What is the HIV Diagnostics Market Size?
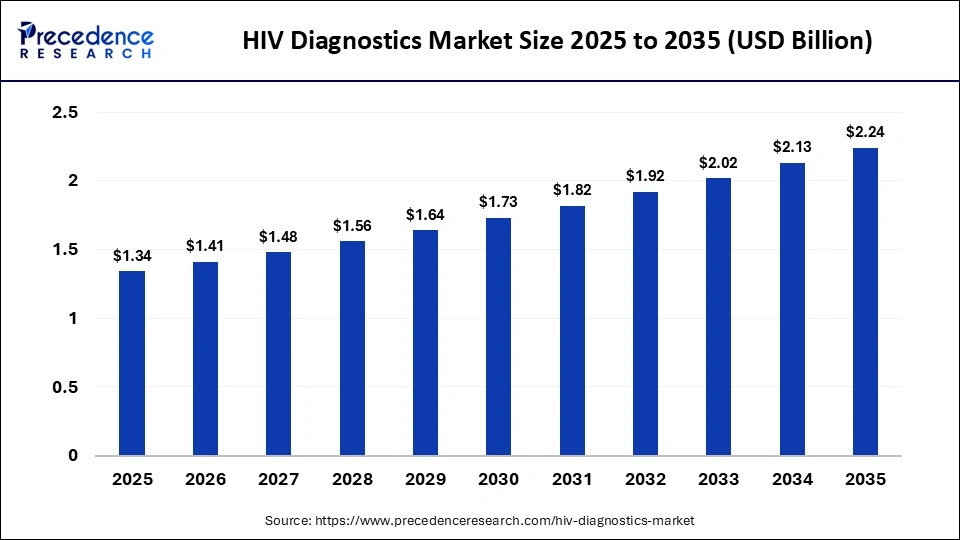
The global HIV diagnostics market size was calculated at USD 1.34 billion in 2025 and is predicted to increase from USD 1.41 billion in 2026 to approximately USD 2.24 billion by 2035, expanding at a CAGR of 5.26% from 2026 to 2035.The market is driven by surging awareness of AIDS among people, along with rapid investments in healthcare infrastructure.
Key Takeaways
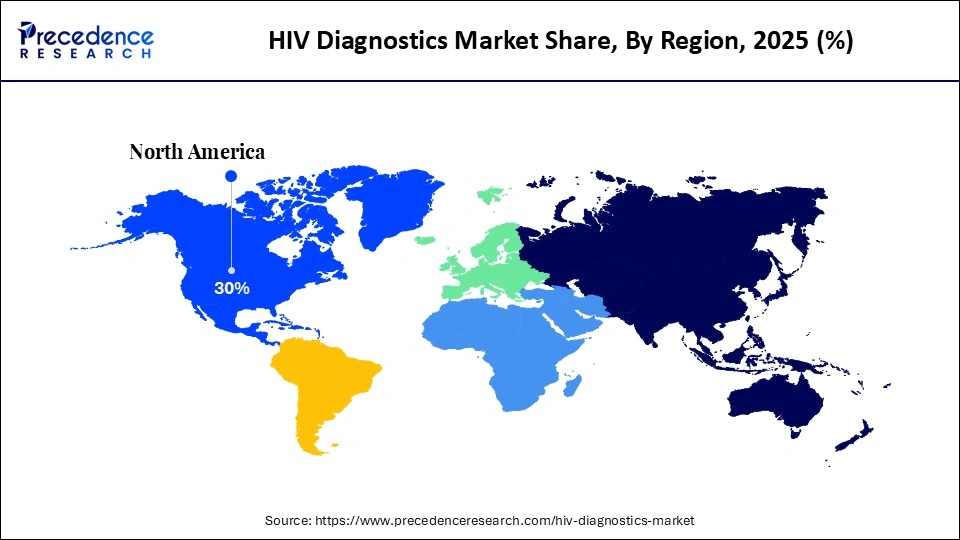
- North America led the market with the largest market share of 30% in 2025.
- Asia Pacific is expected to grow at the fastest CAGR during the forecast period.
- By product type, the test kits segment held the largest share of the market in 2025.
- By product type, the instruments segment is expected to expand with the highest CAGR during the forecast period.
- By test type, the antibody tests segment held the largest share of the market in 2025.
- By test type, the nucleic acid tests segment is expected to grow with the highest CAGR between 2026 and 2035.
- By technology, the ELISA segment held the highest share of the market in 2025.
- By technology, the rapid testing segment is expected to grow with the fastest CAGR during the forecast period.
- By end user, the hospitals segment dominated the market in 2025.
- By end user, the home care settings segment is expected to grow at a significant CAGR during the forecast period.
Market Overview
The HIV diagnostics market deals with the manufacturing, developing, and supplying of equipment, testing services, and software to detect HIV/AIDS (antibodies, antigens, or RNA) in humans. HIV diagnostics are performed primarily on blood or oral fluid to detect the human immunodeficiency virus (HIV), including antibodies, antigens (p24), or genetic material (RNA). The growing focus of the government on developing the HIV testing infrastructure, along with the rapid adoption of the ELISA/EIA test by the clinical experts to check AIDS in human beings, has boosted industrial growth. This industry is expected to grow significantly as the healthcare sector expands worldwide.
What is the Role of AI in the HIV Diagnostics Industry?
AI is playing a vital role in the growth of the HIV diagnostics industry. In the HIV diagnostics sector, AI delivers predictive risk modelling and provides improved screening and image analysis. AI-enabled algorithms help to analyze complex data, such as lab reports and multi-omics data, to detect HIV-related complications earlier, for enhancing timely diagnosis. Additionally, AI is used in forecasting treatment responses by analyzing genomic data to enable drug resistance and optimize antiretroviral therapy (ART).
- In October 2024, the McGill University Health Centre launched MARVIN. MARVIN is an AI-enabled chatbot designed to provide 24/7 expert-validated information on self-management-related solutions for people living with HIV.
HIV Diagnostics Market Trends
- Establishment of New HIV Centers: Numerous healthcare institutions are engaged in establishing HIV testing centers to detect AIDS in human beings. For instance, in June 2025. Damien Center opened a new HIV testing unit in Central Indiana, U.S. This new center was inaugurated to cater to patients across the U.S. region.
- Growing Cases of HIV: The surging prevalence of HIV globally has increased the demand for advanced testing procedures to accelerate treatment methodologies. According to the World Health Organization, in 2024, around 1.3 million people acquired HIV globally.
- Product Launches: Various home diagnostics solution providers are continuously engaged in launching a wide range of products to enhance HIV testing capabilities. For instance, in February 2026, OraSure Technologies, Inc launched OraQuick HIV Self-Test. The OraQuick HIV self-test has received approval from Health Canada for enhancing AIDS testing among the Canadian population.
- Government Initiatives: The governments of several countries, such as the UK, India, Argentina, and South Africa, have launched new initiatives to enhance HIV diagnostics capabilities. For instance, in December 2025, the government of the UK launched a new initiative. This new initiative aims at improving HIV testing in prominent cities such as London and Manchester.
- Partnerships: Various pharma brands are partnering with local governments to strengthen the HIV testing procedure in different parts of the globe. For instance, in September 2025, the local government of Manila partnered with Abbott. Through this partnership, Abbott will deliver a wide range of HIV testing kits across the Philippines.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.34 Billion |
| Market Size in 2026 | USD 1.41 Billion |
| Market Size by 2035 | USD 2.24 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 5.26% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product Type, Test Type, Technology, End User, and region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Product Type Insights
Why Did the Test Kits Segment Dominate the HIV Diagnostics Market?
The test kits segment dominated the HIV diagnostics market with a major share in 2025. This is mainly due to their affordability, applicability, and accuracy in delivering HIV results. Testing kits are rapidly used across numerous healthcare settings, such as diagnostic laboratories, hospitals, and community screening programs.
The instruments segment is expected to grow at the highest CAGR during the forecast period, owing to the surging adoption of automated HIV testing equipment in healthcare settings. Top companies dealing in HIV diagnostics equipment, such as Abbott Laboratories, Roche, and Siemens Healthineers, are developing novel instruments, contributing to the segmental growth.
Test Type Insights
What Made Antibody Tests the Leading Segment in the HIV Diagnostics Market?
The antibody tests segment led the HIV diagnostics market in 2025, owing to their surging adoption as the primary screening process for HIV detection across hospitals. Antibody-based testing is cost-effective, reliable, and suitable for large-scale screening initiatives. These testing solutions detect the body's immune response to HIV infection in diagnostic laboratories and blood banks. It is compatible with ELISA and rapid testing kits, for enhancing clinical performance and scalability.
The nucleic acid tests segment is expected to expand with the highest CAGR between 2026 and 2035, due to its ability to detect viral DNA at early stages of HIV infections by reducing the diagnosis time period. Nucleic acid tests are mostly adopted by modern hospitals to enable early diagnosis, prevent HIV transmission, and enhance patient monitoring capabilities.
Technology Insights
What Made ELISA the Leading Segment in the Market?
The ELISA segment led the HIV diagnostics market with the largest share in 2025, due to its specificity, high sensitivity, and rapid adoption in clinical laboratories. ELISA tests are designed for antibody and antigen testing in hospitals and blood banks. These testing solutions can process large samples with reliable results for enabling mass screening initiatives and public health programs.
The rapid testing segment is expected to grow at the highest CAGR during the forecast period, owing to the surging demand for quick and point-of-care diagnostics solutions. Rapid HIV testing delivers results within several minutes to enable immediate clinical decisions for controlling transmission rates. These testing technologies are mainly used in emergency settings and antenatal clinics, where laboratory infrastructure is not developed properly.
End User Insights
Why Did the Hospitals Segment dominate the Market?
The hospitals segment dominated the HIV diagnostics market in 2025, due to their superior role in large-scale screening, integrated patient management, and confirmatory testing. Hospitals are equipped with advanced technologies and modern screening solutions to handle large patient volumes across numerous departments, including emergency, outpatient, inpatient, and maternal care units, which increases the demand for routine HIV-testing. Private hospitals conduct a wide range of HIV tests, such as ELISA, viral load monitoring, and antigen testing.
The home care settings segment is expected to grow at a significant CAGR during the forecast period, owing to the growing consumer preference for confidential testing solutions. The increasing awareness about early HIV detection and preventive healthcare has increased the demand for self-testing kits for residential use. Moreover, the surging adoption of home-based diagnostics solutions among teenagers for reducing social stigma and improving HIV testing capabilities bolsters segmental growth.
Regional Insights
North America HIV Diagnostics Market Size and Growth 2026 to 2035
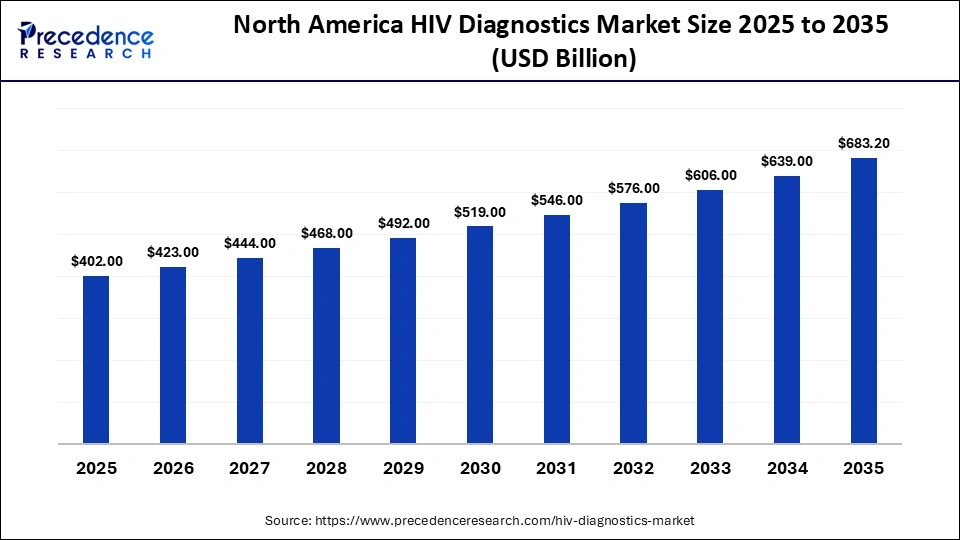
The North America HIV diagnostics market size is estimated at USD 402.00 billion in 2025 and is projected to reach approximately USD 683.20 billion by 2035, with a 5.45% CAGR from 2026 to 2035.
Why Did North America Dominate the HIV Diagnostics Market?
North America dominated the HIV diagnostics market in 2025, due to the rising emphasis of the healthcare organizations in opening new HIV testing centers, coupled with the rapid adoption of western blot test by diagnostic laboratories to carry out AIDS testing among the people. The prominent companies, such as Abbott, Alere Inc., and Cepheid, are engaged in designing a wide variety of HIV testing solutions, coupled with supportive governmental policies to enhance HIV diagnosis capabilities, have contributed to the development of the industry. Additionally, the rise in the number of startup companies dealing in medical diagnostics solutions, as well as the surging expansion of telehealth services, has driven the market expansion.
- In June 2025, Cepheid announced that Health Canada had approved Xpert HIV-1 Viral Load XC. Xpert HIV-1 Viral Load XC is a next-generation extended-coverage (XC) test used for monitoring HIV-1 infection.
U.S. HIV Diagnostics Market Size and Growth 2026 to 2035
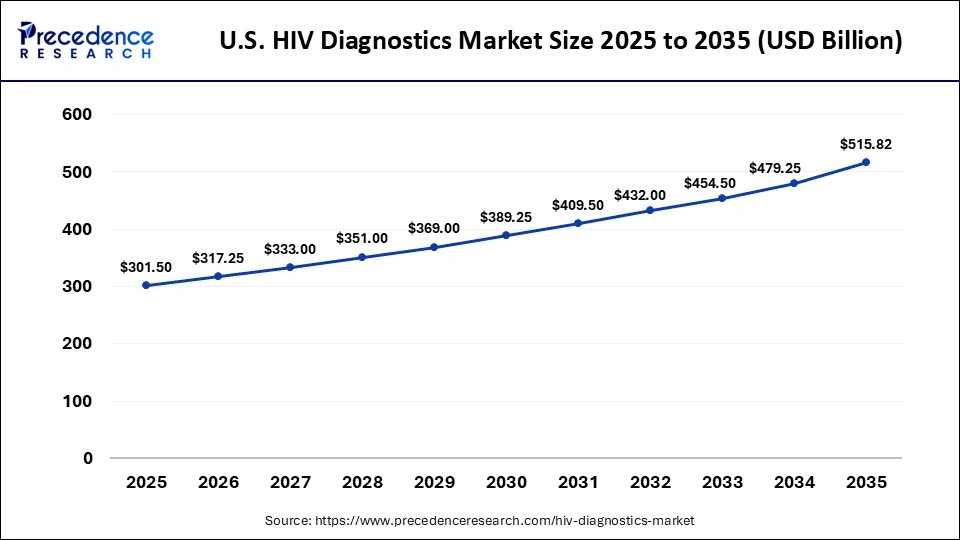
The U.S. HIV diagnostics market size is calculated at USD 301.50 billion in 2025 and is expected to reach nearly USD 515.82 billion in 2035, accelerating at a strong CAGR of 5.52% between 2026 and 2035.
U.S. HIV Diagnostics Market Trends
The U.S. leads the North American HIV diagnostics industry due to the rising awareness of people related to HIV disorders, coupled with the growing sales of home-based diagnostics solutions. Additionally, collaborations among hospital chains and government organizations to deploy advanced solutions in HIV testing units, along with technological advancements in the clinical diagnostics sector, have contributed to the market expansion.
Why is Asia Pacific Expanding with a Significant CAGR in the Market?
Asia Pacific is expected to expand with a significant CAGR during the forecast period, due to the growing cases of HIVAIDS in numerous countries, including Thailand and Papua New Guinea, along with the rapid establishment of HIV testing units. The integration of AI in diagnostic laboratories, as well as partnerships among software companies and healthcare providers to design advanced HIV testing software, has led the market to expand rapidly. Moreover, the surging preference of doctors to adopt PCR and ELISA technology for testing HIV in human beings is expected to foster the industry in the coming years. According to ourworldindata.org, around 1.5% population of Papua New Guinea had suffered from HIV AIDS during 2024.
China HIV Diagnostics Market Analysis
China is the major contributor to the Asia Pacific HIV diagnostics industry, owing to the availability of HIV testing kits on online platforms, as well as the rapid expansion of the pharma industry. Moreover, increased government funding for introducing HIV prevention initiatives, along with the surging adoption of molecular testing platforms by clinicians, has driven the market growth.
What are the Advancements in the HIV Diagnostics Market in Europe?
Europe is expected to have significant growth during the forecast period, driven by a combination of targeted testing initiatives and policy reforms across the region. The resurgence of HIV testing and the implementation of new testing policies in the region have driven the HIV diagnostics market, leading to high levels of research and development efforts. Countries like Germany, France, and the UK are leading players.
UK Market Trends
The adoption of digital solutions is a significant market driver in this region. This integration of technology into health services, including HIV diagnostics, reflects the UK's commitment to improving patient experiences and meeting the growing demand for efficient, accessible healthcare solutions.
What are the Key Trends in the HIV Diagnostics Market in Latin America?
Latin America is expected to have substantial growth in the market, driven by the region's progress in reducing HIV-related deaths. This reflects the growing availability of modern treatment options and early diagnostic tools that enable timely interventions. There is also a high focus on expanding access to diagnostic services and enhancing the effectiveness of treatment strategies. Countries like Brazil and Mexico are leading players.
Brazil Market Trends
The country's growth and development are driven by significant investments and international collaborations in HIV diagnostics. This initiative addresses critical gaps in national HIV programs and facilitates the adoption of cutting-edge technologies, enhancing the country's capacity to combat HIV.
How is the Middle East and Africa Region Growing in the HIV Diagnostics Market?
The Middle East and Africa region is expected to have lucrative growth throughout the forecast period, driven by advancements in HIV testing and treatments. There is also a steep decline in new infections among children, highlighting the effectiveness of expanded diagnostic and prevention programs. Countries such as Saudi Arabia, South Africa, and the UAE are leading players in the region.
Saudi Arabia Market Trends
The country's market growth and development are propelled by advancements in the availability and distribution of rapid testing solutions. This facilitates widespread access to reliable diagnostic tools, supporting national efforts to improve HIV detection and treatment.
HIV Diagnostics Market Value Chain Analysis

HIV Diagnostics Market Companies
- Alere Inc
- Abbott
- Bristol-Myers Squibb Company
- Janssen Global Services, LLC
- Gilead Sciences, Inc.
- Merck & Co. Inc.
- VIIV Healthcare
- BD
- Beckman Coulter, Inc.
- Sysmex Europe SE
- Apogee Flow Systems
- Point Care
- Siemens Healthcare GmbH
- QIAGEN
- Zyomtronix, Inc.
- Mylan N.V.
- F. Hoffmann-La Roche Ltd
Recent Developments
- In December 2025, the Wisconsin Department of Health Services launched a new initiative. This initiative aims at expanding access to HIV and STI testing in the United States of America.(Source: https://www.wbay.com)
- In December 2025, the Department of Health (DOH) Cordillera partnered with the Baguio City Health Services Office. This partnership aims at launching a drive-through ‘Park and Test' initiative that allows on-the-go HIV testing. (Source: https://new.baguio.gov.ph)
- In October 2025, the AIDS Healthcare Foundation (AHF) opened a new HIV/AIDS specialty facility in Tennessee, U.S. This new center was inaugurated to deliver free STI/STD prevention and treatment services in the U.S. (Source: https://www.aidshealth.org)
Segments Covered in the Report
By Product Type
- Tests Kits
- Instruments
- Software
By Test Type
- Antibody Tests
- Antigen Tests
- Nucleic Acid Tests
By Technology
- ELISA
- Rapid Testing
- Polymerase Chain Reaction
By End User
- Hospitals
- Diagnostic Laboratories
- Home Care Settings
By Region
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
