Neurology Clinical Trials Market Size and Forecast 2025 to 2034
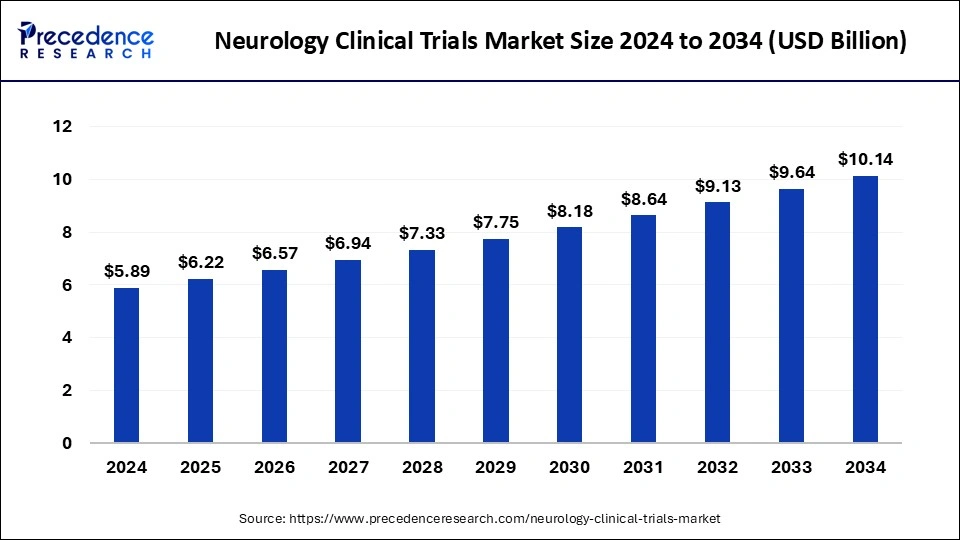
The global neurology clinical trials market size was estimated at USD 5.89 billion in 2024 and is predicted to increase from USD 6.22 billion in 2025 to approximately USD 10.14 billion by 2034, expanding at a CAGR of 5.58% from 2025 to 2034. The benefits of neurology clinical trials include access to the latest therapies, reduced cost, a chance to help others, closer monitoring, and careful and regular monitoring from the research team that includes healthcare professionals, doctors, and others. These factors help to the growth of the market.
Neurology Clinical Trials Market Key Takeaways
- The global neurology clinical trials market was valued at USD 5.89 billion in 2024.
- It is projected to reach USD 10.14 billion by 2034.
- The neurology clinical trials market is expected to grow at a CAGR of 5.58% from 2025 to 2034.
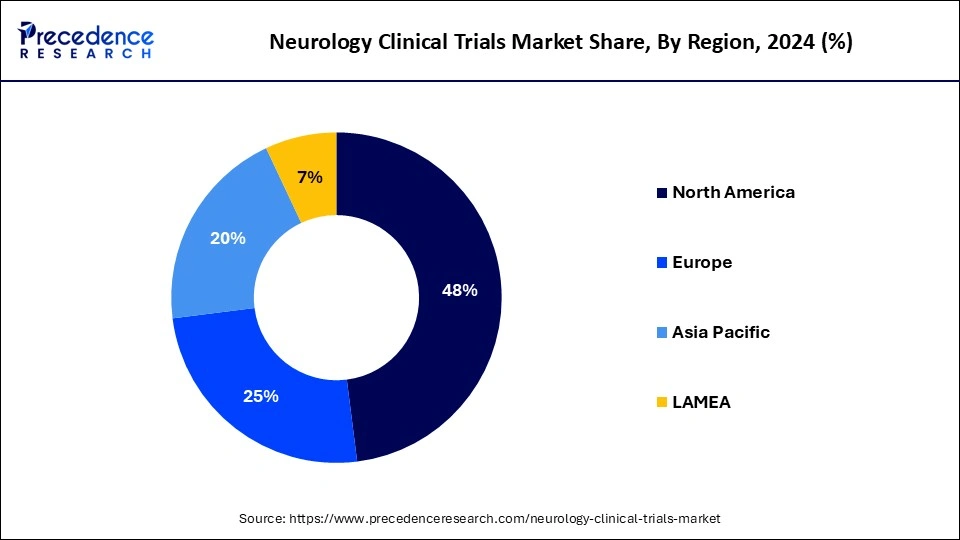
- North America dominated the neurology clinical trials market with the largest revenue share of 48% in 2024.
- Asia Pacific is estimated to grow at a solid CAGR of 5.93% during the forecast period of 2025-2034.
- By phase, the phase II segment has generated more than 47% of revenue share in 2024.
- By phase, the phase III segment is projected to expand at a CAGR of 5.62% during the forecast period.
- By indication, the epilepsy segment has recorded more than 23% of revenue share in 2024.
- By indication, the huntington's disease segment growing at a CAGR of 6.04% during the forecast period.
- By study design, the interventional segment dominated the market with the biggest revenue share of 96% in 2024.
- By study design, the observational segment is expected to grow at a CAGR of 5.83% during the forecast period.
U.S. Neurology Clinical Trials Market Size and Growth 2025 to 2034
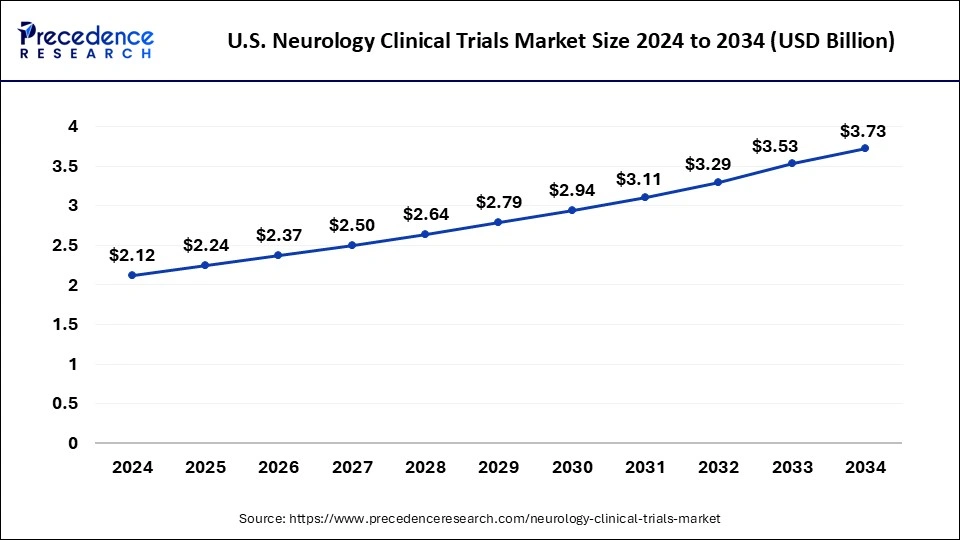
The U.S. neurology clinical trials market size was exhibited at USD 2.12 billion in 2024 and is projected to be worth around USD 3.73 billion by 2034, poised to grow at a CAGR of 5.81% from 2025 to 2034.
North America dominated the neurology clinical trials market in 2024. The presence of a high number of players in neurological clinical trials and the increasing prevalence of neurological diseases leads to the growth of the market in the North American region. In the United States, from 1990-2017, many people suffered from neurological disorders like stroke, TTH, SCI, migraine, AD, and other dementias, which helped the growth of the market. To improve brain health, the government of Canada helps with research.
- In April 2023, in Canada, there are more than 1000 brain disorders and diseases like brain cancer, epilepsy, Parkinson's disease, Alzheimer's disease, etc. The government of Canada is aware of the necessity of supporting the neuroscience community and its role in improving the brain health of Canadians.
- In February 2024, a phase I/ phase II clinical trial investigating an allogenic neural exosome AB126 derived from neural stem cells was launched by a biotechnology company, Aruna Bio (GA US), for the treatment of neurogenerative diseases.
Asia Pacific is estimated to be the fastest-growing during the forecast period of 2024-2033. Researchers are actively developing new treatments for neurological disorders. In the Asia Pacific region, neurological disorders like migraine, stroke, and other dementias are high, which leads to the growth of the neurology clinical trials market. In India, 1 % population out of the total population suffers from the prevalence of epilepsy, which leads to the growth of the market. In January 2024, the Siemens Healthineers-CDS (computational data science) collaborative lab for AI (artificial intelligence) in precision medicine at IISc Bengaluru was launched by the Indian Institutes of Science (IISc) and Siemens Healthineers.
Market Overview
Neurology clinical trials are research studies that examine behavioral, medical, or surgical interventions in people related to disorders or diseases that are linked with brain or cognitive behavior. The neurology clinical trials market deals with pharmaceutical & biotechnology companies, clinical research laboratories, contract research organizations, etc., for the researchers to determine whether the prevention or treatment of new drugs, medical devices, or diet is effective and safe to use for people. Neurology clinical trials are research studies that involve volunteer participants. These human research studies help physician-scientists to better understand, treat, diagnose, and prevent conditions and diseases. These factors help to the growth of the market.
Neurology Clinical Trials Market Growth Factors
- The neurology clinical trial can provide necessary information related to new treatments and also help to enhance the quality of care for people who suffer from neurological conditions.
- The human research studies help scientists or physicians to better understand, treat, diagnose, and prevent conditions and diseases.
- It helps to provide more information about diseases or conditions, which helps the growth of the market.
- It also helps to get information about resources and support groups. These factors help to the growth of the neurology clinical trials market.
Market Scope
| Report Coverage | Details |
| Market Size by 2034 | USD 10.14 Billion |
| Market Size in 2025 | USD 6.22 Billion |
| Growth Rate from 2025 to 2034 | CAGR of 5.58% |
| Largest Market | North America |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Phase, Study Design, Indication, Study Design, Phase, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Driver
Increasing prevalence of neurological diseases
Neurological disorders are types of nervous system disorders that affect neurons and the brain. The causes of the increasing prevalence of neurological disorders include exposure to pollutants, stroke, infections, genetic disorders, changing lifestyles, and the aging population. The increasing number of neurological diseases like stroke, traumatic brain injury, muscle regeneration, Parkinson's disease, etc. These factors help to the growth of the neurology clinical trials market.
Restraint
Limitations of neurology clinical trials
Limitations of neurology clinical trials include regulatory complexities governing trial data and protocols reporting, retention & recruitment of suitable patient populations, and high costs linked with trial design and implementation. Limited understanding of the basic biology of disease, understanding of how nosology can constrain innovation, insufficient sharing of expertise, data, and knowledge, clinical trials high failure rates, clinical trials across therapeutic areas high costs, lack of clarity on regulatory requirements, etc. These barriers can restrict the growth of the neurology clinical trials market.
Opportunity
Future scope of neurology clinical trials
Opportunities to enhance neurology clinical trials include understanding the basic biology of neurological diseases, obtaining all essential evidence across all phases of drug development, developing new endpoints, tools, and statistical approaches to enhance the effectiveness of clinical studies, implementing operational changes to neurology clinical trials, arranging innovative research programs, collaborating across countries and disciplines, making neurology clinical trials more patient-centric, and meeting regulatory requirements. These factors help the neurology clinical trials market grow.
Phase Insights
The phase II segment dominated the market in 2024. Phase II plays an important role in neurology clinical trials. It allows close monitoring of a relatively homogeneous patient population, involves participants selected by using narrow criteria, normally randomized and controlled studies, and evaluates the efficacy and safety of a drug for a particular condition. Phase II of the clinical trial helps in drug development by bridging the gap between safety assessments in Phase I and efficacy evaluations in Phase III. Phase II is also helpful for the understanding of new treatments' therapeutic potential and obtaining detailed data on safety, effectiveness, and dosage profiles. These factors help the growth of the phase II segment and contribute to the growth of the neurology clinical trials market.
The phase III segment is estimated to be the fastest-growing during the forecast period. Phase III plays an important role in neurology clinical trials. It is generally the last phase of testing before drug approval. It generally involves a large number of participants, assesses safety and efficiency information simultaneously, monitors side effects, confirms treatment effectiveness, and also compares new treatments with standard or reference treatments. These factors help the growth of the phase III segment and contribute to the neurology clinical trials market.
Indication Insights
The epilepsy segment dominated the neurology clinical trials market in 2024. Neurology clinical trials play an important role in epilepsy disease, which is a neurological disease. The causes of epilepsy include abnormal activities in the brain that can affect any area of the brain, increasing disorders in the brain like autism, injury or damage to the brain at the time of birth, brain infectious diseases like meningitis, medical conditions like brain stroke or tumor that can affect the brain, trauma or injury to the head, etc. To overcome this disease, the development of new tests and procedures like neurological examination, neuropsychological examination, functional MRI, electroencephalogram, etc., is essential, which leads to neurological clinical trials. According to WHO (World Health Organization), nearly 50 million people suffer from epilepsy, and around 80% of them live in low- or middle-income countries. These factors help to the growth of the epilepsy segment and contribute to the growth of the market.
- In March 2022, a new study to evaluate the effect of lifestyle interventions like cognitive behavioral therapy, yoga, and music therapy to control epileptic seizures was launched by the Cleveland Clinic.
The Huntington's disease segment is estimated to be the fastest-growing during the forecast period. Huntington's disease is caused by a gene defect inherited from parents. Huntington's disease causes disorders like psychiatric, movement, and cognitive disorders with symptoms highly different between individuals. To overcome this disease and its disorders, there is a need for new therapies, medications, prevention methods, genetic testing, etc., which leads to neurology clinical trials. Past epidemiological studies on Huntington's Disease have shown a collective incidence of 0.38 per 100000 person-years and a global prevalence of 2.71 per 100000 person-years. These factors help the growth of the Huntington's disease segment and contribute to the growth of the neurology clinical trials market.
- In August 2023, for the treatment of adults with chorea-linked with Huntington's Disease (HD), INGREZZA (valbenazine) capsules were approved by the U.S. FDA, announced by the Neurocrine Biosciences, Inc.
Study Design Insights
The interventional segment dominated the neurology clinical trials market in 2024. Interventional study design plays an important role in neurology clinical trials. The benefits of interventional study design include eliminating cofounding towards creating groups that are comparable for all factors that hold known, unknown, outcome, or difficult to measure, eliminating selection bias, any guideline differences that exist between study groups are accountable to chance rather than bias, it also gives statistical tests validity based on probability theory, etc.
An increased number of interventional studies on CNS (central nervous system) conditions conducted worldwide leads to the growth of the market. These factors help to the growth of the interventional segment and contribute to the growth of the market.
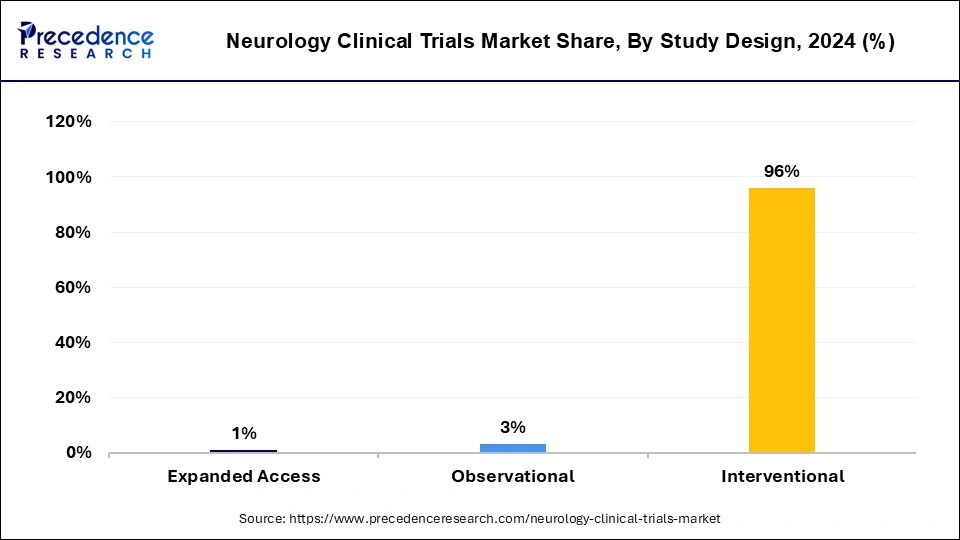
The observational segment is estimated to be the fastest-growing during the forecast period. The benefits of observational study design include providing information about complicated topics in an efficient manner and with low cost, enabling the study of topics that may not be randomized ethically, safely, or efficiently, may normally be completed inexpensively and quickly, enabling change to be recorded, more easily designed and completed than randomized clinical trials (RCTs), straightforward to conduct, etc. These factors help the growth of the observational segment and contribute to the growth of the neurology clinical trials market.
Neurology Clinical Trials Market Companies
- IQVIA
- Biogen
- Aurora Healthcare
- GlaxoSmithKline Plc.
- Icon Plc.
- Syneous Health
- Charles River Laboratories
- Med pace
- Covance
- Novartis AG
- Sanofi
- Merck & Co., Inc.
- AbbVie Inc.
- Teva Pharmaceutical Industries Ltd.
- Annovis Bio
- Athira Pharma, Inc.
- Zydus Group
- Eli Lilly and Company
- Eisai Co., Ltd.
- AstraZeneca
- Supernus Pharmaceuticals, Inc. (Adamas Pharmaceuticals)
Recent Developments
- In August 2023, the phase II RECOVER-NEURO clinical trial study to evaluate the combination of REMOTE-transcranial direct current stimulation (tDCS) and a brain training program for long covid was launched by Soterix Medical.
- In March 2024, an innovative and new pTau217 blood test, ALZpath Dx, was launched by a specialized clinical laboratory, Neurocode USA, Inc., that offers world-class testing solutions for neurological disorders. This new test may be used in the monitoring, screening, and diagnosis of Alzheimer's disease. In the US, Neurocode is the first laboratory to make this test as LDT (laboratory-developed test) for clinical trials, clinical diagnostics use, and other research causes.
Segments Covered in the Report
By Phase
- Phase I
- Phase II
- Phase III
- Phase IV
By Study Design
- Interventional
- Observational
- Expanded Access
By Indication
- Epilepsy
- Parkinson's Disease (PD)
- Huntington's Disease
- Stroke
- Traumatic Brain Injury (TBI)
- Amyotrophic Lateral Sclerosis (ALS)
- Muscle Regeneration
- Others
By Study Design
- Epilepsy
- Interventional
- Observational
- Expanded Access
- Parkinson's Disease (PD)
- Interventional
- Observational
- Expanded Access
- Huntington's Disease
- Interventional
- Observational
- Expanded Access
- Stroke
- Interventional
- Observational
- Expanded Access
- Traumatic Brain Injury (TBI)
- Interventional
- Observational
- Expanded Access
- Amyotrophic Lateral Sclerosis (ALS)
- Interventional
- Observational
- Expanded Access
- Muscle Regeneration
- Interventional
- Observational
- Expanded Access
- Others
- Interventional
- Observational
- Expanded Access
by Phase
- Epilepsy
- Phase I
- Phase II
- Phase III
- Phase IV
- Parkinson's Disease (PD)
- Phase I
- Phase II
- Phase III
- Phase IV
- Huntington's Disease
- Phase I
- Phase II
- Phase III
- Phase IV
- Stroke
- Phase I
- Phase II
- Phase III
- Phase IV
- Traumatic Brain Injury (TBI)
- Phase I
- Phase II
- Phase III
- Phase IV
- Amyotrophic Lateral Sclerosis (ALS)
- Phase I
- Phase II
- Phase III
- Phase IV
- Muscle Regeneration
- Phase I
- Phase II
- Phase III
- Phase IV
- Others
- Phase I
- Phase II
- Phase III
- Phase IV
By Geography
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
