What is the Cardiac Troponin Market Size?
The global cardiac troponin market size is calculated at USD 4.30 billion in 2025 and is predicted to increase from USD 4.75 billion in 2026 to approximately USD 11.44 billion by 2035, expanding at a CAGR of 10.28% from 2026 to 2035.
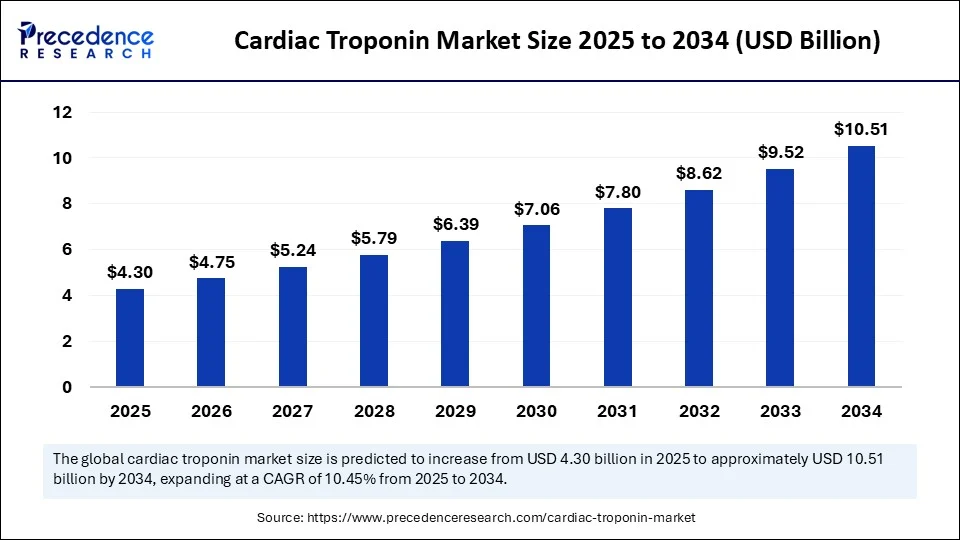
Cardiac Troponin Market Key Takeaways
- In terms of revenue, the global cardiac troponin market was valued at USD 4.30 billion in 2025.
- It is projected to reach USD 11.44 billion by 2035.
- The market is expected to grow at a CAGR of 10.28% from 2026 to 2035.
- North America dominated the global cardiac troponin market with the largest market share of 38% in 2025.
- Asia Pacific is expected to expand at the fastest CAGR between 2026 and 2035.
- By type, the troponin I (cTnI) segment held the biggest market share of 56% in 2025.
- By type, the troponin T (cTnT) segment is anticipated to grow at a remarkable CAGR between 2026 and 2035.
- By test type, the laboratory-based testing segment captured the highest market share of 62% in 2025.
- By test type, the point-of-care testing (POCT) segment is expected to expand at a notable CAGR over the projected period.
- By sample type, the serum segment contributed the largest market share of 45% in 2025.
- By sample type, the other segment is expected to expand at a significant CAGR over the projected period.
- By technology, the chemiluminescence immunoassay (CLIA) segment generated the major market share of 41% in 2025.
- By technology, the other (biosensors, electrochemical) segment is expected to expand at a notable CAGR over the projected period.
- By end use, the hospitals & clinics segment held a remarkable market share of 49% in 2025.
- By end use, the homecare settings segment is expected to expand to a notable CAGR over the projected period.
- By application, the myocardial infarction diagnosis segment accounted for significant market share of 58% in 2025.
- By application, the ACS risk stratification segment is expected to expand at a notable CAGR over the projected period.
Market Overview
The cardiac troponin market refers to the global landscape of diagnostic solutions used for detecting cardiac troponin proteins (cTnI and cTnT) in blood, which are highly specific and sensitive biomarkers for myocardial infarction (MI) and acute coronary syndromes (ACS). Troponins are structural proteins released into the bloodstream when heart muscles are damaged, and their rapid and accurate detection is vital for early diagnosis and timely treatment. The market includes instruments, reagents, and assays used in point-of-care and laboratory settings across hospitals, clinics, and diagnostic laboratories.
How is AI Revolutionizing the Cardiac Troponin Market?
Artificial intelligence is disrupting the cardiac troponin market by enhancing speed, accuracy, and availability of the test. AI optimizes troponin test results accurately, reducing errors and enhancing decision-making. A rapid troponin test, combined with machine learning algorithms, can identify the risks of a heart attack more quickly and accurately.
- In February 2025, Researchers at UCLA constructed an AI-enabled chemiluminescence sensor that produces troponin I results equivalent to lab results in 25 min, improving the timeliness of emergency care and furthering early intervention strategies. (Source: https://www.ee.ucla.edu)
What are the Major Growth Factors of the Cardiac Troponin Market?
- Rising Cases of Cardiovascular Disease: The rising incidence of heart attacks and other cardiac-related disorders around the globe is increasing the demand for reliable biomarkers such as troponins so that early diagnosis and effective clinical response can occur.
- Availability of High-Sensitivity Tests: The emergence of high-sensitivity troponin assays enables the detection of damaged heart tissue sooner. In turn, it improves the diagnostic accuracy and clinical treatment approach for patients. Ultimately, the understanding that high-sensitivity assays improve patient outcomes is leading to the global acceptance and use by clinicians in emergency and acute care settings.
- Expansion of Emergency Services: As emergency departments and cardiac care units expand around the world, there is a growing need for rapid, reliable biomarkers, such as cardiac troponins, to inform timely clinical decision-making and treatment.
- Aging Population Worldwide: With an increasing population of elderly people, the propensity for cardiac events increases, leading to the utilization of cardiac biomarkers both as a routine and acute standard for diagnosis and ongoing well-being.
- Increased Use of Point-of-Care Testing: The growing use of portable diagnostic equipment in ambulatory or even home care settings, such as troponin testing, is enhancing access to these cardiac biomarkers for a larger population, particularly those residing in under-resourced or more remote areas.
Market Scope
| Report Coverage | Details |
| Market Size by 2035 | USD 11.44 Billion |
| Market Size in 2025 | USD 4.3 Billion |
| Market Size in 2026 | USD 4.75 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 10.28% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Type, Test Type, Sample Type, Technology, End Use, Application and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Rising Demand for Rapid Diagnostics
One of the major factors driving the growth of the cardiac troponin market is the rising demand for rapid, high-sensitivity troponin (hs-cTn) testing. Recent reports have demonstrated that U.S. entities have shown hs-cTn assays to have not only improved analytical specificity for myocardial injury indications, but also to improve patient outcomes and processing efficiencies in the emergency care continuum. Laboratory guidelines recommend hs-cTn test results be communicated to physicians in less than sixty minutes from the receipt of the sample, and some point-of-care devices can deliver results in less than twenty minutes.
(Source: https://www.sciencedirect.com)
The clinical practice framework of health systems across North America has now incorporated rapid troponin-based clinical pathways that safely rule out myocardial infarction in 1-2 hours, relieving some congestion in emergency rooms and increasing patient flow. Diagnostic protocols sanctioned by the government, as well as Laboratory Standards, have only solidified the utility and impact of fast and reliable troponin assays, creating a large demand for the global supply chain and infrastructure supporting and testing.
Restraint
Regulatory Hurdles
A significant factor limiting the growth of the cardiac troponin market is the strict regulatory requirements imposed by Europe's In Vitro Diagnostic Medical Device Regulation (IVDR), which has heightened approval barriers and hindered the availability of new or laboratory-developed troponin assays. The IVDR imposes stringent performance validation, clinical evidence, and post-market surveillance requirements, necessitating confirmation of follow-up activities in relation to the requirements under the previous IVDD scheme.
Many former in-house tests, including laboratory-developed tests, must now either be fully compliant or must be reclassified, while the notifying bodies under the member state's capacity are constrained. These regulatory cultures limit the speed-to-market of new high-sensitivity and point-of-care troponin assays, especially in Europe and nearby markets, and curtail overall accessibility for clinicians as demand rapidly increases.
Opportunity
Will Point-of-Care Troponin Testing Change the Future of the Market?
One of the greatest opportunities in the cardiac troponin market is the rapid adoption of point-of-care (POC) technologies, particularly in emergency settings or for remote care. In November 2024, the U.S. FDA approved Abbott's i-STAT TnI-Nx assay for use in ER settings, providing troponin I results, allowing much faster diagnosis of myocardial infarction. POC testing is beneficial for hospitals that lack adequate testing solutions. POCT allows rapid troponin testing at the patient's bedside, reducing turnaround times and improving patient management. (Source: https://clinicaltrials.gov)
Furthermore, the World Health Organization (WHO) has prioritized the expansion of decentralized diagnostic testing tools as part of its Global Health Emergency preparedness plan. Cardiovascular diseases are still the leading cause of mortality globally, which makes fast and portable troponin diagnostics a strong opportunity to hasten clinical decisions and increase access to care for patients in need.
Segment Insights
[[segment_insights]]
Regional Insights
[[regional_insights]]
Value Chain Analysis
- Raw material sourcing
Sourcing raw materials for this market involves the procurement of high purity recombinant proteins, buffer regeants, antibodies and surface chemistries. Suppliers are seen shifting towards advanced recombinant protein platforms in order to improve batch consistency as there is a high demand for high sensitivity troponin tests all across emergency care. Rising costs for raw materials and strict regulatory bodies further push the market.
Key Players: Merck, Abcam, Bio Rad - Manufacturing and Processing
This manufacturing process involves preparing reageants, fitting them into cartridges and then building analyzer systems that are able to read results. Production facilities must maintain strict sterile conditions and follow international safety standards. Many companies produce both, central lab assays as well as point of care kits. The rise of automation software is improving the consistency of this stage.
Key Players: Roche, Abbott, Siemens - Distribution and Logistics
The next stage is distribution, which involves the shipping of kits, tests, analyzers and consumables to hospitals, diagnostics labs, point of care sites and emergency care centers. Because these troponin kits are temperature sensitive, companies usually rely on cold chain logistics and validated supply partners. MNCs also often use direct distribution processes for major hospitals.
Key Players: Cardinal Health, Fisher Healthcare, Medline Industries
Cardiac Troponin Market Companies

- Abbott Laboratories: Known for high-sensitivity troponin assays, Abbott Laboratories offers a comprehensive portfolio of cardiac markers, including troponin tests, which are crucial for diagnosing acute myocardial infarction and other cardiac conditions.
- Siemens Healthineers: Siemens Healthineers provides rapid, reliable testing solutions that integrate into their diagnostic ecosystem, making them a preferred choice for hospitals and laboratories.
- Roche Diagnostics: Roche Diagnostics offers a broad portfolio of cardiac markers, including troponin tests, which are essential for the early detection and prevention of heart failure and other cardiac conditions.
Other Major Key Players
- Beckman Coulter (Danaher Corporation)
- bioMérieux SA
- Ortho Clinical Diagnostics (QuidelOrtho)
- Becton, Dickinson and Company (BD)
- Radiometer Medical (Danaher)
- Bio-Rad Laboratories
- LSI Medience Corporation
- Tosoh Corporation
- Randox Laboratories
- Thermo Fisher Scientific
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
- Singulex, Inc.
- HyTest Ltd
- Response Biomedical Corp.
- Getein Biotech Inc.
- LifeSign LLC
- Creative Diagnostics
Recent Developments
- In September 2025, Roche's sixth-generation Troponin T test offered a new level of accuracy critical for diagnosing heart attacks. The test helps clinicians quickly identify heart attack and rule out non-cardiac causes, ensuring patients receive the care they need at the earliest opportunity. Results showed that the Elecsys Troponin Gen 6 test was able to identify acute myocardial infarction (AMI), or heart attack, and identify those not having an AMI, with a high level of precision. This supports the efficient triage of patients arriving at the emergency department, ensuring healthcare resources can be focused where they are needed most.
(Source: taiwannews.com ) - In November 2025, QuidelOrtho Corporation recently received 510(k) clearance from the U.S. Food and Drug Administration for its VITROS Troponin I Assay, the company. The news comes as the stock trades at $26.99, significantly below its 52-week high of $49.45, with shares down over 8% in the past week. According to InvestingPro analysis, the company appears undervalued based on its Fair Value assessment.
(Source: investing.com ) - In March 2024, Polymedco received FDA clearance for the PATHFAST hs-cTnI-II test as the first high-sensitivity cardiac troponin I assay that has been approved for point-of-care use in the US. The test provides accurate results within 17 minutes to help with rapid heart attack diagnosis.
(Source: prnewswire.com) - In October 2023, HyTest launched a new recombinant human cardiac troponin complex that combines cTnI and TnC, that can have improved accuracy on the calibrators and controls. This advancement offers the opportunity to better reproduce troponin forms seen in clinical patient samples with myocardial infarction.
(Source: hytest.fi) - In October 2023, Mindray expanded its cardiac biomarker offering with the launch of hs-cTnI and NT-proBNP assays developed with HyTest. The aim of the new assays is to facilitate earlier identification, diagnosis, and monitoring of cardiovascular disease - with the application throughout global healthcare settings.
(Source: dicardiology.com)
Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
