What is the Transdermal Skin Patches Market Size in 2026?
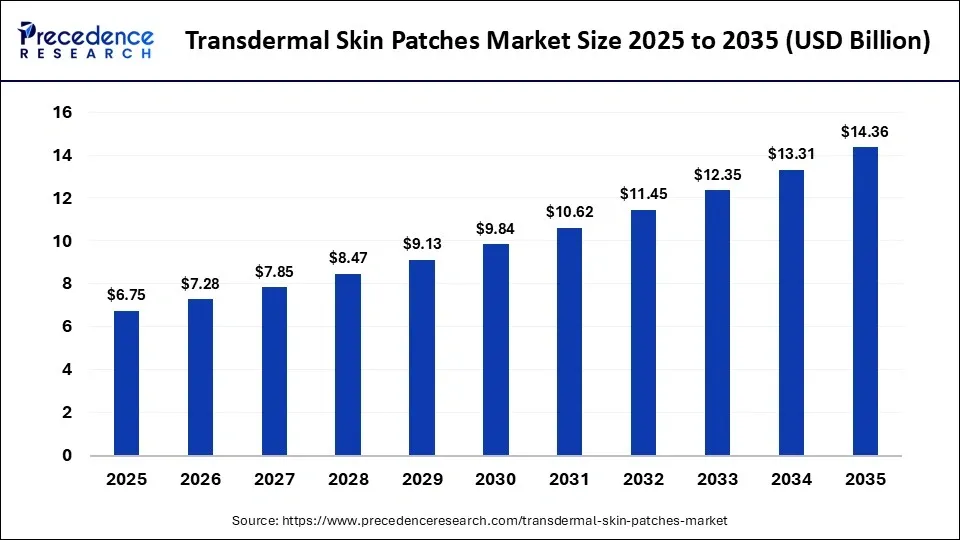
The global transdermal skin patches market size was calculated at USD 6.75 billion in 2025 and is predicted to increase from USD 7.28 billion in 2026 to approximately USD 14.36 billion by 2035, expanding at a CAGR of 7.84% from 2026 to 2035. The market is driven by growing preference for non-invasive drug delivery and improved patient compliance.
Key Takeaways
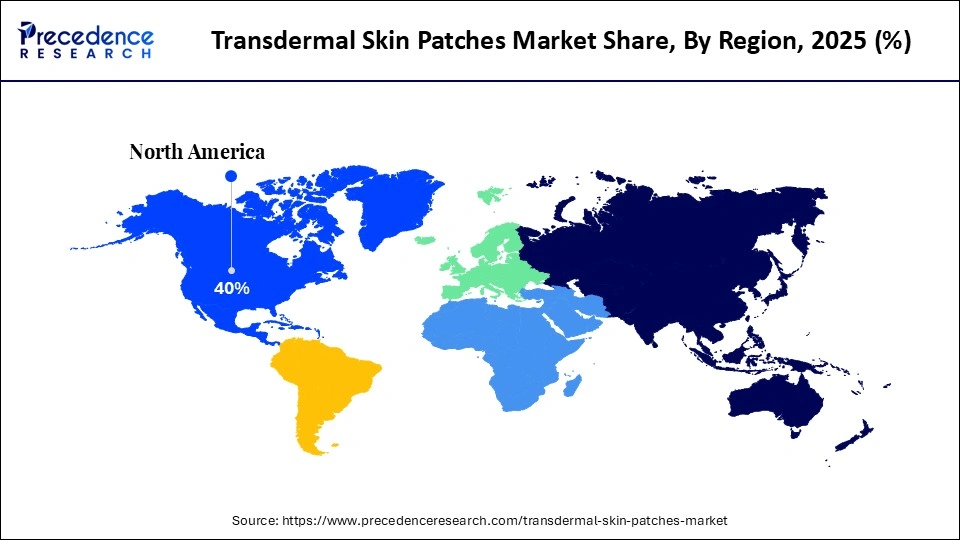
- North America accounted for the largest share of 40% in 2025.
- The Asia Pacific is expected to witness the fastest growth during the forecasted years.
- By type, the multi-layer drug-in-adhesive segment held a significant market share of 38% in 2025.
- By type, the matrix segment is expected to grow at a significant CAGR in the market over the forecast period.
- By application, the pain relief segment held a significant share of 25% in 2025.
- By application, the smoking reduction and cessation aid segment is anticipated to show considerable growth in the market over the forecast period.
- By distribution channel, the hospital pharmacies segment contributed the highest market share of 41% in 2025.
- By distribution channel, the online pharmacies segment is anticipated to grow at the fastest rate in the market over the forecast period.
Market Overview
The transdermal skin patches market revolves around the production, application, and use of drug-containing adhesive patches that deliver medication through the skin into the bloodstream over a controlled period. These patches present an alternative to either oral or injectable drug intake, and provide sustained release, constant therapeutic levels, and are far more convenient to the patients. They are commonly used for pain management, hormone therapy, smoking cessation, and neurological and cardiovascular treatments. The market is growing as a result of the rising prevalence of chronic illnesses, which drives the need for long-term medication. Technological innovations in patch design, such as improved permeability, smart features, and advanced adhesives, are also fueling market growth.
How is AI Integration Influencing the Transdermal Skin Patches Market?
The transdermal skin patches market is being transformed by artificial intelligence (AI), enabling more precise drug delivery, product development, and patient monitoring. AI-based modeling and simulation optimize drug formulations, skin permeability, and release profiles, supporting the development of more effective and personalized therapies. Additionally, AI facilitates intelligent patches equipped with sensors that track physiological indicators, such as body temperature, glucose levels, and medication adherence, in real time. In manufacturing, AI enhances quality control by detecting defects, improving production efficiency, and ensuring product consistency.
Transdermal Skin Patches Market Trends
- Advanced transdermal technologies are improving controlled drug release, skin penetration, dosing accuracy, and therapeutic efficacy, while reducing side effects and enhancing treatment outcomes for chronic disease management.
- The increase in the prevalence of chronic diseases like diabetes, cardiovascular diseases, and neurological conditions is creating the need for long-term drug delivery solutions such as transdermal patches in hospitals and home care.
- Innovations in patch design, such as enhanced adhesives, microneedle integration, and smart monitoring, boost drug absorption, enable personalized therapies, and allow real-time tracking of treatment outcomes, improving patient compliance and clinical effectiveness.
- Growing focus on patient comfort and self-administration is increasing demand for user-friendly transdermal systems that simplify medication regimens, reduce dosing frequency, minimize gastrointestinal side effects, and support adherence across diverse patient groups.
- Rising pharmaceutical R&D investments in new transdermal formulations are expanding therapeutic applications, improving drug stability and bioavailability, and accelerating the commercialization of innovative patch-based therapies.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 6.75 Billion |
| Market Size in 2026 | USD 7.28 Billion |
| Market Size by 2035 | USD 14.36 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 7.84% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Type, Application, Distribution Channel, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Type Insights
Why Did the Multi-Layer Drug-In-Adhesive Transdermal Patches Segment Lead the Market?
The multi-layer drug-in-adhesive segment led the transdermal skin patches market, holding the largest share in 2025. This is because of its superior drug delivery, controlled release capabilities, and broad therapeutic applications. These patches are sophisticated systems that feature several adhesive layers with the active pharmaceutical ingredient, enabling even and prolonged diffusion of medication through the skin. This type of design prevents the variation in the delivery of drugs within the bloodstream, and it also reduces the danger of dose dumping and variability that is usually seen in oral therapies. They are more convenient, patients adhere more, and do not require such a frequent dose, which positively affects the effectiveness of treatment.
The matrix segment is expected to grow at a significant CAGR over the forecast period because of its cost-effective production, versatile formulation development, and expanding therapeutic applications. In matrix systems, the drug is uniformly distributed within a polymer matrix, allowing predictable and consistent release through passive diffusion. Compared to multi-layer systems, matrix patches are more economical to produce, making them suitable for large-scale manufacturing and wider market access. They are increasingly used in areas such as smoking cessation, hormone therapy, and chronic disease management.
Application Insights
Why Did the Pain Relief Segment Contribute the Most Revenue in the Market?
The pain relief segment held the largest share of the transdermal skin patches market in 2025. The segment's dominance is attributed to the increasing prevalence of chronic pain conditions around the world, which significantly creates the need for long-term and effective remedies for managing these conditions. Arthritis, musculoskeletal injuries, nerve-related pain, and lower back conditions are disorders affecting millions of people that usually need constant medication. Transdermal patches offer a regulated and steady delivery of analgesics and maintain uniform therapeutic concentrations with no peaks and troughs. These patches are also very useful to elderly patients and long-term treatment patients because of their non-invasive nature.
The smoking reduction and cessation aid segment is expected to grow at a significant CAGR in the coming years, owing to the rising global awareness of the health risks of tobacco use and the need for effective quitting solutions. Public health campaigns, regulatory measures, and educational efforts have highlighted smoking-related diseases such as respiratory disorders, cardiovascular complications, and cancer. Nicotine patches, a clinically proven and non-invasive option, help manage withdrawal symptoms and reduce cravings through controlled nicotine delivery. Growing initiatives by governments and health organizations to curb smoking are fueling strong demand for transdermal cessation therapies, supporting rapid segment growth.
Distribution Channel Insights
Why Did the Hospital Pharmacies Segment Lead the Transdermal Skin Patches Market?
The hospital pharmacies segment led the market while holding the largest share in 2025, owing to the high volume of patients, high level of clinical trust, and integrated delivery of healthcare services. Hospitals serve as primary care centers for conditions requiring prescription therapies, including pain management, hormonal disorders, and chronic disease treatment. Patients often receive medications directly through hospital pharmacies following consultations, surgeries, or inpatient care, ensuring reliable distribution. Professional supervision also enhances treatment safety, adherence, and compliance, particularly for first-time users.
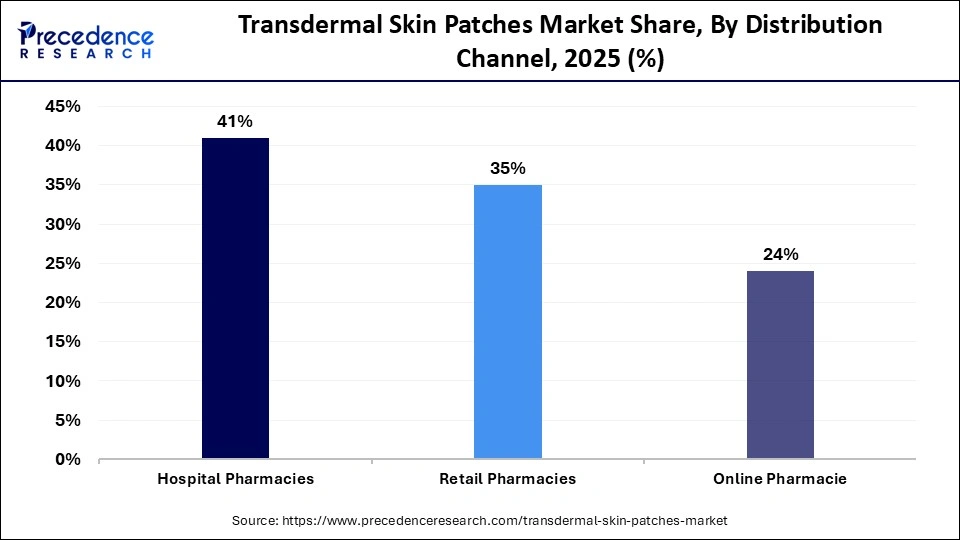
The online pharmacies segment is expected to grow at the fastest CAGR over the forecast period, driven by increasing digital adoption, expanding e-commerce infrastructure, and demand for convenient access to medications. Patients with chronic conditions are increasingly using digital platforms to order prescriptions from home, reducing the need for frequent pharmacy visits. Online channels are especially valuable for individuals in remote or underserved areas with limited medical facilities. Competitive pricing, home delivery, and subscription-based medication programs further enhance convenience, adherence, and patient compliance.
Regional Insights
North America Transdermal Skin Patches Market Size and Growth 2026 to 2035
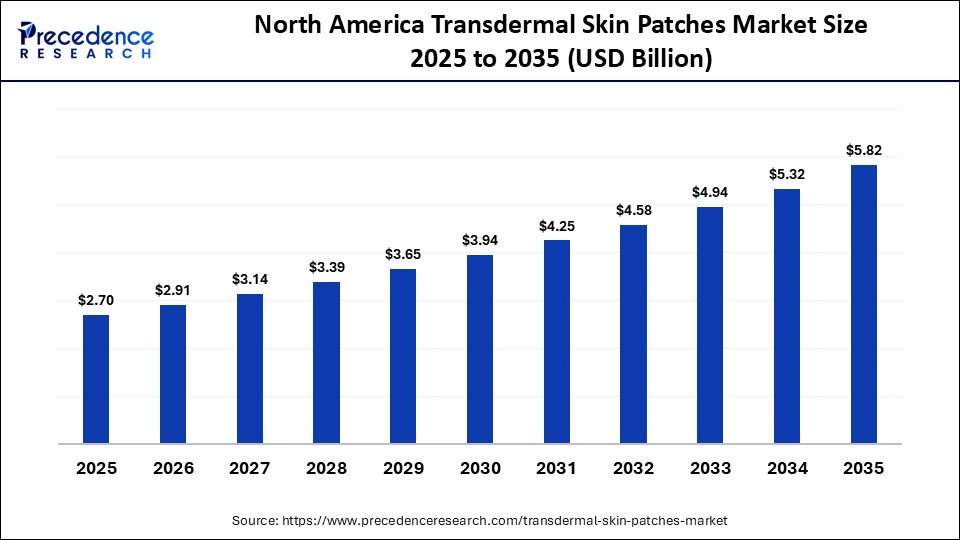
The North America transdermal skin patches market size is estimated at USD 2.70 billion in 2025 and is projected to reach approximately USD 5.82 billion by 2035, with a 7.98% CAGR from 2026 to 2035.
What Made North America the Leading Region in the Transdermal Skin Patches Market?
North America led the transdermal skin patches market by holding the largest share in 2025 because of its well-developed healthcare system, ongoing pharmaceutical innovation, and high acceptance of new drug delivery systems. The area enjoys an established legal regulatory framework that facilitates quick approval and commercialization of transdermal therapies and rapid entry and availability of new pharmaceutical products. Rising prevalence of chronic disorders, including pain, cardiovascular diseases, and hormonal imbalances, has driven demand for long-term, controlled drug delivery solutions. Additionally, the presence of large pharmaceutical and biotechnology firms investing in R&D is accelerating product innovation and commercialization.
U.S. Transdermal Skin Patches Market Size and Growth 2026 to 2035
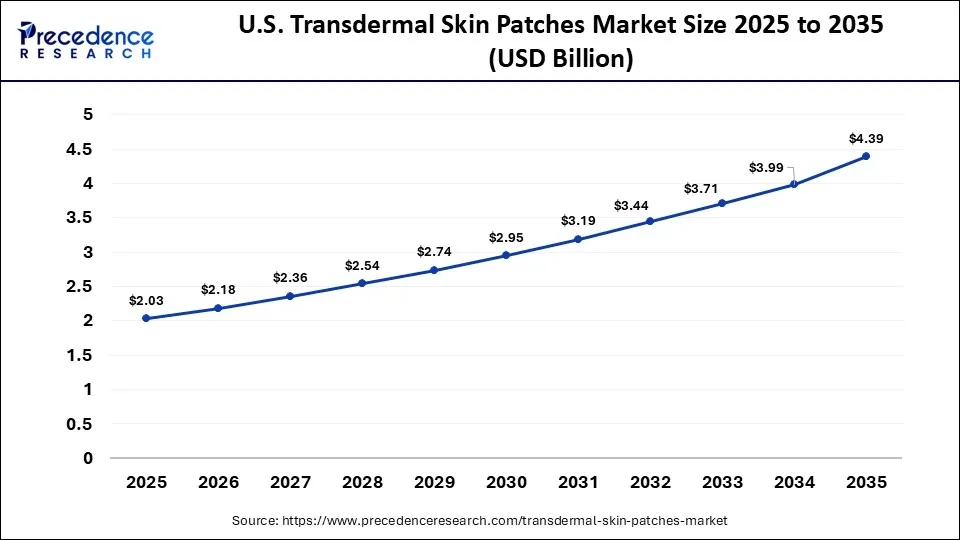
The U.S. transdermal skin patches market size is calculated at USD 2.03 billion in 2025 and is expected to reach nearly USD 4.39 billion in 2035, accelerating at a strong CAGR of 8.02% between 2026 and 2035.
U.S. Market Analysis
The U.S. transdermal skin patches market is growing due to the rising prevalence of chronic diseases, such as cardiovascular disorders, diabetes, and pain-related conditions, which increase demand for long-term, controlled drug delivery solutions. Strong healthcare infrastructure, advanced regulatory frameworks, and widespread adoption of innovative drug delivery technologies are accelerating market growth. Additionally, growing patient preference for convenient, non-invasive therapies and increasing investments by pharmaceutical companies in R&D are further driving market expansion.
Why is Asia Pacific Undergoing the Fastest Growth in the Transdermal Skin Patches Market?
Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period because of expanding healthcare infrastructure, growing population health demands, and the adoption of modern drug delivery methods. A large and rising population, along with increasing prevalence of chronic diseases such as diabetes, cardiovascular disorders, and pain-related conditions, is fueling demand for long-term medication solutions. Rising disposable incomes and a growing middle class are also increasing patient demand for convenient and efficient treatments. Additionally, regional government investments in healthcare modernization, pharmaceutical manufacturing, and medical research are supporting local innovation and product availability.
China Market Analysis
China is the major contributor to the Asia Pacific transdermal skin patches market due to substantial government investment in healthcare infrastructure, supportive policies promoting pharmaceutical innovation, and its position as the world's largest producer of lithium batteries, which indirectly supports medical device manufacturing. The country also has a large and growing patient population with a rising prevalence of chronic diseases, driving demand for long-term, controlled drug delivery solutions. Additionally, strong collaborations between state-owned enterprises and private companies accelerate the development and deployment of advanced transdermal therapies.
Why Is the European Transdermal Skin Patches Market Experiencing Notable Growth?
Europe is expected to grow at a notable CAGR in the foreseeable future, driven by well-developed healthcare services, rising demand for patient-friendly treatments, and ongoing technological advancements in drug delivery. The region's advanced regulatory framework ensures safety, quality, and faster adoption of high-tech pharmaceutical products. The increasing prevalence of chronic diseases, particularly among the elderly, is driving demand for long-term, convenient, and predictable medication solutions. Additionally, active collaboration between pharmaceutical companies, academic institutions, and healthcare providers is accelerating innovation in patch design and drug development.
Transdermal Skin Patches Market Companies
- Teva Pharmaceutical Industries Ltd
- Novartis AG
- Teikoku Pharma USA Inc.
- Viatris Inc.
- Johnson & Johnson
- Luye Pharma Group
- Purdue Pharma Manufacturing LP
- Henan Lingrui Pharmaceutical Ltd
- Samyang Biopharmaceuticals Corp.
- Hisamitsu Pharmaceutical Co, Inc.
Recent Developments
- In April 2025, Luye Pharma Group launched Rotigotine Luye, a rotigotine transdermal patch, in the UK. Indicated for early- and advanced-stage Parkinson's disease and moderate-to-severe restless legs syndrome in adults, it is the first generic UK patch demonstrating bioequivalence to Neupro.(Source: https://www.luye.cn )
- In March 2025, the U.S. Food and Drug Administration approved Aveva Drug Delivery Systems for their generic of ZTlido (0.18% lidocaine patch) in treating post-herpetic neuralgia. The non-hydrogel patch is a non-opioid patch and results in up to 12 hours of sustained specific nerve pain relief.(Source: https://firstwordpharma.com )
- In October 2024, Tonik announced the launch of transdermal patches formulated with homeopathic recipes. These patches are intended for sleep and stress relief.(Source: https://www.nutraingredients.com )
- In August 2024, Zydus Lifesciences Limited (and its affiliates) received final U.S. FDA approval to market Scopolamine Transdermal System 1 mg/3 days (Transderm Scop). This marks Zydus' fifth ANDA approval in its transdermal portfolio, leveraging its expertise in complex drug-device dosage forms. The system is indicated for preventing nausea and vomiting caused by anesthesia, narcotic pain medications, surgery, and motion sickness.
Segments Covered in the Report
By Type
- Single-layer Drug-in-Adhesive
- Multi-layer Drug-in-Adhesive
- Matrix
- Other Types
By Application
- Pain Relief
- Smoking Reduction and Cessation Aid
- Cardiovascular Disorders
- Neurological Disorders
- Hormonal Therapy
- Other Applications
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting