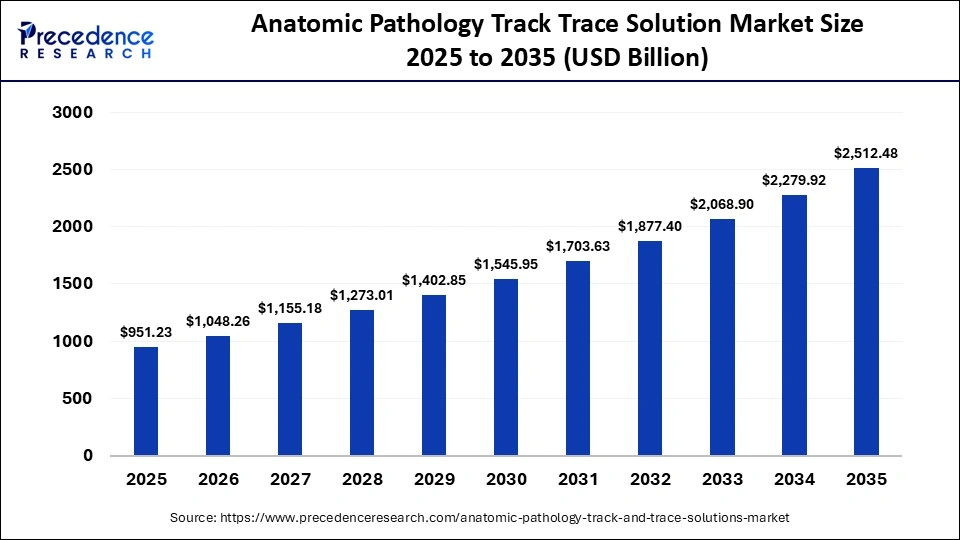
What is the Anatomic Pathology Track Trace Solution Market Size in 2026?
The global anatomic pathology track trace solution market size was calculated at USD 951.23 billion in 2025 and is predicted to increase from USD 1048.26 billion in 2026 to approximately USD 2,512.48 billion by 2035, expanding at a CAGR of 10.20% from 2026 to 2035.Key market drivers include rapid technological innovations, increasing adoption of advanced healthcare technologies, continued digitalization, and rising healthcare awareness.
Key Takeaways
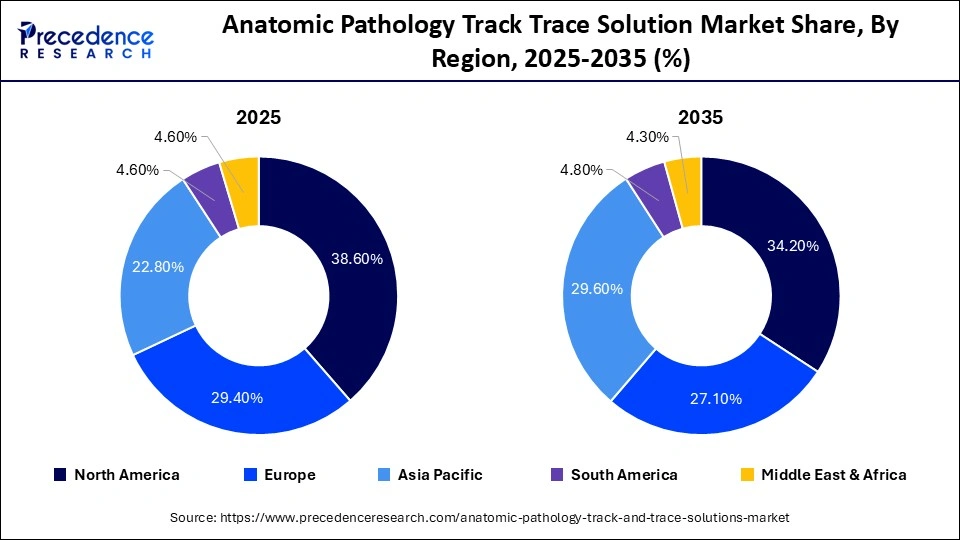
- North America led the anatomic pathology track trace solution market in 2025 with approximately 38.6% share.
- Asia-Pacific is expected to be the fastest-growing region in the forecasted period with about 13.4% CAGR.
- By component, the software segment held a dominant position in the market with a share of 42.7% in 2025.
- By component, the consumables segment is expected to grow at the fastest CAGR of 12.8% in the market between 2026 and 2035.
- By technology, the barcode tracking segment registered its dominance over the global market with a share of 68.4% in 2025.
- By technology, the RFID tracking segment is expected to expand rapidly in the market with a CAGR of 14.1% in the coming years.
- By application, the specimen tracking segment dominated the global market with a share of 36.8% in 2025.
- By application, the chain of custody and compliance tracking segment is expected to grow with the highest CAGR of 13.7% in the market during the studied years.
- By end user, the hospital laboratories segment held the largest revenue share of 47.5% in the market in 2025.
- By end user, the specialty cancer and diagnostic centers segment is expected to witness the fastest growth in the market with a CAGR of 14.4% over the forecast period.
Anatomic Pathology Track Trace Solution Market Overview
Anatomic pathology track trace solution industry comprises software, hardware, consumables, and integrated workflow systems used to identify, label, track, verify, and document pathology specimens, cassettes, blocks, slides, and related assets across accessioning, grossing, processing, microtomy, staining, archiving, and reporting. These solutions improve specimen integrity, chain of custody, patient safety, workflow efficiency, regulatory compliance, and error reduction in hospital laboratories, reference laboratories, academic centers, and specialized pathology networks adopting barcode, RFID, connectivity, and digital workflow automation platforms for visibility and analytics.
Impact of Artificial Intelligence on the Anatomic Pathology Track Trace Solution
Artificial Intelligence (AI) shows considerable potential in anatomic pathology by enhancing diagnostic precision through AI based image analysis, virtual staining, and generating synthetic data. AI also boosts workflow efficiency by facilitating the automation of routine tasks, quality control, and reflex testing.
AI-powered software can derive essential insights from high content imaging, leading to more informed decisions in drug discovery. Furthermore, robotic systems can manage sample preparation, labeling, and scanning. By examining extensive datasets, AI can predict disease progression and foresee how patients might react to therapies. AI aids in the organization, documentation, and long-term viability of pathological work.
Anatomic Pathology Track Trace Solution Market Trends
- Automated Wax Embedding System: New automated wax embedding modules support standardized histopathology workflow by managing paraffin block preparation efficiently, while enabling operators to focus on accurate tissue orientation in pathology processes.
- Integration of LIMS and Tracking Software: Integration of a laboratory information management system (LIMS) with pathology tracking software improves data security, sample traceability, and workflow efficiency in handling sensitive pathology information.
- Sustainable Track and Trace Solution: Increasing demand for environmentally responsible solutions is encouraging the development of track and trace systems with reduced plastic use, energy efficient operations, and recyclable materials in laboratory settings.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 951.23 Billion |
| Market Size in 2026 | USD 1048.26 Billion |
| Market Size by 2035 | USD 2,512.48 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 10.20% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Component, Technology,Application,End User ,and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Adoption of Identification and Tracking Systems
Growing use of printers, labelling systems, barcode scanners, RFID readers, and mobile computing systems supports enhanced sample tracking driven by the need for diagnostic accuracy and operational efficiency in pathology labs.
Constraint
Interoperability Challenges
Interoperability remains a key challenge, as compatibility issues between systems can increase costs, cause data inaccuracies, and disrupt workflow, limiting seamless adoption of advanced tracking solutions.
Opportunity
Rising Diagnostic Demand and Digitization
Increasing disease incidence, wider use of the biopsy technique, and ongoing digitization in pathology are creating growth opportunities for advanced track and trace and laboratory management solutions.
Segmental Insights
Component Insights
Anatomic Pathology Track Trace Solution Market Share, By Component , 2025-2035 (%)
| Component | 2025 | 2035 |
| Software | 42.7% | 39.2% |
| Hardware | 34.8% | 31.4% |
| Consumables | 22.5% | 29.4% |
The Software Segment Dominated the Market with 42.7% of Market Share in 2025
The software segment dominated the anatomic pathology track trace solution market with a share of 42.7% in 2025, driven by its ability to reduce turnaround times, eliminate bottlenecks, and deliver consistently accurate results, thereby improving patient care. These solutions address the diverse needs of all diagnostic disciplines, including anatomic pathology, clinical laboratories, molecular diagnostics, genomics, and toxicology testing.Software platforms form the backbone of complex pathology laboratories, supporting more efficient and standardized operations. Epic Beaker, Oracle Cerner PathNet, and Sunquest PowerPath are key examples of advanced tools used in leading labs. The broad spectrum of available solutions, from end-to-end LIMS platforms to EHR-integrated systems, makes identifying the right fit critical for enhancing laboratory efficiency and overall patient care.
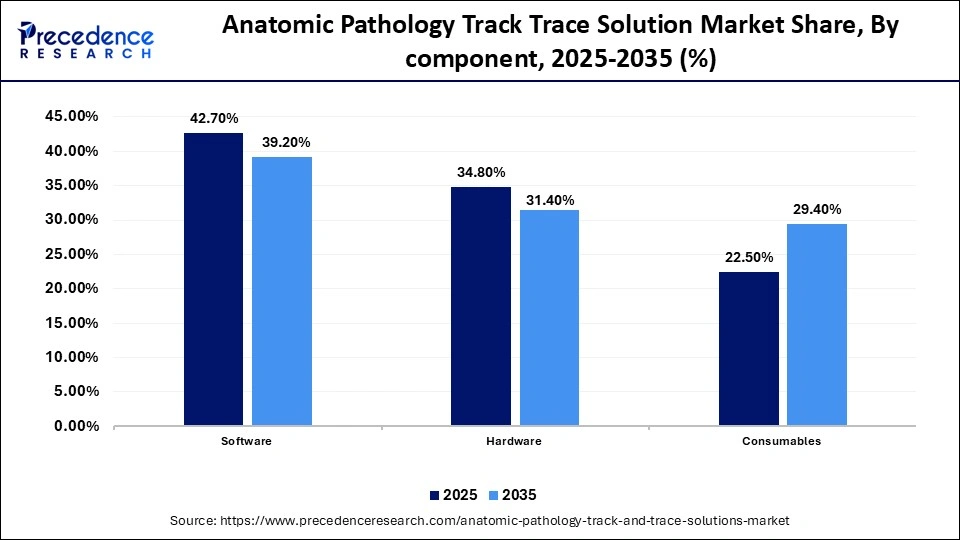
The consumables segment is expected to show the fastest growth with a CAGR of 12.8% over the forecast period, driven by rising demand for high quality and customized products in pathological testing. These consumables support multiple steps of the workflow, from formalin handling using vacuum sealed bags and buckets to all in one tissue processing and embedding, and a wide range of options to meet diverse sectioning requirements. They are designed to meet the highest quality standards, ensuring seamless integration, consistent performance, and reliability across routine and specialized anatomic pathology workflows.
Technology Insights
Anatomic Pathology Track Trace Solution Market Share, By Technology , 2025-2035 (%)
| Technology | 2025 | 2035 |
| Barcode Tracking | 68.4% | 54.6% |
| RFID Tracking | 12.6% | 19.8% |
| Mobile and Cloud Enabled Tracking | 19.0% | 25.6% |
The Barcode Tracking Segment Led the Market With 68.4% of Market Share in 2025
The barcode tracking segment led the anatomic pathology track trace solution market with a share of 68.4% in 2025, as barcode tracking enables surgical pathology practices to rely on computer readable information for accurate and consistent specimen management. Barcode systems can capture and manage data across the workflow and help histology standardize its processes.
By encoding the identification of specimen parts at accession, grossing, histology, and pathology sign-out, barcode tracking supports standardized and automated work processes. It remains the preferred choice for many users due to its efficiency, ease of implementation, and relatively lower cost compared with alternative technologies.
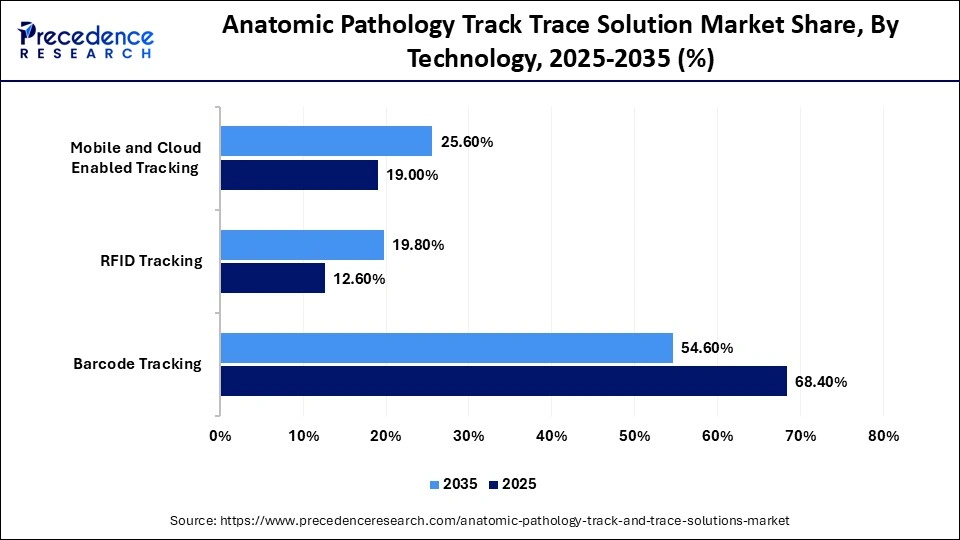
The RFID tracking segment is expected to gain the highest market share with a CAGR of 14.1% between 2026 and 2035. It helps reduce specimen identification and track errors through continuous, automated specimen monitoring. Key advantages of RFID technology include automated reading, reduced labor requirements, faster reading rates, the ability to electronically write data to tags, greater resistance to harsh chemicals and processing environments, no line-of-sight requirement, and higher data capacity. Together, these benefits provide pathology laboratories with a pathway from reactive crisis management to proactive specimen control, thereby supporting operational efficiency.
Application Insights
Anatomic Pathology Track Trace Solution Market Share, By Application , 2025-2035 (%)
| Application | 2025 | 2035 |
| Hospital Laboratories | 47.5% | 43.2% |
| Independent and Reference Laboratories | 24.8% | 23.6% |
| Academic and Research Institutes | 14.6% | 12.4% |
The Specimen Tracking Segment Held the Largest Market Share of 36.8% in 2025
The specimen tracking segment held the largest revenue share of 36.8% in the anatomic pathology track trace solution market in 2025, as it eliminates the manual tracking of specimens, slides, and blocks, allowing technologists to dedicate more time to developing and refining their histologic techniques. By minimizing dependence on paper logs, phone calls, and labor intensive searches, specimen tracking creates additional capacity for technical training and quality enhancement.
Specimen tracking solutions are designed to be highly user-friendly, incorporating universal icons and straightforward text to facilitate adoption among various laboratory teams. These systems empower laboratories to transition towards a more streamlined, automated workflow while enhancing patient safety through precise sample identification at every phase of the laboratory process. Furthermore, this technology contributes to improved ergonomics at the workbench by simplifying and optimizing daily tasks.
The chain of custody and compliance tracking segment is expected to witness the fastest growth in the market with a CAGR of 13.7% over the forecast period. In laboratory environments, the chain of custody entails documenting the handling, storage, and transfer of samples or data throughout their lifecycle. This includes tracking items involved, timestamps, transfer purposes, storage conditions, testing procedures, security measures against tampering, and authorized personnel signatures.
Such meticulous documentation is essential in regulated fields like pharmaceuticals, clinical research, forensics, and biobanking, as any traceability gaps can lead to inaccuracies, regulatory issues, or invalidated studies. Growing regulatory demands and the need for audit readiness are increasing the need for effective chain of custody and compliance tracking solutions.
End User Insights
Anatomic Pathology Track Trace Solution Market Share, By End User, 2025-2035 (%)
| End User | 2025 | 2035 |
| Specimen Tracking | 36.8% | 31.2% |
| Tissue Cassette and Block Tracking | 24.7% | 22.1% |
| Slide Tracking | 22.6% | 21.4% |
| Chain of Custody and Compliance Tracking | 15.9% | 25.3% |
The Hospital Laboratories Segment Dominated the Market with 47.5% of Market Share in 2025
The hospital laboratories segment accounted for the highest revenue share of 47.5% in the anatomic pathology track trace solution market in 2025, as hospital laboratories play a critical role in patient care, requiring highly reliable tracking systems to manage specimens collected across diverse diagnostic tests.
As demand for precise and timely diagnostics continues to rise, the importance of efficient specimen tracking solutions in this setting is growing. These solutions integrate a range of technologies and systems to ensure end-to-end traceability of specimens, samples, and associated data throughout the entire laboratory workflow, supporting both clinical accuracy and operational efficiency.
The specialty cancer and diagnostic centers segment is expected to expand rapidly in the market with a CAGR of 14.4% in the coming years. Rising global cancer burden and the adoption of advanced diagnostic technologies are driving the need for robust track and trace systems in these centers. Such systems enhance the timeliness and accuracy of diagnosis and treatment, supporting more effective cancer care delivery, even in health systems that already have established cancer screening programs.
Regional Insights
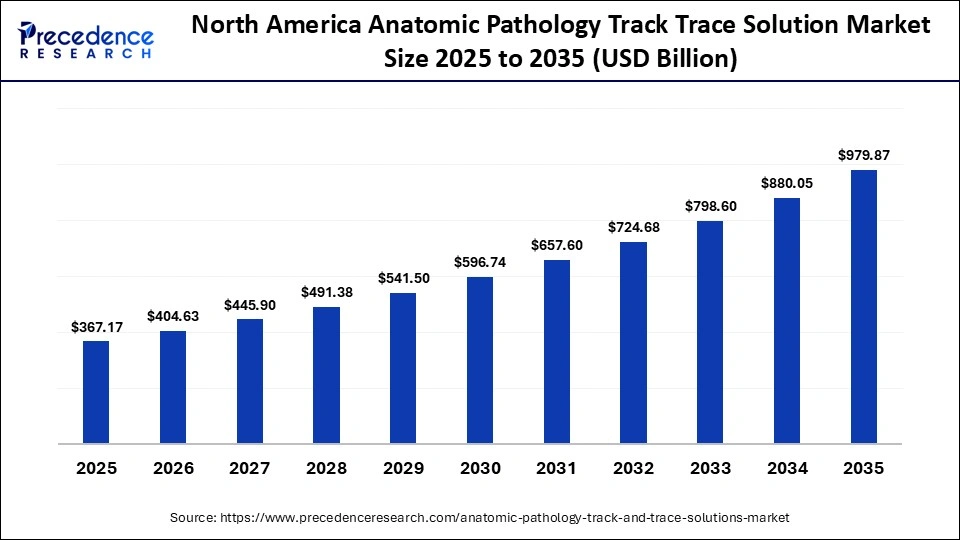
North America Anatomic Pathology Track Trace Solution Market Size and Growth 2026 to 2035
The North America anatomic pathology track trace solution market size is estimated at USD 367.17 billion in 2025 and is projected to reach approximately USD 979.87 billion by 2035, with a 10.31% CAGR from 2026 to 2035.
Why North America Dominated the Anatomic Pathology Track Trace Solution Market?
North America held a major market share of 38.6% in 2025, due to the increased number of diagnostic tests conducted in anatomic pathology labs, the prevalence of chronic diseases, which is driving up demand for high quality diagnostic services, and the presence of major market competitors in the area. The increasing prevalence of cancer across the North American population boosts the market. An estimated 2.1 million new cancer cases and 701,000 cancer deaths occur in North America each year. As the prevalence increases, diagnostic centers and healthcare facilities are adapting by investing in infrastructure.
U.S. Anatomic Pathology Track Trace Solution Market Size and Growth 2026 to 2035
The U.S. anatomic pathology track trace solution market size is calculated at USD 275.38 billion in 2025 and is expected to reach nearly USD 739.80 billion in 2035, accelerating at a strong CAGR of 10.39% between 2026 and 2035.
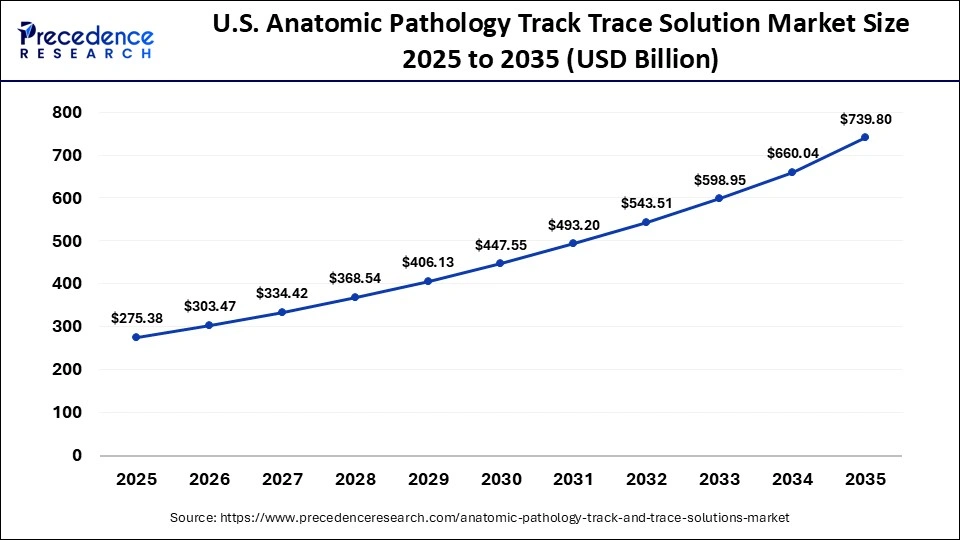
U.S. Market Trends
The U.S. leads the North American market. This leadership is driven by the high and growing burden of cancer, which has a major impact on society and underscores the need for precise and timely diagnostics. In addition, rising investment in precision medicine initiatives, focused on patient-specific treatment planning, is creating significant demand for advanced track and trace solutions. The country's advanced healthcare infrastructure, strong adoption of innovative technologies, stringent regulatory requirements for specimen tracking and management, and substantial healthcare spending further support market growth.
How is Asia-Pacific Growing in the Anatomic Pathology Track Trace Solution Market?
Asia-Pacific is expected to experience the fastest growth with a CAGR of 13.4% during the predicted timeframe. Rising demand for high-quality healthcare services, expanding healthcare infrastructure, greater awareness of diagnostic accuracy, and increasing healthcare expenditures are collectively driving the adoption of anatomic pathology track and trace solutions. The region is also benefiting from growing disposable incomes and strong government initiatives to promote advanced technologies in pathology trace and tracking, further supporting robust market growth.
China Market Trends
China's market is driven by the growing emphasis on early disease detection and preventive healthcare, which is driving demand for sophisticated diagnostic tools and robust tracking systems. National healthcare priorities increasingly align with track and trace technologies that enable earlier illness detection, better prevention strategies, and more personalized treatment, thereby accelerating adoption across Chinese pathology laboratories and cancer centers.
Anatomic Pathology Track Trace Solution Market Companies
- Leica Biosystems
- Roche Diagnostics
- Sakura Finetek
- Epredia
- PHC Holdings Corporation
- Epic Systems Corporation
- Hamamatsu Photonics
- Sectra AB
- Indica Labs
- Inspirata
- Gestalt Diagnostics
- Corista
- LigoLab
- NovoPath
- XIFIN
Recent Developments
- In March 2026, Ligolab organized a two-day event named Ligoverse 2026, which concentrated on the changing role of AI and automation in contemporary laboratory operations. The gathering focused on converting operational complexity into practical workflows, emphasizing the application of integrated laboratory informatics platforms in diagnostic settings.(Source: https://www.openpr.com)
- In September 2025, StatLab Medical Products, located in McKinney, a worldwide developer and manufacturer of pathology supplies and equipment, introduced a new product. The company revealed the global launch of its PiSmart S1 single-hopper slide printer, thereby enhancing its current PiSmart labeling and tracking solutions for both slide and cassette printers. Additionally, it is planning to expand its manufacturing operations at its facility in Arlington.(Source: https://dallasinnovates.com)
Segments Covered in the Report
By Component
- Software
- Hardware
- Consumables
By Technology
- Barcode Tracking
- RFID Tracking
- Mobile and Cloud Enabled Tracking
By Application
- Specimen Tracking
- Tissue Cassette and Block Tracking
- Slide Tracking
- Chain of Custody and Compliance Tracking
By End User
- Hospital Laboratories
- Independent and Reference Laboratories
- Academic and Research Institutes
- Specialty Cancer and Diagnostic Centers
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
