What Bronchial Biopsy Devices Market Size?
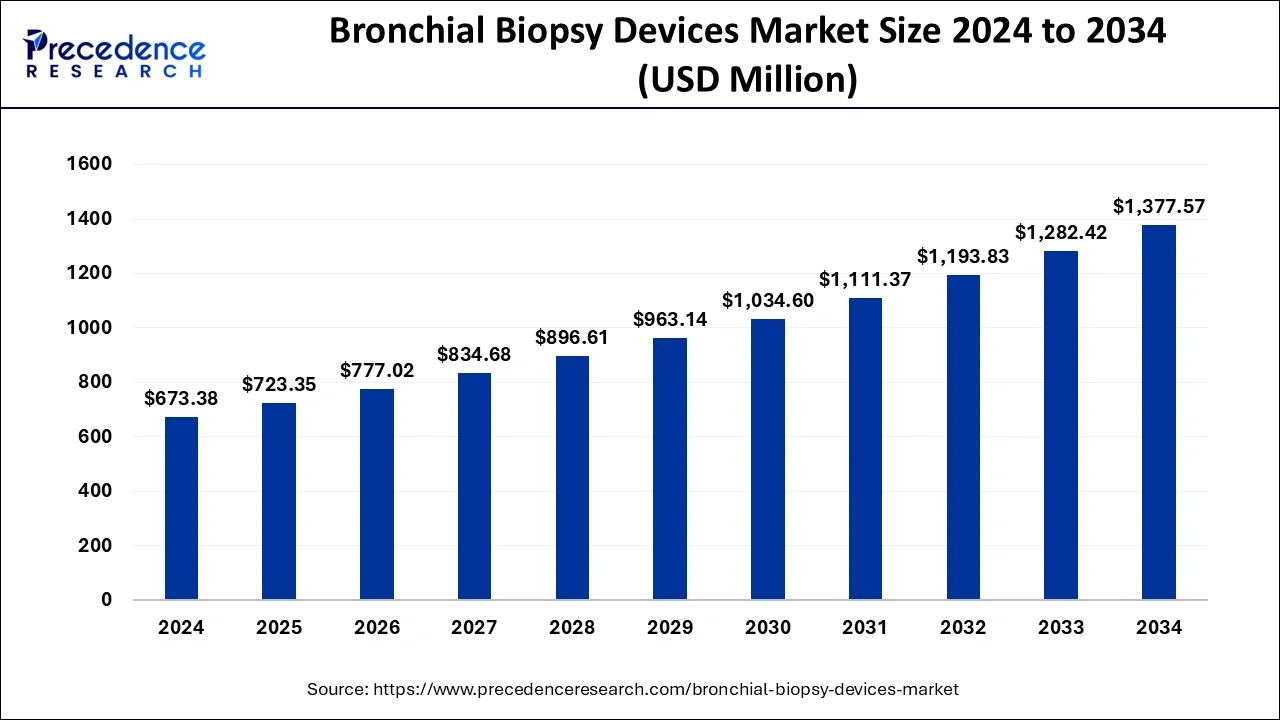
The global bronchial biopsy devices market size is calculated at USD 723.35 million in 2025 and is predicted to increase from USD 777.02 million in 2026 to approximately USD 1,468.35 million by 2035, expanding at a CAGR of 7.34% from 2026 to 2035.The bronchial biopsy devices market is growing because of the rising incidence of respiratory illnesses and lung cancer, as well as improved technologies, an increasing number of elderly, and growing awareness of people towards health, which also contributes to growth in demand.
Bronchial Biopsy Devices Market Key Takeaways
- In terms of revenue, the global bronchial biopsy devices market was valued at USD 723.35 million in 2025.
- It is projected to reach USD 1,468.35 million by 2035.
- The market is expected to grow at a CAGR of 7.34% from 2026 to 2035.
- North America dominated the global market with the largest market share of 41% in 2025.
- Asia Pacific is expected to grow at the fastest CAGR during the forecast period.
- By product, the transbronchial needle aspiration (TBNA) needles segment contributed the highest market share of 63% in 2025.
- By product, the biopsy forceps product segment is expected to grow at a notable CAGR during the forecast period.
Market Overview
A biopsy is defined as a medical operation carried out to remove tissues from an organ to determine the existence of a disease, specifically inflammatory and neoplastic diseases. Lung Biopsy is the extraction of the lung tissues to diagnose any lung disease, infection, or cancer. Increased incidence of cancer, especially the breast, prostate, and lung, coupled with increased technological development in biopsy, has propelled the bronchial biopsy devices market. This surgical procedure is mandatory for detecting some diseases, including coeliac disease, malignancies, etc, and is mainly carried out by radiologists, surgeons, and interventional cardiologists.
The constantly rising incidence of lung diseases requires new diagnostic tools. The cases of respiratory diseases such as lung cancer and interstitial lung disease are on the rise worldwide, and there is a need for prompt and accurate lung biopsies. This diagnostic procedure is necessary for determining the presence, extent, and nature of lung diseases, posing insights that are crucial for effective treatment planning.
How is AI Impacting the Bronchial Biopsy Devices Market?
The increasing accuracy and reducing tissue trauma in the bronchial biopsy devices market when AI-powered systems were used. Compared to other surgical methods, the robotic system allows precise control, which is essential, especially when the lesion is hard to locate. In addition to enhancing the physician diagnostic capabilities, the ability of patient treatment and management is also impacted positively by AI integration in the robotic system to add further value to these biopsies. They use AI algorithms for target identification, image processing, calculation of the optimal path for needle placement, and operation feedback. Robotic technology and AI can be potential implementations to overcome human mistakes.
- In April 2022, the MicroPort Trans-bronchial Surgical Robot, a robotic-assisted bronchoscopy navigation system jointly developed by Shanghai MicroPort MedBot (Group) Co., Ltd., was used in a robot-assisted trans-bronchial biopsy for lung nodules. It offers a system to perform lung disease treatment because it integrates all the intelligent support for the operation, including preoperative planning, respiratory compensation, and intraoperative surgery.
Bronchial Biopsy Devices Market Growth Factor
- Rising incidence of respiratory diseases: COPD, asthma, and lung cancer are becoming more prevalent, and bronchial biopsy is needed to confirm these diseases more often.
- Aging population: The average global population is aging, and as people age, they are more likely to have respiratory diseases that require diagnostic or therapeutic procedures that require biopsy devices.
- Improved healthcare infrastructure: Rising healthcare infrastructure and the development of technologies in developing nations are aiding in the availability of various complicated medical devices , such as bronchial biopsy tools, that are helping the growth of the bronchial biopsy devices market.
- Government and insurance coverage: As more governments and insurance companies recognize the importance of covering respiratory diseases with treatment and diagnostic procedures, bronchial biopsy devices are becoming available to a new population base.
Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Market Size by 2026 | USD 777.02 Million |
| Market Size in 2025 | USD 723.35 Million |
| Market Size in 2035 | USD 1,468.35 Million |
| Market Growth Rate from 2026 to 2035 | CAGR of 7.34% |
| Leading Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Driver
Rising incidence of respiratory diseases
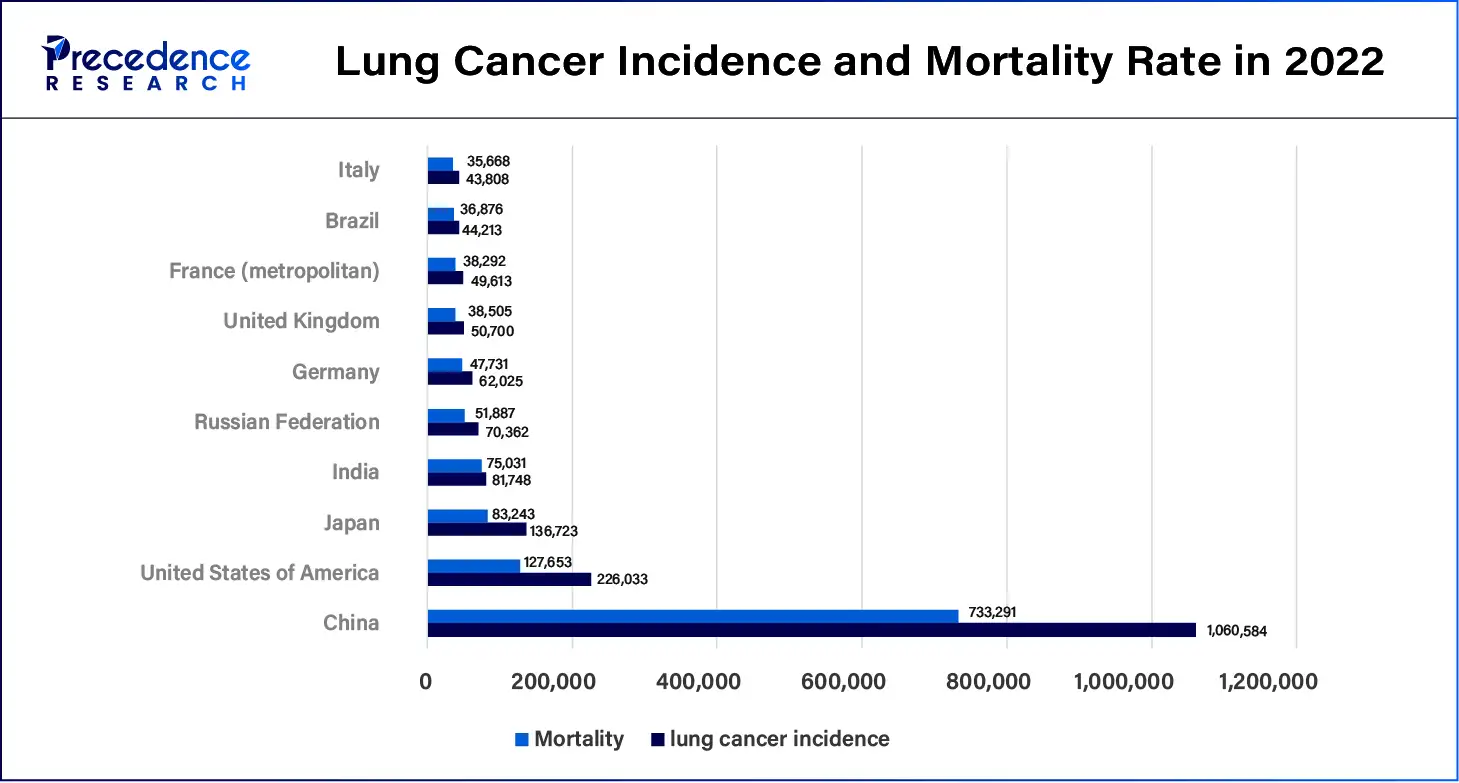
Lung cancer continues to become one of the most prevalent types of cancer, and the incidence rate is still rising. Long-term respiratory diseases include chronic obstructive pulmonary disease (COPD), lung cancer, asthma, and interstitial lung disease. Such an increase in respiratory diseases has put more demand for higher and more accurate methods of diagnosis. The leading factors that may have resulted in this rise include increased environmental pollution, smoking, working conditions, and demographic factors. Bronchial biopsy devices facilitate accurate diagnosis early enough for intervention. Lung ailments, including COPD, asthma, and lung malignancy, are progressively being realized in the global populace. The growing operation of bronchial biopsies for diagnosing lung cancer and related conditions.
- The GLOBOCAN 2022 database reveals that lung cancer was the most common cancer identified in 2022, with almost 2.5 million people diagnosed, or one in eight cancers identified globally (12.4% of all cancers globally).
Restraints
Cost constraints
The growth of the bronchial biopsy devices market is limited by high production and purchasing costs, especially for devices that are equipped with the latest technologies. These advanced devices are expensive to develop. The maintenance, training, and other operational costs of these advanced biopsy devices also augment the overall medical care expenses. Many of these devices are costly, putting a financial burden on both healthcare centers and patients to acquire the diagnostic tools that are needed to improve the generation and application of diagnostic procedures.
Opportunity
Rising demand for minimally invasive device
This shift towards minimally invasive techniques is contributing to the expansion of the bronchial biopsy devices market. The biopsy devices are tissue-sparing, which means that the recovery is quicker, skin closures are better with less scar tissue, and the chances of complications are much less than with an open biopsy. As a result, there is a rising preference for these devices among both patients and healthcare providers.
Innovations in bronchial biopsy devices are making processes less invasive, which aids both patients and healthcare providers. Further specific instruments and techniques lessen patient discomfort and recovery times. The trend towards minimally invasive procedures will continue to gain traction. The bronchial biopsy devices market is estimated to develop significantly over the period due to the rising inclination for minimally invasive diagnostic procedures.
Segment Insights
[[segment_insights]]
Region Insights
[[regional_insights]]
Value Chain Analysis
[[value_chain]]
Bronchial Biopsy Devices Market Companies
[[market_company]]

Other Major Key Players
- Olympus Corporation
- Becton, Dickinson and Company (BD)
- Cook Medical
- Telemed Systems, Inc.
- HOBBS MEDICAL INC
- Argon Medical Devices (WEIGAO GROUP)
- Horizons International Corp.
- Erbe Elektromedizin GmbH
Latest Announcement by Industry Leader
- In October 2024, Cook County Health invited hosted Cook County Board President Toni Preckwinkle to celebrate the launch of the Intuitive Surgical ION Navigational Bronchoscopy platform in John H. Stroger, Jr. Hospital in Chicago.
- The technique of robotic navigational bronchoscopy is a cutting-edge tool that makes diagnosing lung cancer safer, quicker, and more accurate. The technology permits less invasive biopsies, which means less pain and risk of complications for patients, as well as a faster time to treatment and recovery.
Recent Developments
- In October 2025, Invenio Imaging received the FDA's Breakthrough Device Designation for NIO Lung Cancer Reveal, an AI module designed to assist doctors in evaluating bronchoscopic lung forceps biopsies during surgery.
(Source: respiratory-therapy.com ) - In October 2025, Intuitive Surgical received FDA clearance for new software upgrades for its Ion robotic bronchoscopy system, improving minimally invasive lung biopsies with accurate location and shape information through fiber optic technology.
(Source: massdevice.com ) - In April 2024, Single Pass declared that its Class II Kronos biopsy closure device had received clearance from the U.S. Food and Drug Administration (FDA). The FDA has determined that devices are significantly equivalent to predicted devices in terms of their safety and effectiveness.
- In May 2023, Argon Medical Devices, Interventional solutions for Radiology, surgery intervention, cardiology, and oncology, announced the release of the SuperCore Advantage Semi-Autocused biopsy device as the latest addition to their soft tissue biopsy devices in the United States market.
- In September 2022, Serpex Medical received a U.S. FDA 510(k) clearance on Compass Steerable Needles, steerable biopsy needles that enable precise access to lung nodules in the intrapulmonary region. Serpex Medical aims to use the advantage of steerable instruments to allow enhanced accuracy and reach to facilitate the detection and management of lung cancer. The clearance of the Compass Steerable Needles follows the recent clearance of Serpex's Recon Steerable Sheath.
Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
