What is the Follicular Lymphoma Market Size?
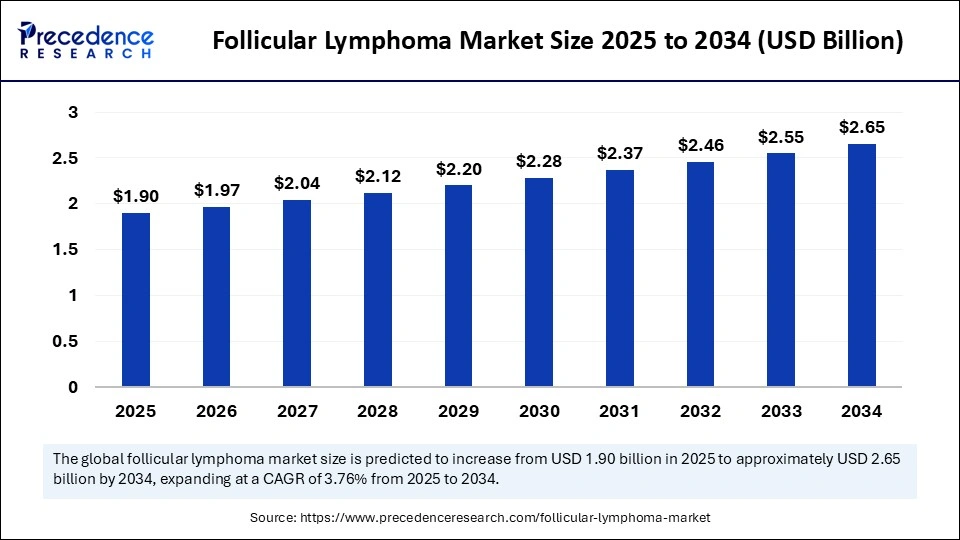
The global follicular lymphoma market size is calculated at USD 1.90 billion in 2025 and is predicted to increase from USD 1.97 billion in 2026 to approximately USD 2.65 billion by 2034, expanding at a CAGR of 3.76% from 2025 to 2034. The follicular lymphoma market is witnessing unprecedented growth, driven by the rising prevalence of follicular lymphoma as a common type of non-Hodgkin lymphoma, which is expanding the patient pool requiring treatment.
Market Highlights
- North America dominated the market, holding the largest market share in 2024.
- The Asia-Pacific is expected to expand at the fastest CAGR between 2025 and 2034.
- By treatment type, the chemotherapy segment dominated the market in 2024.
- By treatment type, the targeted therapy segment is expected to grow at the fastest CAGR from 2025 to 2034.
- By end user, the hospitals segment held the largest market share in 2024.
- By end user, the cancer research institutes segment is expected to grow at a significant CAGR between 2025 and 2034.
Follicular Lymphoma Market Landscape
Follicular lymphoma (FL) is one of the most common types of slow-growing (indolent) non-Hodgkin lymphoma (NHL) and arises from germinal center B cells. It has a long-term disease course, owing to the severe chronic and incurable nature of the disease in the majority of patients. Although the prognosis for early and advanced stage disease has improved due to the therapeutic advances, the choice of treatment for follicular lymphoma is generally dependent on patient and disease characteristics. It is generally characterized by significant lymph node enlargement, an enlarged spleen, and bone marrow involvement.
Many patients also present with fatigue, night sweats, or unintentional weight loss, although some remain asymptomatic for long periods and are monitored under a watchful waiting approach. Modern therapies such as anti-CD20 monoclonal antibodies, targeted inhibitors, and combination chemoimmunotherapy have extended survival, but relapses are common and require individualized long-term management strategies.
How Are AI-Driven Innovations Reshaping the Follicular Lymphoma Market?
In the rapidly evolving technological landscape, the integration of Artificial Intelligence (AI) holds great potential to accelerate the growth of the follicular lymphoma market by enhancing diagnosis, prognosis, drug discovery, and treatment planning. By effectively analyzing large datasets, AI-powered tools provide more efficient insights, helping clinicians make more accurate, data-driven decisions. Deep learning models, such as convolutional neural networks (CNNs), assist pathologists in analyzing digital histopathological images. AI accelerates the discovery and development of new therapies for follicular lymphoma, such as biomarker discovery and effective clinical trial design. AI algorithms can effectively detect and quantify lymphoma cells, analyze morphological features, and perform image analysis of immunohistochemical (IHC) stains faster and more consistently, thereby overcoming inter-observer variability among pathologists.
Follicular Lymphoma Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.90 Billion |
| Market Size in 2026 | USD 1.97 Billion |
| Market Size by 2034 | USD 2.65 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 3.76% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Treatment Type, End User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Follicular Lymphoma Market Segmental Insights
[[segment_insights]]
Follicular Lymphoma Market Regional Insights
[[regional_insights]]
Follicular Lymphoma Market Value Chain

Follicular Lymphoma Market Companies
[[market_company]]
Recent Developments
- In November 2025, Genmab A/S announced today that EPKINLY (epcoritamab-bysp) in combination with rituximab and lenalidomide (EPKINLY + R2) was approved by the U.S. Food and Drug Administration (FDA) for adult patients with relapsed or refractory (R/R) follicular lymphoma (FL). The approval is based on results from the pivotal Phase 3 EPCORE FL-1 study that evaluated fixed duration EPKINLY + R2 compared to standard of care R2.i(Source: https://ir.genmab.com)
- In November 2025, Tempus AI, Inc., a technology company leading the adoption of AI to advance precision medicine, announced a new research study sponsored by Tempus and being run in collaboration with the Institute for Follicular Lymphoma Innovation (IFLI), a global, nonprofit, private foundation dedicated to accelerating the development of innovative treatment options for patients with follicular lymphoma (FL), marking Tempus first study collaboration with a non-profit foundation.(Source: https://www.businesswire.com)
Follicular Lymphoma Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
