What is the C Reactive Protein Testing Market Size in 2026?
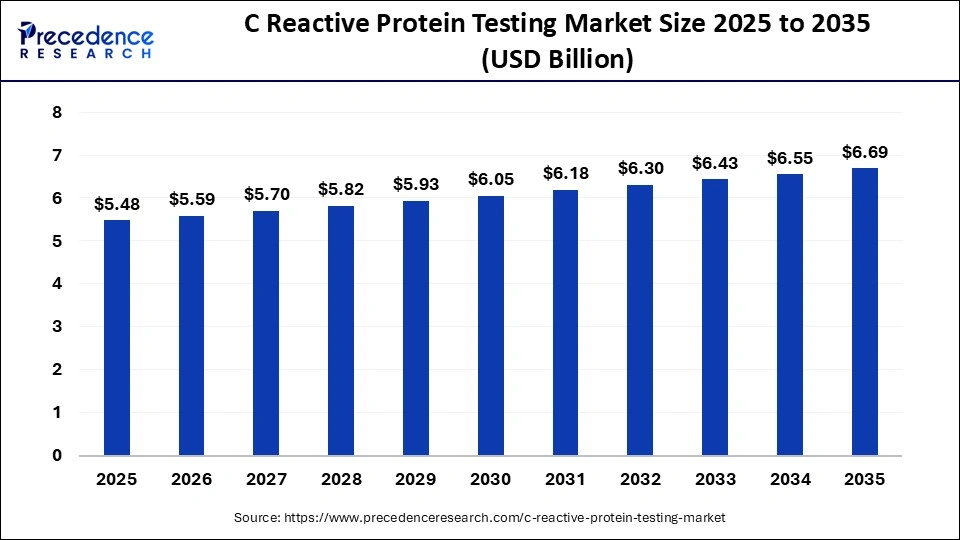
The global C reactive protein testing market size was calculated at USD 5.48 billion in 2025 and is predicted to increase from USD 5.59 billion in 2026 to approximately USD 6.69 billion by 2035, expanding at a CAGR of 2.01% from 2026 to 2035. The market is driven by the rising prevalence of inflammatory diseases and the growing demand for early diagnostics.
Key Takeaways
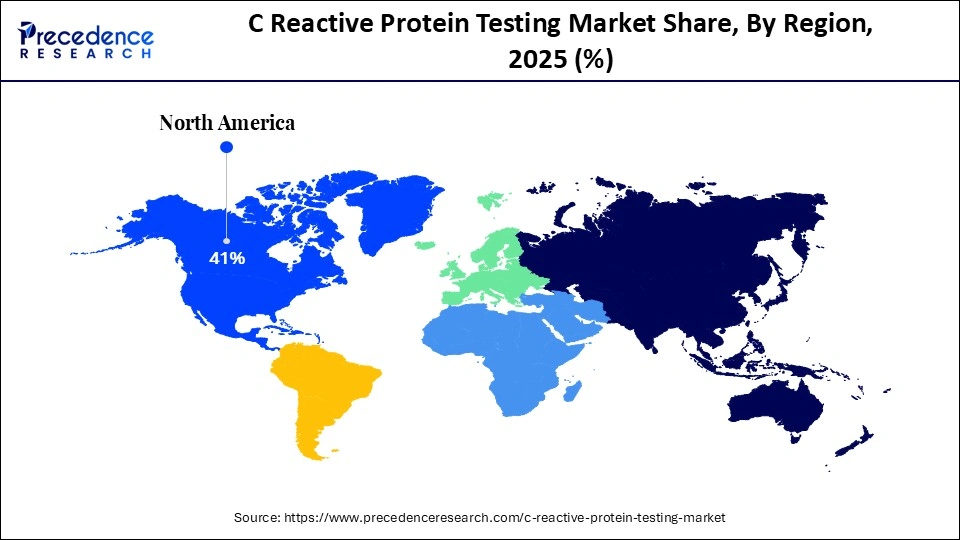
- North America accounted for the largest share of the C reactive protein testing market with the largest market share of 41% in 2025.
- Asia Pacific is anticipated to witness the fastest growth during the forecast period.
- By assay type, the immunoturbidimetric assays segment contibuted a major share of 49% in 2025.
- By assay type, the ELISA segment is expected to grow at a significant rate over the forecast period.
- By detection range, the hs-crp segment contributed the highest market share of 62.02% in 2025.
- By detection range, the conventional CRP segment is anticipated to grow at a significant rate in the market over the forecast period.
- By disease area, the rheumatoid arthritis segment held a major market share of 25.78% in 2025.
- By disease area, the cardiovascular diseases segment is expected to expand at a significant CAGR in the market over the forecast period.
- By end use, the hospitals segment accounted for the biggest market share of 38.17% in 2025.
- By end use, the laboratories segment is anticipated to expand at the fastest rate over the forecast period.
Market Overview
C-reactive protein (CRP) testing is a diagnostic procedure that has gained significant traction as a form of testing the quantity of CRP in blood, which is a body biomarker of inflammation. In diagnosing infections, heart diseases, and other inflammatory diseases, it is important, as it enables healthcare practitioners to start treatment early. With the global shift toward preventive care, CRP testing has become an integral part of routine health assessments and risk evaluations. Moreover, the level of health awareness among individuals has been on the rise, which has led to the rise in the demand for preventative diagnostic tests such as the CRP test. This trend is further supported by the necessity to reduce the long-term cost of health care and improve the outcomes of patients.
How Does AI Integration Impact the C Reactive Protein Testing Market?
The emergence of artificial intelligence is radically changing the C reactive protein testing market due to its ability to improve diagnostic accuracy and efficiency and enhance clinical decision-making. AI-based algorithms are finding more applications to analyze the results of the CRP test and patient data, allowing to identify inflammatory conditions, infections, and cardiovascular risks with increased accuracy. Furthermore, the use of AI in conjunction with point-of-care devices offers real-time processing of the data and remote monitoring of the patients, enhancing their management, particularly in decentralized and resource-restricted environments.
C Reactive Protein Testing Market Trends
- The rise of point of care testing devices is transforming CRP diagnostics, enabling clinics and emergency departments to obtain rapid results, improve clinical decision making, reduce turnaround times, and enhance patient management efficiency.
- Advanced technologies, such as high sensitivity CRP assays, are improving accuracy in diagnosis.
- There is a rising need for early diagnostic tools, particularly for cardiovascular risk assessment and preventive care programs worldwide.
- Growing interest in preventive care and routine health check-ups is increasing the adoption of CRP testing, as individuals seek early detection of inflammatory conditions to avoid chronic complications and costly treatments.
- Expansion of diagnostic services in emerging economies is fueled by improvements in healthcare infrastructure, supportive government initiatives, increased healthcare spending, and rising awareness of the benefits of early disease detection.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 5.48 Billion |
| Market Size in 2026 | USD 5.59 Billion |
| Market Size by 2035 | USD 6.69 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 2.01% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Assay Type, Detection Range, Disease Area, End-use, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Increased Need for Early Diagnosis
Increased need for early disease diagnosis is driving the market. CRP testing is very crucial in the diagnosis of infections, cardiovascular diseases, and inflammatory disorders at an early stage to prevent medical intervention and enhance patient outcomes. The demand for CRP testing has increased considerably due to the importance of regular health check-ups and active management of diseases among individuals. Also, governments and healthcare institutions are encouraging massive screening campaigns to identify high-risk groups and minimize the risk of chronic illnesses.
Restraint
Limited Accessibility and Affordability
The market faces significant challenges related to accessibility and cost, particularly in low- and middle-income countries. Most of the healthcare facilities in developing regions do not have sophisticated diagnostic infrastructure and qualified personnel to conduct proper CRP tests. Moreover, small clinics and under-invested healthcare systems may be unable to afford the cost of high-sensitivity CRP tests, as well as the expensive latest diagnostic equipment. This limits its large-scale adoption, particularly in rural and remote areas where the resources of healthcare are already constrained.
Opportunity
Technological Innovations and Growth of Point-of-Care Testing
Ongoing advances in diagnostic technology and the rising demand for point-of-care testing create immense opportunities in the market. Innovations that are making CRP tests faster, more accurate, and more convenient include portable diagnostic machines, high-sensitivity tests, and computerized test systems. All these developments allow healthcare providers to deliver immediate results even in decentralized settings, i.e., clinics, emergency rooms, and home care organizations.
Segment Insights
Assay Type Insights
Why Did the Immunoturbidimetric Assays Segment Lead the C Reactive Protein Testing Market?
The immunoturbidimetric assays segment led the market with the largest share in 2025, as these assays are highly efficient, scalable, and cost-effective. These assays are extensively used in clinical laboratories due to their advantage of low processing time and high throughput results. Their capability to be combined with automated workflows is furthermore more effective in terms of workflow and minimal interactions with human color, which is particularly advantageous in the hospital and diagnostic laboratory settings. They are also cheap to operate compared with other complex procedures, making them a preferred choice among healthcare providers.
The ELISA segment is expected to grow at a significant CAGR over the forecast period due to the high sensitivity and specificity of ELISA assays in the detection of CRP levels in human serum and plasma. The quality of ELISA is most appreciated in clinical diagnostics and research because it produces specific and reproducible data even at extremely low levels of CRP concentrations. The current developments have improved its automation, accuracy, and efficiency in its functioning, and it has become a preferred choice in contemporary labs. The increasing rate of inflammatory diseases, cardiovascular disorders, and autoimmune diseases is increasing the use of ELISA-based CRP testing kits.
Detection Range Insights
Why Did the hs-CRP Segment Dominate the C Reactive Protein Testing Market?
The hs-CRP segment dominated the market with the largest share in 2025. This is primarily due to its high sensitivity, making it the preferred choice to detect minimal levels of CRP of about 0.3 to 10mg/L. This range is necessary to detect low-grade inflammation and determine the risk of cardiovascular disease at a very early stage. The growing worldwide epidemiology of cardiovascular conditions has greatly contributed to the need to test hs-CRP because it offers adequate information on the risk of heart diseases in addition to the conventional lipid profiling. The CRP test is normally used in conjunction with other diagnostic techniques, such as rheumatoid factor, anti-CCP antibodies, and imaging techniques, which make it more important in disease assessment.
The conventional CRP segment is expected to grow at a significant rate in the upcoming period, owing to its low cost, widespread availability, and ability to identify greater levels of inflammation. Conventional CRP tests have the ability to detect CRP levels between 10 mg/L and 1, 000mg/L. Hence, they are applicable especially in the diagnosis of acute infections and injuries, as well as chronic inflammatory diseases. Immunoassays and laser nephelometry are widely used in conventional CRP testing and provide effective and affordable diagnostic tests. The demand for conventional CRP testing is increasing, driven by the rising prevalence of infectious diseases and inflammatory disorders, particularly in developing areas.
Disease Area Insights
Why Did the Rheumatoid Arthritis Segment Lead the C Reactive Protein Testing Market?
The rheumatoid arthritis segment led the market with the largest revenue share in 2025. This is because CRP testing has become popular in measuring the levels of inflammation in rheumatoid arthritis patients, enabling health practitioners to monitor the disease progression as well as the effectiveness of the treatment. The rapid increase in the prevalence of rheumatoid arthritis around the world, as well as growing awareness of early diagnosis, has played a major role in the growth of the segment. CRP testing is often combined with other diagnostic methods, such as rheumatoid factors, anti-CCP antibodies, and imaging, enhancing its role in comprehensive disease evaluation.
The cardiovascular diseases segment is expected to grow at a significant CAGR over the forecast period, since the burden of heart-related diseases around the world is on the rise. This increases the necessity to detect the potential danger at an early stage. Elevated CRP levels are closely linked to an increased risk of cardiovascular events, including heart attacks and strokes, making CRP testing a valuable tool in preventive cardiology. The adoption of high-sensitivity CRP (hs-CRP) tests alongside traditional lipid profiling improves risk prediction and enables early intervention, with demand further driven by growing awareness of heart health and the importance of regular screenings.
End-use Insights
Why Did the Hospitals Segment Contribute the Most Revenue in the C Reactive Protein Testing Market?
The hospitals segment held the largest share of the market in 2025, since hospitals serve as primary centers for diagnosing, monitoring, and managing inflammatory, infectious, and cardiovascular diseases. Equipped with laboratory facilities, trained staff, and a high patient volume, hospitals are well-positioned to conduct CRP testing effectively. The growing prevalence of infectious, autoimmune, and cardiovascular conditions, along with increased focus on point-of-care testing, particularly in emergency departments, has further driven the adoption of high-sensitivity CRP tests in hospital settings.
The laboratories segment is expected to grow at a substantial rate in the coming years. This is because CRP tests are frequently conducted in controlled lab settings for monitoring infections, inflammatory conditions, and postoperative complications using advanced analytical tools. The growth of the segment is further driven by increasing demand for accurate, timely, and accessible diagnostic services, particularly in urban and semi-urban areas. Additionally, laboratories play a key role in research, preventive healthcare programs, and disease monitoring campaigns, positioning them for strong growth as healthcare systems emphasize early diagnosis and personalized treatment plans.
Regional Insights
North America C Reactive Protein Testing Market Size and Growth 2026 to 2035
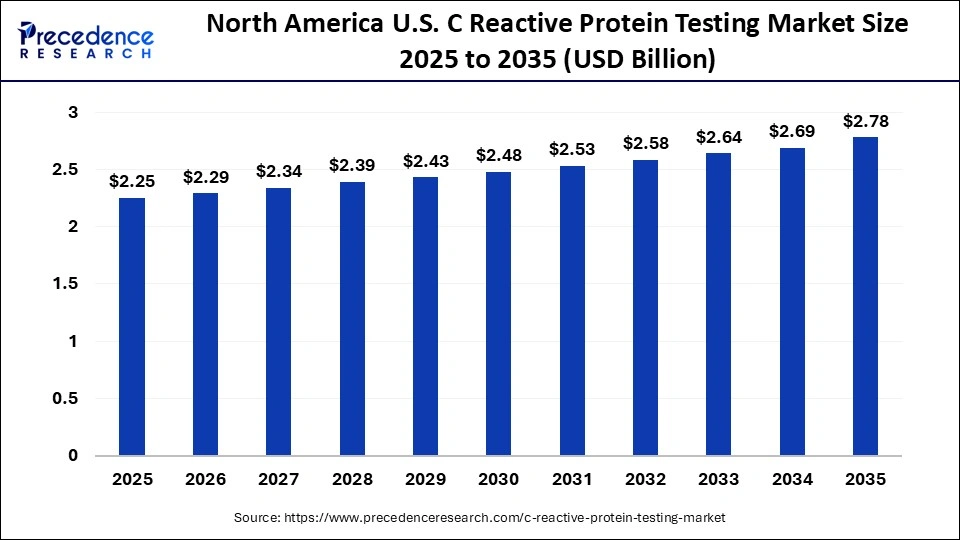
The North America C reactive protein testing market size is estimated at USD 2.25 billion in 2025 and is projected to reach approximately USD 2.78 billion by 2035, with a 2.14% CAGR from 2026 to 2035.
What Made North America the Leading Region in the Global C Reactive Protein Testing Market?
North America led the C reactive protein testing market with the largest share in 2025 due to the high rate of cardiovascular diseases, autoimmune diseases, and other inflammatory diseases in the region. Market leadership has been enhanced by sophisticated healthcare facilities, high proliferation of advanced diagnostic laboratories, and high-density integration of automated immunoturbidimetric and high-sensitivity CRP. The positive reimbursement policies in countries such as the U.S. and Canada promote the use of preventive diagnostics, whereas the rising interest in personalized medicine and early detection of diseases stimulates growth. Also, the increasing awareness among patients about preventive healthcare and frequent screenings has boosted the use of CRP testing.
U.S. C Reactive Protein Testing Market Size and Growth 2026 to 2035
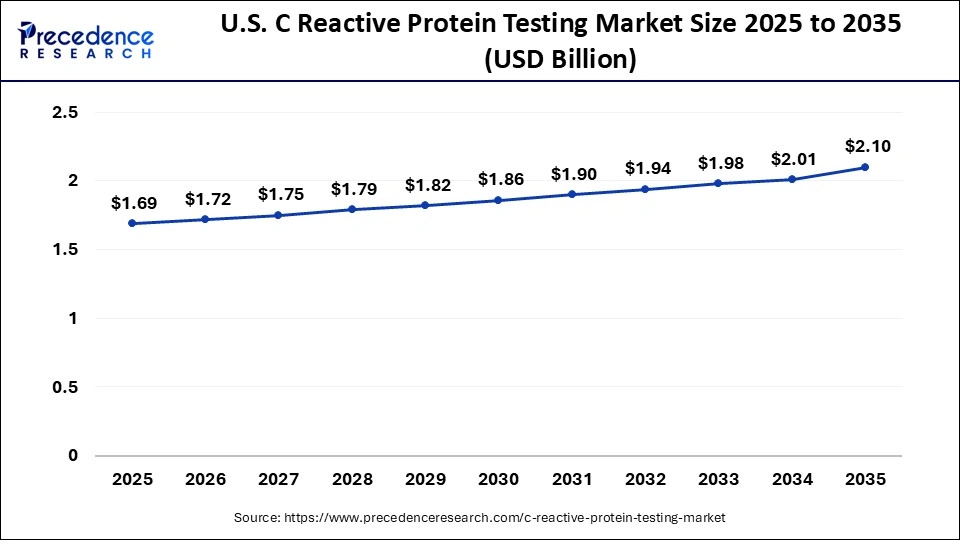
The U.S. C reactive protein testing market size is calculated at USD 1.69 billion in 2025 and is expected to reach nearly USD 2.10 billion in 2035, accelerating at a strong CAGR of 2.20% between 2026 and 2035.
U.S. C Reactive Protein Testing Market Analysis
The U.S. holds a substantial share of the market and is the major contributor to the market. The country has a well-developed healthcare IT infrastructure. Significant investment in research and development provides the backbone for rapid technological advancements. The country is home to leading market players such as Veeva Systems, MasterControl Inc., Oracle Corporation, ArisGlobal, Parexel International, Merative, Xybion Corporation, ClinOne (Clindex), EXTEDO GmbH, and others. These companies are recognized for providing secure, validated, and compliant document management tools. Additionally, the increasing integration of Artificial Intelligence (AI) and Machine Learning (ML) assists in automating workflows and enhancing document quality control, which is expected to fuel the market's growth in the country.
Why is Asia Pacific Experiencing the Fastest Growth in the C Reactive Protein Testing Market?
Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period due to the rapid expansion of healthcare facilities and laboratories. Rising populations, particularly the growing geriatric demographic in countries like Japan and China, have increased demand for diagnostic services. Additionally, the high prevalence of cardiovascular, infectious, and inflammatory diseases, coupled with growing health awareness, government initiatives promoting preventive check-ups, and rising healthcare spending, is fueling market growth. The expanding private healthcare infrastructure and improved access to quality diagnostics in urban and semi-urban areas also drive the market across the region.
C Reactive Protein Testing Market Companies
- Thermo Fisher Scientific, Inc.
- F. Hoffmann-La Roche Ltd.
- Danaher
- Quest Diagnostics
- Siemens Healthineers AG
- Abbott
- Merck KGaA
- Zoetis
- Ortho Clinical Diagnostics
- Getein Biotech, Inc.
- HORIBA, Ltd
- Randox Laboratories Ltd.
- Boditech Med, Inc.
- Aidian
Recent Developments
- In September 2025, HORIBA entered into a strategic alliance with GeodAIsics to create an AI-based system to detect sepsis early and combine their CRP-compatible hematology analyzers to identify the patterns of infection and inflammation. This partnership aimed at improving quick clinical decision-making and patient outcomes in intensive care units. (Source: https://www.horiba.com )
- In August 2025, Quest Diagnostics bought some of the clinical testing holdings of Spectra Laboratories, the chronic kidney disease diagnostic unit of Fresenius Medical Care, enhancing its diagnosis expertise. The acquisition enlarges the network of labs that Quest has and gives it more ability to provide special testing services in order to manage patients better. (Source: https://freseniusmedicalcare.com )
- In September 2024, Monod Bio, a life sciences company leveraging AI for protein discovery in RUO and IVD markets, launched NovoLISA, a rapid biosensor assay platform delivering results in just 15 minutes. NovoLISA (“de Novo protein-Linked Instantaneous Solution-based Assay”) significantly reduces testing time compared to traditional methods like ELISA, with its first offering being a C-Reactive Protein (CRP) assay.(Source: https://www.prnewswire.com )
Segments Covered in the Report
By Assay Type
- Immunoturbidimetric Assay
- ELISA
- Clinical
- Nonclinical
- Chemiluminescence Immunoassay
- Others
By Detection Range
- hs-CRP
- Conventional CRP
- cCRP
By Disease Area
- Cardiovascular Diseases
- Cancer
- Rheumatoid Arthritis
- Inflammatory Bowel Disease
- Endometriosis
- Lupus
- Others
By End-use
- Clinics
- Hospitals
- Laboratories
- Assisted Living Healthcare Facilities
- Homecare Settings
- Others
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
