the fastest rate of 4.4% from 2025 to 2034.
- By indication, the candidiasis segment contributed the biggest market share of 65.5% in 2024.
- By indication, the dermatophytosis segment is set to grow at the highest CAGR of 4.1% from 2025 to 2034.
- By route of administration, the oral segment generated the major market share of 55.5% in 2024.
- By route of administration, the injectable segment is expected to grow at the fastest CAGR of 4.2% from 2025 to 2034.
- By end user, the hospitals segment held the biggest market share of 45.5% in 2024.
- By end user, the specialty clinics segment is growing at a CAGR of 4.5% from 2025 to 2034.
What Is the Antimycotics Market?
The antimycotics market is expanding rapidly due to the growing demand for novel and enhanced treatments, as fungal infection rates rise from immunosuppression and hospital-acquired infections. The safety and effectiveness of treatments are being improved by technological developments such as new antifungal compounds, improved delivery methods, and quick diagnostic tools.
- In September 2025, Propedix launched Dryello (tolnaftate 1%), the first dry-stick over-the-counter antifungal for athlete's foot with a novel format that aims to enhance user convenience and adherence.
- In April 2025, the WHO released its first comprehensive pipeline review of antifungal agents, providing a strategic view of clinical and preclinical antifungal development and gaps in the sector.
Key Technological Shifts in the Antimycotics Market
- Rise of Topical Antimycotics: Introducing topical combinations and extended-release antifungal formulations to increase patient compliance and efficacy. Additionally, these innovations seek to improve patient convenience and decrease the frequency of dosing.
- Rapid Diagnostics Integration: Identification of fungal infections is facilitated by the use of molecular assays and point-of-care diagnostics, which allow for prompt treatment. Preventing severe or invasive infections is another benefit of early detection.
- Biological and Immunotherapy Approaches: Increasingly invasive or resistant fungal infections are being targeted by manipulatory drugs and monoclonal antibodies. These methods give patients who don't respond to traditional antifungal medications new choices.
Antimycotics Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 17.50 Billion |
| Market Size in 2026 | USD 18.21 Billion |
| Market Size by 2034 | USD 25.04 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 4.06% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Drug Class, Infection Type, Indication, Route of Administration, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Antimycotics Market Segment Insights
[[segment_insights]]
Antimycotics Market Regional Insights
[[regional_insights]]
Antimycotics Market 2024 - Who is Leading and Who is Catching Up?
| Region | Market Status | Key Growth Drivers | Government/Public Health Support | Strategic Opportunities |
| North America | Dominating | Strong R&D ecosystem, high prevalence of Candida auris infections, and early adoption of advanced antifungal drugs | CDC's Antimicrobial Resistance Lab Network (ARLN) and NIH funding for fungal disease genomics | Expansion of antifungal resistance monitoring networks; AI-based infection diagnostics are being integrated in hospitals |
| Europe | Fastest Growing | High incidence of hospital-acquired infections, growing elderly population, and strong AMR surveillance | European Center for Disease Prevention and Control (ECDC) and Horizon Europe grants supporting antifungal R&D | Rapid adoption of long-acting echinocandins; the EU's focus on local drug manufacturing under the EU4Health policy |
| Asia Pacific | High Growth Potential | Increasing cases of superficial fungal infections, expansion of hospital infrastructure, and rising generic manufacturing | India's National AMR Action Plan, Japan's PMDA Innovation Program, and China's Healthy China 2030 initiative | Opportunities in developing topical antifungals and affordability-based pricing strategies; regional biotech collaborations are growing |
| Latin America | Notably Emerging | Urbanization, tropical climates, and limited access to fungal diagnostics create unmet demand. | Pan American Health Organization (PAHO) programs are improving fungal disease surveillance in Brazil, Argentina, and Chile. | Potential for expansion through public-private partnerships; growing e-pharmacy channel for OTC antifungal creams |
Antimycotics Market Value Chain
[[value_chain]]
Antimycotics Market Companies
[[market_company]]
Recent Developments
- In March 2025, Zydus Lifesciences received final approval from the U.S. Food and Drug Administration (USFDA) to manufacture and distribute Ketoconazole Shampoo 2%. This antifungal medication is used to treat dandruff, seborrheic dermatitis, and other fungal scalp infections. The approval positions Zydus to compete directly with the reference listed drug, Nizoral Shampoo 2%, in the U.S. market. (Source: https://zyduslife.com)
- In June 2024, Biocon received approval from the USFDA for its complex injectable antifungal medication, Micafungin. This drug is used to treat various fungal and yeast infections, including invasive candidiasis and esophageal candidiasis. The approval enhances Biocon's portfolio of vertically integrated, complex drug products.(Source: https://www.biocon.com)
Antimycotics Market Segment Covered in Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
