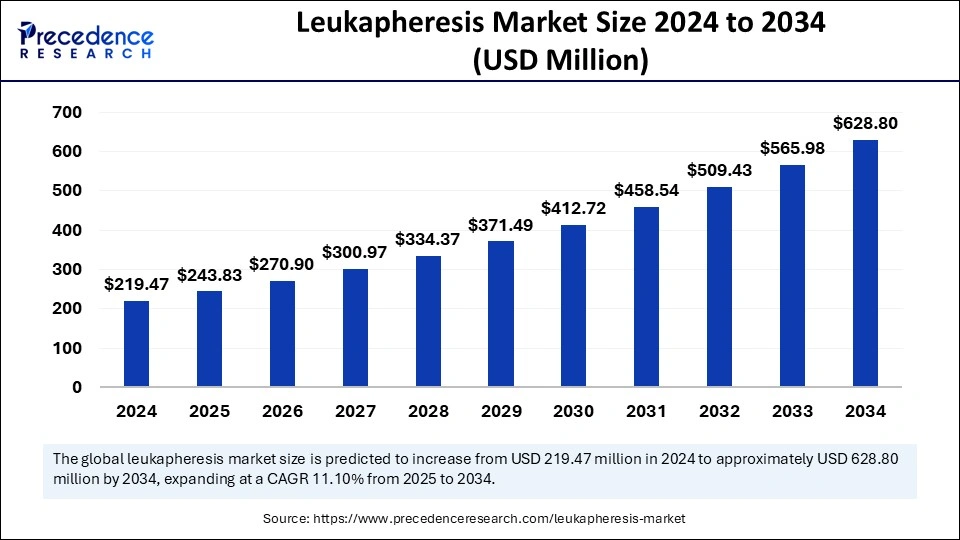
What is the Leukapheresis Market Size?
The global leukapheresis market size accounted for USD 243.83 million in 2025 and is predicted to increase from USD 270.90 million in 2026 to approximately USD 687.44 billion by 2035, expanding at a CAGR of 10.92% from 2026 to 2035. The rising incidence of blood disorders across the globe is the key factor driving the growth of the market. Also, technological innovations in leukapheresis procedures, coupled with the increasing need for personalized medicine, can fuel market growth further.
Leukapheresis Market Key Takeaways
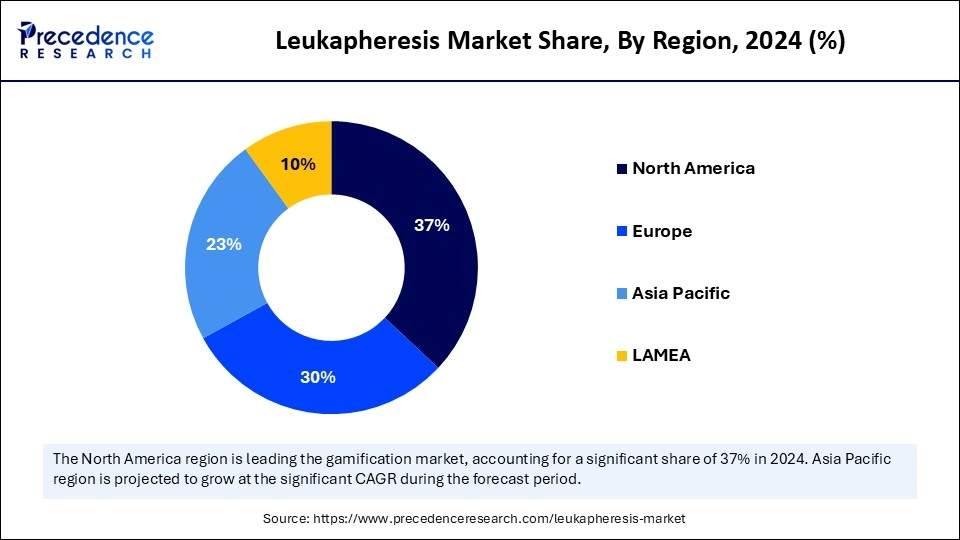
- North America dominated the leukapheresis market with the largest share of 37% in 2025.
- Asia Pacific is expected to show the fastest CAGR of 12.4% over the projected period.
- By product type, the leukapheresis devices segment held the biggest market share of 70% in 2025.
- By product type, the leukapheresis disposables segment is projected to grow at a solid CAGR of 11.3% during the forecast period.
- By application, the research application segment contributed the highest market share of 78% in 2025.
- By application, the therapeutic application segment is expanding at the fastest CAGR of 12% over the forecast period.
- By end use, the hospitals and clinics segment generated the major market share of 65% in 2025.
- By end use, the pharmaceutical and biotechnology companies segment is growing at a double digit CAGR of 12.3% during the projected period.
Market Overview
Leukapheresis is a procedure that includes the separation and collection of WBCs from an individual's blood. It is generally performed utilizing catheter devices that use filtration or centrifugation techniques to remove or isolate the white blood cells. Leukapheresis is mainly utilized around oncology and hematology for different purposes, such as the collection of WBCS for research purposes and the treatment of several cancers and blood disorders.
Applications of Artificial Intelligence (AI) in CAR-T Cell Therapy
Artificial Intelligence plays an essential role in optimizing the production of CAR-T cells, and enhancing the efficacy and quality of these therapies for treating lymphoma and leukemia can help to find proper patients for CAR-T cell therapy to forecast treatment responses and monitor therapy progression. Furthermore, AI-powered systems can automate different aspects of leukapheresis procedures, decreasing the need for manual intervention and enhancing overall efficiency.
Leukapheresis Market Growth Factors
- The growing investments by major players in various research activities are expected to boost market growth shortly.
- The ongoing collaboration between manufacturers and healthcare providers can soon propel market growth.
- The growing emphasis on outpatient techniques will likely contribute to market expansion.
Market Scope
| Report Coverage | Details |
| Market Size by 2035 | USD 687.44Billion |
| Market Size in 2025 | USD 243.83Billion |
| Market Size in 2026 | USD 270.90 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 10.92% |
| Dominated Region | North America |
| Fastest Growing Market | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Application, End-use, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America and Middle East & Africa |
Market Dynamics
Drivers
Increasing demand for stem cell transplantation
Leukapheresis plays a crucial part in stem cell transplantation procedures. As the need and demand for stem cell -based therapies rise to treat various diseases such as blood disorders, the leukapheresis market will witness significant expansion. In addition, favorable reimbursement policies by private and government insurance companies for leukapheresis procedures have facilitated their adoption.
- In March 2025, Zydus Lifesciences launched ANVIMO (Letermovir), a new and innovative treatment for the prevention of Cytomegalovirus (CMV) infection in hematopoietic stem cell transplant (HSCT) and kidney transplant patients.CMV infection remains one of the most serious complications in transplant patients, often leading to graft failure, decreased survival, and prolonged hospital stays.
Restraint
Risk of complications
Therapeutic leukapheresis comes with many complexities, such as reduced white blood cell (WBC) count, hypocalcemia, adverse reactions to anticoagulants , and potential risks of platelet loss. These side effects can cause dizziness and severe pain at the venipuncture site among some patients, which limits the procedure's usage and acceptance.
Opportunity
Growing demand for leukopaks in oncology research
The most substantial factor creating opportunities in the leukapheresis market is the growing demand for leukoplakias diagnostic and clinical research. These are products of leukapheresis, rich in different types of immune cells like monocytes, lymphocytes, and dendritic cells, and are used extensively in immunotherapy, oncology, and cell biology research. Furthermore, leukopaks are very important in an extensive range of research and therapeutic applications.
- In November 2024, QPS Holdings, LLC announced that their Springfield campus, QPS Missouri, has launched a new Cell Therapy business unit. The unit's first achievement is the opening of a new Leukopak collection and blood product processing center. This facility is designed to meet the exponentially growing demand for blood products to support cell and gene therapy work.
Segment Insights
Product Type Insights
The leukapheresis devices segment dominated the leukapheresis market in 2025. The dominance of the segment can be attributed to the increasing incidence of blood disorders like lymphoma and leukemia across the globe. The segment is sub-segmented into membrane filtration-based devices and centrifugation-based devices. The growing prevalence of chronic conditions and autoimmune diseases necessitating therapeutic apheresis is propelling the demand for membrane filtration-based devices.
The leukapheresis disposables segment is expected to grow the fastest during the forecast period. The growth of the segment can be credited to the surge in awareness regarding the blood component separation procedures along with the improved procedure volume. With the increase in the individual population with hematologic conditions and solid tumors, the number of procedures is also rising. Additionally, innovations in medical research and increased funding in healthcare technology impact segment growth positively.
- In April 2023, Fresenius Kabi announced the availability of a single-needle venous access option for the Amicus Extracorporeal Photopheresis (ECP) System at the 49th annual meeting of the European Society for Blood and Marrow Transplantation (EBMT). Fresenius Kabi is a global healthcare company that specializes in medicines and technologies for infusion, transfusion, and clinical nutrition.
Application Insights
The research application segment held the largest leukapheresis market share in 2025. The dominance of the segment can be linked to the growing inclination toward the development and launch of novel therapies for conditions such as autoimmune disorders and cancer. Moreover, this research application includes studies on personalized medicine, CAR T-cell therapy, and new leukoplakia collection methods. Such research applications are anticipated to boost the growth in demand for leukapheresis procedures.
The therapeutic application segment is expected to grow at the fastest rate over the forecast period. The growth of the segment can be driven by the rising adoption of personalized medicine and cell-based therapy approaches. This segment covers numerous disease areas where therapeutic interventions are used, such as autoimmune diseases, cancer, hematological disorders, etc.
End-Use Insights
In 2025, the hospitals and clinics segment dominated the market by holding the largest leukapheresis market share. The dominance of the segment is due to the wide range of roles of this segment in healthcare, including therapeutic procedures, direct patient care, and diagnostic services. Furthermore, the rising incidence of blood-related conditions and the increasing need for efficient treatment protocols are fueling the demand for leukapheresis products in hospitals.
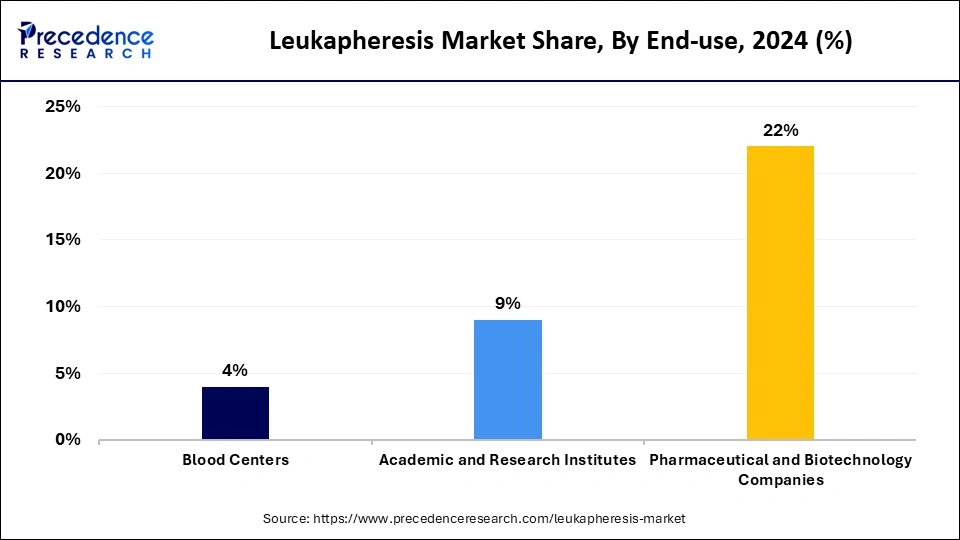
The pharmaceutical and biotechnology companies segment is estimated to show the fastest growth during the projected period. The growth of the segment is owing to the growing emphasis on cell-based therapies and personalized medicine across all research facilities. In addition, these companies play an essential role in the market as they use leukapheresis products for drug development and research. Major pharmaceutical and biotechnology companies in the market are Terumo BCT, Macopharma, Asahi Kasei Medical, Fresenius, and Miltenyi Biotec.
Regional Insights
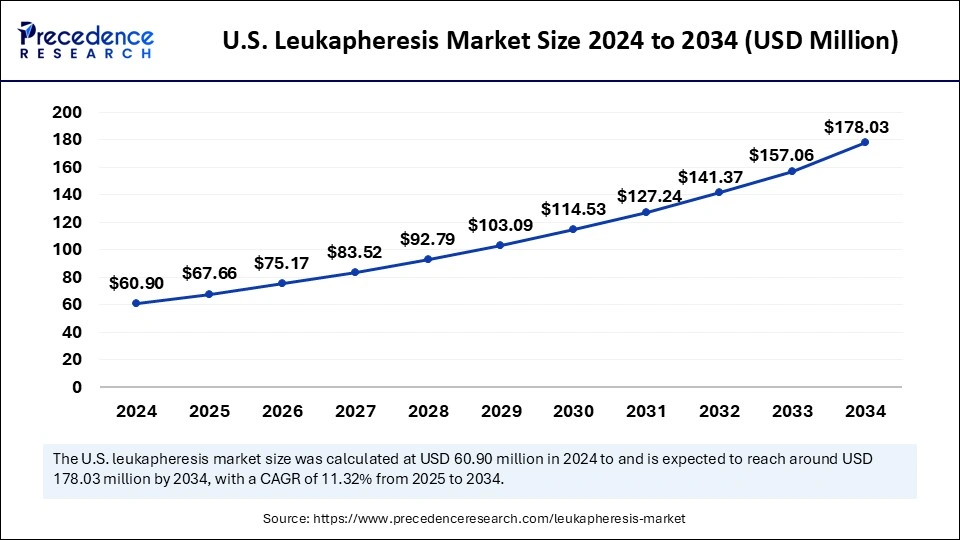
What is the U.S. Leukapheresis Market Size?
The U.S. leukapheresis market size was exhibited at USD 67.66 million in 2025 and is projected to be worth around USD 195.48 million by 2035, growing at a CAGR of 11.19% from 2026 to 2035.
North America held the largest leukapheresis market share in 2025. The dominance of the region can be attributed to the rising incidence of diseases such as autoimmune disorders and cancer. The region also has a strong healthcare infrastructure and high healthcare spending. Moreover, enhancements in healthcare technology have further impacted positive regional expansion.
In North America, the U.S. led the leukapheresis market owing to the ongoing and rapid collaboration between pharmaceutical firms, research institutions, and healthcare providers. Also, the demand for leukapheresis is increasing in the U.S. due to growing incidence rates of autoimmune diseases and blood cancers.
- In August 2023, the FDA approved the Reveos Automated Whole Blood Processing System by Terumo Blood and Cell Technologies. This system improves the efficiency of processing whole blood into white blood cells and other components, supporting efforts to increase the U.S. blood supply.
Asia Pacific is expected to show the fastest growth in the leukapheresis market over the projected period. The growth of the region can be credited to the rising incidence of autoimmune diseases and blood disorders in the developing countries in the region. Furthermore, emerging nations such as China, Japan, and India are going to witness exponential growth in medical infrastructures due to a surge in investment and a growing incidence of chronic diseases.
In Asia Pacific, China dominated the leukapheresis market in 2025. The dominance of the country can be driven by growing investments in innovative medical technologies coupled with the surge in healthcare infrastructure. There is also an exponential surge in the number of individuals undergoing stem cell transplantation procedures.
Role of Leukapheresis in CAR-T Cell Therapy Manufacturing in Europe
Europe shows a significant growth during the forecast period. It includes the separation and collection of a patient's white blood cells, mainly lymphocytes, to be engineered into therapeutic CAR-T cells in European countries. It allows high-yield harvesting of T cells, which are then shipped to production facilities to manufacture the final treatment.
Germany Leukapheresis Market Trends
The rising prevalence of leukemia and a few blood cancers, mainly among the aging population in France, is driving the demand for therapeutic leukapheresis. Hospitals are adopting advanced, automated closed-system, as well as continuous-flow apheresis devices that offer enhanced cell separation accuracy and thus, increased donor safety or comfort.
Single-Use Kits and Consumables in Leukapheresis Procedures in Latin America
Latin America shows a notable growth during the forecast period. It is primarily to ensure patient safety, comply with strict infection control regulations, and maintain the high quality of collected blood components. As the region sees a rise in advanced therapies, such as CAR T-cell therapy, the demand for sterile, disposable tubing sets and collection bags has risen to remove cross-contamination risks and guarantee procedural consistency.
Brazil Leukapheresis Market Trends
Brazil's market is driven by the surge in cellular as well as gene therapy research and treatments demands high-quality leukopaks, driving the need for specialized collection, a trend seen globally. There is a growing shift towards automated apheresis machines that enhance procedural efficiency and improve patient safety during white blood cell separation.
Advancements in Automated Leukapheresis Systems in MEA
MEA shows a rapid growth during the forecast period. It is driven by a rapid modernization of healthcare infrastructure, the rising prevalence of hematological disorders, and the increasing adoption of specialized, personalized therapies such as CAR-T. These systems are replacing manual, time-intensive methods to improve procedure efficiency, enhance patient safety, and increase cell yield for, among other things, stem cell transplantation.
Leukapheresis Market Companies

- StemExpress, LLC.: StemExpress, LLC offers both Research Use Only (RUO) and GMP-compliant products, funding the entire pipeline from drug development to clinical trials and commercialization.
- Haemonetics Corporation: Haemonetics Corporation offers comprehensive technologies for the leukapheresis market, aiming for automated, closed-system cell collection and processing.
- Caltag Medsystems Limited: Caltag Medsystems Limited offers a range of high-quality human cellular starting materials, specifically aiming at leukapheresis products to support research along with development in cell and gene therapy (CGT).
- Lonza Group AG: Lonza Group AG provides a comprehensive portfolio for the leukapheresis market, aiming for both high-quality cellular starting materials for research and, thus, GMP-compliant products for clinical or commercial cell therapy manufacturing.
Other Major Key Players
- Charles River Laboratories International, Inc.
- Adacyte Therapeutics
- AllCells, LLC
- Asahi Kasei Medical
- Haemonetics Corporation
- Macopharma
- Cerus Corporation
- SB-Kawasumi Laboratories, Inc.
Latest Announcements by Market Leaders
- In October 2024, Cerus Corporation announced updates on the INTERCEPT red blood cell (RBC) programs in the U.S. and Europe: In the U.S., Cerus has entered into a new six-year agreement with the Biomedical Advanced Research and Development Authority (BARDA), part of the U.S. Department of Health and Human Services Administration for Strategic Preparedness and Response, to further advance the development of the INTERCEPT RBC system.
Recent Developments
- In March 2024, the FDA approved Bristol Myers Squibb's Breyanzi, an innovative CAR T cell therapy for adult patients with specific types of leukemia and lymphoma. The treatment process begins with leukapheresis, where patients' white blood cells are collected to create the therapy.
- In May 2023, Akadeum Life Sciences launched a new product line for cell therapy research, advancing its BACS Microbubble technology. This innovation enables the straightforward isolation of T cells and PBMCs from leukapheresis materials, eliminating the need for traditional lysis or centrifugation steps.
- In June 2023, Hemanext Inc., a leading innovator in blood processing, storage, and transfusion technology, announced a new partnership with Vitalant, one of the nation's largest nonprofit blood and biotherapies healthcare organizations.
Segments Covered in the Report
By Product
- Leukapheresis Devices
- Centrifugal Devices
- Membrane Separators
- Leukapheresis Disposables
By Application
- Research Application
- Cancer Research
- Immunology Research
- Others
- Therapeutics Application
- Hematologic Disorders
- Autoimmune Diseases
- Others
By End-use
- Blood Centers
- Academic and Research Institutes
- Pharmaceutical and Biotechnology Companies
- Hospitals and Clinics
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
