What is the Antibody Production Market Size?
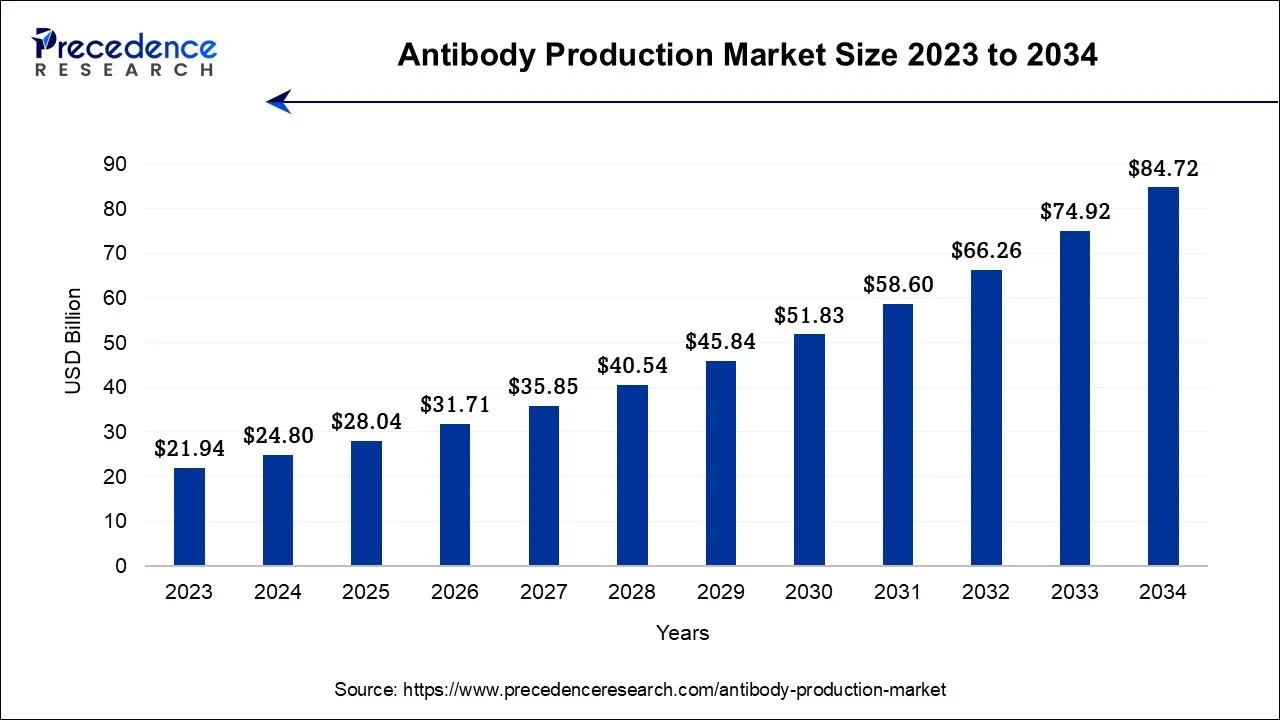
The global antibody production market size was valued at USD 28.04 billion in 2025, and is projected to be worth USD 31.71 billion by 2026, and is anticipated to reach around USD 93.76 billion by 2035, expanding at a CAGR of 12.83% over the forecast period from 2026 to 2035.
Antibody Production Market Key Takeaways
- In terms of revenue, the market is valued at $28.04 billion in 2025.
- It is projected to reach $93.76 billion by 2035.
- The market is expected to grow at a CAGR of 12.83% from 2026 to 2035.
- North America contributed more than 40% of revenue share in 2025.
- By Poduct, the consumables segment generated with the largest market share of 57% in 2025.
- By Process, the downstream segment held the highest market share of 68% in 2025.
- By Type, the monoclonal antibody segment recorded more than 65% of revenue share in 2025.
- By End-use, the pharmaceutical and biotechnology companies segment captured more than 56% of revenue share in 2025.
What is the Antibody Production?
The antibody production market refers to the commercial ecosystem encompassing the research, development, manufacturing, and distribution of antibodies for various applications, including research, diagnostics, and therapeutics. Antibodies are specialized proteins produced by the immune system in response to specific antigens, and they play a crucial role in immune defense, disease detection, and targeted treatments. The market involves a range of stakeholders, including pharmaceutical and biotechnology companies, research institutions, diagnostic companies, contract research organizations, and more, who collaborate to advance antibody-related technologies and products.
How is AI contributing to the Antibody Production Industry?
AI enhances the process of generating antibodies by speeding up the process of discovery, engineering, and production decision-making throughout the biologics development process. Neural networks are used to predict antibody structures and antigen interactions, aiding in designing efficient antibodies and optimizing binding. There are advanced algorithms in the development of sequences and avoiding the risk of aggregation. AI (when combined with automated labs and simulators) would allow accelerating the selection of candidates and simplifying the process of producing antibodies.
Antibody Production Market Growth Factors
Antibodies production is not only a central aspect of fighting infections but also a cornerstone of vaccine-induced immunity. The market growth is driven by several factors including the growing product approvals, increasing research and development and rising government initiatives. Moreover, the rising prevalence of cancer across the globe is observed to promote the growth of the global antibody production market. For instance, according to the National Institute of Health's estimation, there will be 1,958,310 cancer cases in 2024 in the United States.
The rising prevalence of cancer across the globe is observed to promote the growth of the global antibody production market. For instance, according to NIH in the United States, the estimated number of new cases in 2024 was 62,770, and the percentage of all new cancer cases was 3.1%. The estimated deaths in 2024 were 23,670, and the percentage of all cancer deaths was 3.9%.
Market Scope
| Report Coverage | Details |
| Market Size by 2035 | USD 93.76 Billion |
| Market Size in 2025 | USD 28.04 Billion |
| Market Size in 2025 | USD 31.71 Billion |
| Growth Rate from 2026 to 2035 | CAGR of 12.83% |
| Largest Market | North America |
| Fastest Growing Market | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Process, Type, End-use and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Rise in chronic disease along with an increasing elderly population
The rising chronic incidence coupled with the increasing aging population is expected to drive the market growth over the forecast period. For instance, according to the PAHO estimated that there will be 20 million new cancer instances worldwide. In the upcoming two decades, the burden of cancer will rise by approximately 60%, placing additional strain on healthcare systems, individuals, and communities. By 2040, it is anticipated that there will be roughly 30 million additional instances of cancer worldwide. Such rising cases of diseases will force the industry to boost its capability of production as well as research activities for innovation and advancements. Therefore, the aforementioned facts drive the market growth over the forecast period.
Restraint
Complexity and high cost of production
The purification of antibodies from complex mixtures can be challenging and time-consuming. High purity requirements and the need to remove impurities can lead to labor-intensive and costly processes. In addition, antibody production, especially for therapeutic purposes, can be expensive due to the need for specialized equipment, cell culture media, and purification methods. The costs associated with large-scale production can impact the accessibility of antibody-based treatments. Thus, this is expected to be a major restraining factor for the market growth over the forecast period.
Opportunity
Growing approvals for monoclonal antibodies
The increasing FDA approvals of monoclonal antibodies are expected to offer a lucrative opportunity for market growth over the forecast period. For instance, in May 2024, the Food and Drug Administation (FDA) approved EPKINLYTM, for the treatment of adult patients with diffuse large B-cell lymphoma. The rising cases of multiple communicable and non-communicable diseases also force FDA or other approving agencies and administrative bodies to boost their pace of approving monoclonal antibodies that can be further used for research and development purposes. Thus, the growing approval of monoclonal antibodies is anticipated to propel the market's growth.
Segment Insights
Product Insights
Based on the product, the global antibody production market is segmented into instruments, consumables and software. The consumables segment is expected to hold the largest market share over the forecast period. In antibody production, various consumables are used to support the growth, maintenance, and purification of cells involved in producing antibodies. These consumables are essential for creating the optimal environment for cell cultures and for facilitating the isolation and purification of the produced antibodies. Cell culture media are nutrient-rich solutions that provide the necessary nutrients, growth factors, and conditions to support the growth of cells, such as B cells or hybridomas, that produce antibodies. Thus, the growing use of consumables in the production of antibodies is expected to drive market growth during the forecast period.
Process Insights
Based on the process, the global antibody production market is segmented into upstream processing and downstream processing. The downstream segment held a significant market share in 2025 and is expected to continue the same pattern during the forecast period. The segment growth is attributed to their benefits including purity enhancement, increased specific activity, consistency and reproducibility and others.
Downstream processing allows for the removal of impurities, contaminants, and unwanted proteins that might be present in the culture medium or produced alongside the antibodies. This results in highly purified antibodies, which are essential for accurate and reliable experimental results and safe therapeutic use. Moreover, the standardized downstream processing methods contribute to consistent and reproducible antibody preparations across batches. This is vital for research and diagnostics, where consistent antibody performance is essential for reliable results. Therefore, these advantages are expected to drive the segment growth.
Type Insights
Based on the type, the global antibody production market is segmented into monoclonal antibodies and polyclonal antibodies. The monoclonal antibody segment is expected to dominate the market over the forecast period. Monoclonal antibodies are a class of antibodies that are produced by identical immune cells, all derived from a single parent cell. These antibodies are highly specific in their recognition of a single antigenic epitope, making them valuable tools in research, diagnostics, and therapeutic applications. Monoclonal antibodies are used to target specific molecules, cells, or pathogens with high precision, and they have revolutionized the fields of medicine and biotechnology.
In addition, monoclonal antibodies are used for different type of cancer and the burden of cancer rises globally, which in turn, drive the segment growth over the projected period. For instance, as per National Cancer Institutes, it is anticipated that by 2040, there would be 16.4 million cancer-related deaths and 29.5 million new cases of cancer annually.
End-use Insights
Based on the end-use, the global antibody production market is segmented into pharmaceutical and biotechnology companies, research laboratories and CROs and CDMOs. The pharmaceutical and biotechnology companies segment is expected to capture the largest market share over the forecast period. Antibody production is a crucial aspect of research and development activities in pharmaceutical and biotechnology companies.
In drug discovery, antibodies are often used to target specific proteins associated with diseases. They can serve as tools to study the function of these proteins, identify potential drug targets, and validate therapeutic concepts. Moreover, the therapeutic potential of antibodies has led to the development of numerous antibody-based drugs for treating various diseases. Thereby, driving the segment growth over the forecast period.
Regional Insights
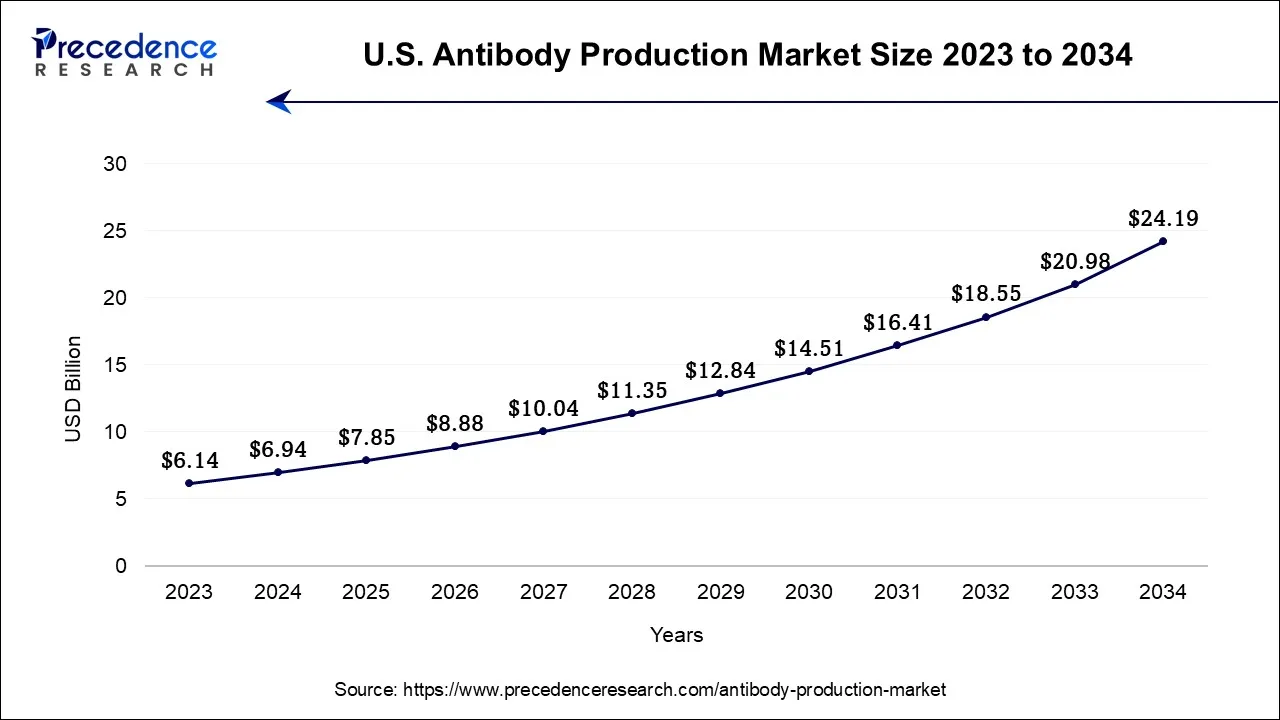
What is the U.S. Antibody Production Market Size?
The U.S. antibody production market size accounted for USD 7.85 billion in 2025 and is projected to be worth around USD 26.88 billion by 2035, poised to grow at a CAGR of 13.10% from 2026 to 2035.
North America is expected to dominate the market over the forecast period. The growth in the region is attributed to the presence of major biotechnology and pharmaceutical companies such as Pfizer, Merck, Sanofi and others. These companies are actively involved in antibody research and production where they develop monoclonal antibodies for a wide range of applications, including cancer therapy, autoimmune diseases, infectious diseases and others.
U.S. Antibody Production Market Trends
The U.S. antibody production industry is growing with intense use of artificial intelligence-based cell line engineering and modern technology in the manufacturing sector. Biopharmaceutical firms are making more sophisticated biologics, such as antibody drug conjugates and bispecific antibodies, and the continuous manufacturing platforms are becoming more flexible and scalable with respect to production.
For instance, in August 2024, Pfizer Inc. reported that the US Food and Drug Administration (FDA) has given ELREXFIOTM (elranatamab-bcmm) accelerated approval for the treatment of adult patients with relapsed or refractory multiple myeloma. The willingness to adopt advanced technologies in the biotechnology sector also promotes the growth of the market in North America. Therefore, the increasing company research and development and rising FDA approvals are expected to drive market growth over the forecast period.
On April 10, 2025, the U.S. Food and Drug Administration (FDA) announced that it will be phasing out the current requirements for animal testing in the development of monoclonal antibody therapies and other drugs, replacing them “with more effective, human-relevant methods.”
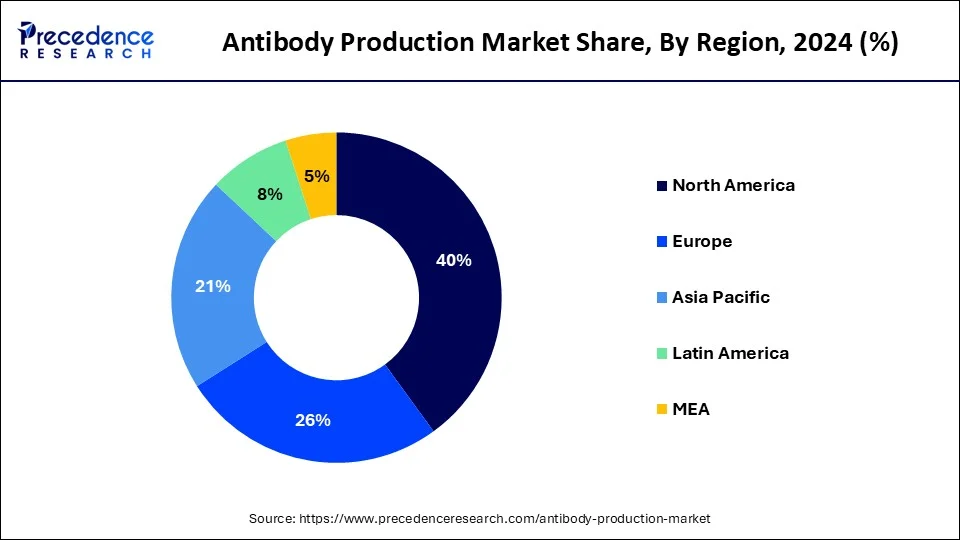
Asia Pacific is expected to witness the fastest rate of growth during the forecast period. The region's ongoing emphasis on clinical trials and drug development is observed to promote the market's growth in the upcoming years. Countries such as India, Japan, South Korea and China carry huge potential to grow in the biotechnology and pharmaceutical sectors. The presence of strict regulatory framework for the biotechnology industry in these countries also play a significant role in the market's development. The rising population, especially in countries including China and India along with the presence of huge geriatric population in Japan supplements the development of potential therapeutics in the region, this element is observed to fuel the market's growth in Asia Pacific.
China Antibody Production Market Trends
Chinas antibody manufacturing ecosystem grows based on robust in-country biotechnology innovation and quick biologics development pipelines. The biopharmaceutical firms also focus on antibody drug conjugates and bio better therapeutics and increase in manufacturing capabilities and licensing alliances that help in wider commercialization plans internationally.
In November 2024, AstraZeneca held a groundbreaking ceremony at their Singapore ADC Manufacturing Facility to commemorate the expansion of their?largest Antibody Drug Conjugate (ADC) Manufacturing Facility in Asia Pacific.? AstraZeneca has committed $1.5 billion, its largest first-stage investment to date, to establish the facility spanning 58 acres. AstraZeneca's first end-to-end Antibody Drug Conjugate (ADC) production facility will span 58 acres and will cover the complete manufacturing process for ADCs by 2029.
Antibody Production Market Companies
- QIAGEN: Offers sample technologies Nucleic acid purification kits, and automated molecular platforms to facilitate the research workflows in recombinant antibody gene cloning.
- Thermo Fisher Scientific, Inc.: Offers combined technologies such as cell culture media, expression system and purification technologies that support efficient monoclonal antibody production.
- PerkinElmer, Inc.: Provides high-throughput screening, automated liquid handling, and immunoassay solutions in therapeutic discovery and cellular analysis.
Other Major Key Players
- BGI
- Illumina, Inc.
- Agilent Technologies
- Pacific Biosciences of California, Inc.
- ProPhase Labs, Inc. (Nebula Genomics)
- Psomagen
- Azenta US, Inc.
Recent Development
- In February 2026, FUJIFILM Biotechnologies inaugurated its expanded site in Teesside, UK, today, enhancing its capabilities as a leading CDMO for biologics and advanced therapies. The expansion, funded by £400m from FUJIFILM Corporation, features the UK's largest single-use biopharmaceutical facility. (https://fujifilmbiotechnologies.fujifilm.com )
- In June 2025, Ecolab Life Sciences launched Purolite™ AP+50, an innovative affinity chromatography resin optimized for antibody manufacturing. Featuring a 50-micron bead size, it offers high dynamic binding capacity and durability. This resin uses Ecolab's patented manufacturing technology, ensuring lot consistency and shorter lead times for purification challenges. (https://cleanroomtechnology.com )
- In October 2024, MilliporeSigma, the U.S. and Canada life sciences arm of Germany's Merck KGaA, continued its march of manufacturing investments this year with plans to pump $76 million into expanding production capacity at its antibody-drug conjugate (ADC) facility in St. Louis. The expansion is expected to create 170 new jobs. Plans include upgrading 34,000 square feet of existing space, plus adding new labs, a dedicated manufacturing buffer preparation facility, cold storage infrastructure and a warehouse.
- In June 2024, South Korean CDMO Lotte Biologics and Merck KGaA signed a partnership deal focused on expanding biopharmaceutical production and process development. Under this agreement, the two companies will work together to ensure supplies of essential raw materials, establish a stable supply chain for raw materials, and create a support system for the “Bio Venture Initiative” at Lotte's Songdo Bio campus in South Korea.
- In December 2024, Chennai-based Omexa Formulary Pvt Ltd announced its plan to set up a new manufacturing unit for monoclonal antibodies and formulation products with an investment of ?95 crore near Tindivanam, Chennai.
- In January 2023, the Biologics License Application (BLA) for nirsevimab was approved by the Center for Drug Evaluation and Research of the U.S. Food and Drug Administration (FDA). Nisevimab is developed for the treatment of respiratory syncytial virus lower respiratory tract disease in newborns and infants beginning or experiencing their first RSV season.
- In May 2023, the U.S. Food and Drug Administration (FDA) approved and authorized LibrelaTM for the treatment of osteoarthritis for dogs. Librela is the first and only once-monthly anti-NGF monoclonal antibody therapy for canine OA pain, and it is both safe and effective in reducing symptoms of the condition in dogs for the long term.
Segments Covered in the Report:
By Product
- Instruments
- Consumables
- Software
By Process
- Upstream Processing
- Downstream Processing
By Type
- Monoclonal Antibody
- Polyclonal Antibody
By End-use
- Pharmaceutical and Biotechnology Companies
- Research Laboratories
- CROs and CDMOs
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
