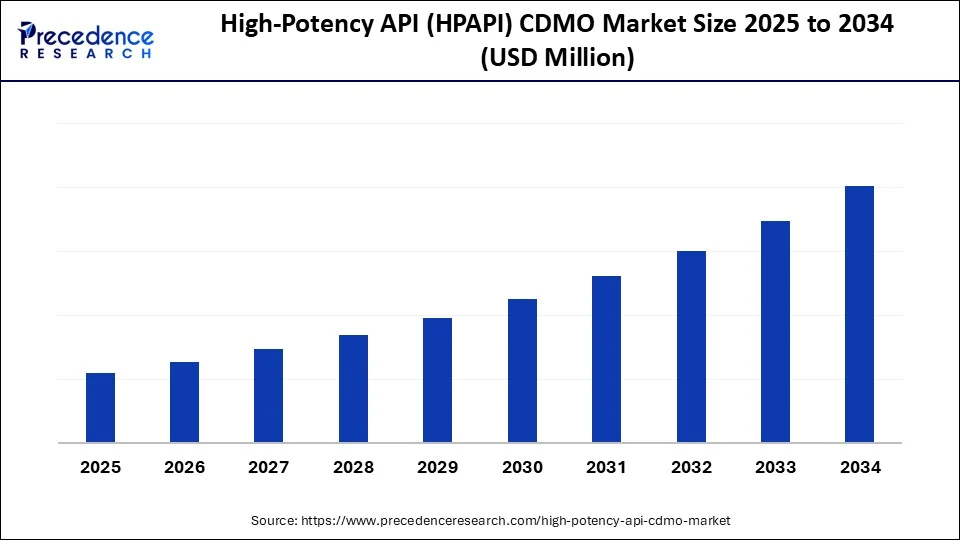
High-Potency API (HPAPI) CDMO Market Size and Forecast 2025 to 2034
The high-potency API (HPAPI) CDMO market is accelerating as pharmaceutical companies focus on oncology, hormonal therapies, and niche drugs, driving demand for specialized manufacturing expertise. The high-potency API (HPAPI) CDMO market is a rapidly growing segment within the pharmaceutical industry, driven by increasing demand for specialized drug manufacturing services. With a strong projected growth trajectory, this market is expected to expand significantly over the coming decade, reflecting its vital role in advanced medical therapies.
High-Potency API (HPAPI) CDMO Market Key Takeaways
- North America dominated the high-potency API (HPAPI) CDMO market in 2024.
- By region, the Asia Pacific is anticipated to grow at the fastest CAGR during the forecast period.
- By type of HPAPI, the innovative HPAPIs segment held a the biggest market share in 2024.
- By Type of HPAPI, the generic HPAPIs segment is expected to grow at the fastest CAGR during the forecast period.
- By the synthesis method, the synthetic HPAPIs segment accounted for a considerable share in 2024.
- By synthesis method, the biotech HPAPIs segment is projected to experience the highest growth CAGR between 2025 and 2034.
- By service type, the contract manufacturing services segment captured the biggest market share in 2024.
- By service type, the contract development services segment is set to experience the fastest CAGR from 2025 to 2034.
- By application type, the oncology segment contributed the highest market share in 2024.
- By application type, the antibody drug conjugates (ADCs) segment is projected to expand rapidly coming years.
- By end-user type, the pharmaceutical companies segment generated the major market share in 2024.
- By end-user type, the biotechnology companies segment is projected to expand rapidly in the coming years.
How Is AI Transforming the High-Potency API (HPAPI) CDMO Market?
Artificial intelligence is emerging as a game-changer for high-potency API (HPAPI) CDMOs, streamlining processes across drug development and manufacturing. AI-driven predictive analytics are being leveraged to optimize formulation strategies, improve containment protocols, and forecast demand with greater accuracy. Machine learning tools enable better design of scalable manufacturing processes, reducing costly trial-and-error approaches. In quality control, AI helps monitor data in real-time to detect deviations early and ensure regulatory compliance. Furthermore, AI enhances supply chain visibility by anticipating disruptions and optimizing management. As a result, CDMOs integrating AI technologies are gaining a competitive edge by ensuring higher productivity, cost efficiency, and speed to market.
- In April 2025, Phlow Corp is advancing its high-potency active pharmaceutical ingredients and antibody drug conjugate capabilities by integrating cutting-edge AI-powered analytical technologies. This investment reflects the growing reliance on AI to deliver greater precision, efficiency, and compliance in the production of highly potent and complex therapies within the CDMO sector. (Source: https://www.contractpharma.com)
Why Are High-Potency API (HPAPI) CDMOs Becoming Indispensable?
The high-potency API (HPAPI) CDMO market refers to specialized third-party service providers engaged in the development, scale-up, and commercial manufacturing of highly potent active pharmaceutical ingredients (HPAPIs) used in oncology, hormonal therapies, antibody-drug conjugates (ADCs), and other targeted therapeutics. Due to their high pharmacological activity at low doses, HPAPIs require specialized containment facilities, advanced technologies, and regulatory-compliant manufacturing practices. CDMOs play a critical role by offering end-to-end services ranging from process development and analytical testing to formulation and packaging.
The high-potency API (HPAPI) CDMO market is expanding rapidly, driven by increasing adoption of highly targeted drugs such as antibody-drug conjugates (ADCs). CDMOs provide the specialized containment facilities and process expertise that pharmaceutical companies often lack internally. With oncology continuing to dominate drug pipelines globally, demand for High-potency production capacity is outpacing supply, creating strong opportunities for service providers. Additionally, the outsourcing model allows pharma companies to reduce risk, minimize capital investment, and access advanced technologies. Regulatory compliance is a defining factor in this market, with CDMOs focusing on achieving and maintaining international standards for safety and quality. As innovation intensifies, the role of CDMOs in the HPAPI space has shifted from manufacturing partners to strategic collaborators.
Market Key Trends
- Rising demand for targeted therapies, especially oncology drugs and ADCs.
- Increasing outsourcing of high-potency API production to reduce capital burden.
- Expansion of CDMO facilities with advanced containment and automation systems.
- Strategic partnerships and acquisitions to expand global footprints.
- Growing regulatory scrutiny, pushing higher standards of quality and safety.
- Adoption of AI, automation, and continuous manufacturing technologies.
Market Scope
| Report Coverage | Details |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Type of HPAPI, Synthesis Method, Service Type, Application, End User and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
The Cancer Therapy Catalyst
The oncology sector continues to be the single biggest driver of the high-potency API (HPAPI) CDMO market. As cancer treatment shifts toward more personalized, highly potent molecules such as ADCs, the need for specialized high-potency production facilities is growing exponentially. Pharmaceutical companies are turning to CDMOs with the necessary expertise and infrastructure to meet these complex requirements for safety. The rising incidence of cancer globally and an increasing focus on targeted therapies ensure steady demand for high-potency API. The ability of CDMOs to offer integrated services, from process development to commercial-scale manufacturing, is positioning them as key enablers in oncology, a central force propelling this market growth.
- In August 2025, Industry leaders, including experts from Veranova, emphasize that the rising demand for complex small-molecule drugs and high-potency API (HPAPI) CDMO is accelerating the adoption of digital transformation in manufacturing. AI-enabled process optimization, predictive analytics, and integrating data platforms are now playing a key role in streamlining workflows, improving efficiency, and enabling smarter, predictive manufacturing approaches.
(Source: https://www.dcatvci.org)
Restraint
The Safety and Cost Challenge
Despite strong growth, the high-potency API (HPAPI) CDMO industry faces key challenges in safety and cost management. The high potency of these molecules demands stringent containment protocols, specialized equipment, and continuous training for staff, all of which drive up operational costs. Regulatory hurdles add another layer of complexity, often leading to longer timelines and higher compliance expenses. Limited global capacity also means that demand often exceeds supply, creating bottlenecks. For smaller CDMOs, competing with larger players who can invest in advanced facilities and AI-driven automation remains a challenge. Ultimately, balancing safety, cost-efficiency, and scalability remains the most pressing restraint in this market.
Opportunity
Unlocking Value Through Specialized Outsourcing
A major opportunity in the high-potency API (HPAPI) CDMO market lies in expanding capacity and capabilities to meet unmet demand. Many mid-size and emerging biotech firms lack in-house containment facilities and are fully dependent on outsourcing. This creates room for CDMOs to differentiate through niche expertise, flexible manufacturing, and end-to-end services models. Furthermore, opportunities exist in geographic expansion, particularly in emerging markets, where demand for advanced therapies is growing. With increasing investments in innovative modalities like ADCs and oligonucleotides, CDMOs with diversified portfolios stand to capture significant excellence with speed and scalability, representing the strongest opportunity for long-term market leadership.
- In August 2025, Laurus Labs revealed a strategic restructuring of its CDMO division, Laurus Synthetics. As part of this move, the API manufacturing unit will be integrated with its subsidiary, Sriam Labs, which specializes in APIs and intermediates production. The consultation is aimed at streamlining operations, optimizing resources, and strengthening overall efficiency. This step is expected to significantly enhance the company's capacity and flexibility in high-potency API manufacturing. By unifying operations, Laurus Labs positions itself in high-potency API manufacturing. By unifying operations, Laurus Labs positions itself for stronger growth potential and improved competitiveness. (Source: https://timesofindia.indiatimes.com)
Type of HPAPI Insight
Why are Innovative HPAPIs Dominating the High-Potency API (HPAPI) CDMO Market?
The innovative HPAPIs have become a dominant force in the high-potency API (HPAPI) CDMO industry, followed by the rising demand for novel, targeted therapies in oncology, immunology, are rare diseases. These APIs are typically developed under strict regulatory frameworks, requiring advanced manufacturing expertise and significant R&D investments. Pharmaceutical companies increasingly focus on innovation to maintain exclusivity and ensure higher profit margins, further fueling the segment's dominance. Additionally, the growing number of FDA and EMA approvals for innovative drugs containing high-potency API (HPAPI) continues to strengthen this market position.
The generic HPAPIs are the fastest-growing in the high-potency API (HPAPI) CDMO sector, because of open opportunities for cost-effectiveness alternatives. Growing healthcare cost pressure globally and the increasing adoption of generics in emerging markets are accelerating this trend. Manufacturers specializing in generics are investing in high-potency API (HPAPI) CDMO capabilities to expand their portfolios and cater to demand. Moreover, government initiatives promoting generic adoption further drive this segment's rapid expansion.
Synthesis Method Insights
Why are Synthetic HPAPIs Dominating the High-Potency API (HPAPI) CDMO Market?
The synthetic HPAPIs have become a dominant force in the high-potency API (HPAPI) CDMO industry, due to well-established production technologies and standardized manufacturing processes. They allow for large-scale, cost-efficient production while meeting stringent quality enhancements. The broad applications across oncology, cardiovascular, and CSN therapies contribute to sustained demand. The scalability and maturity of chemical synthesis platforms make them the backbone of high-potency API manufacturing.
The biotech HPAPIs are the fastest-growing in the high-potency API (HPAPI) CDMO sector, fueled by advancements in biologics, antibody-drug conjugates, and targeted therapies. As personalized medicine gains traction, biologically derived high-potency API (HPAPIs) are increasingly favored for their precision and efficacy. With the expansion of recombinant technologies, cell-based systems, and fermentation processes, biotech-based methods are set to outpace traditional methods. Their relevance in next-generation therapies ensures robust growth potential.
Service Type Insights
Why Contract Manufacturing Services are Dominating the High-Potency API (HPAPI) CDMO Market?
The contract manufacturing services have become a dominant force in the high-potency API (HPAPI) CDMO industry; as a result, companies seek to optimize costs and focus on core competencies like R&D and commercialization. Outsourcing high-potency API (HPAPI) production to specialized CDMOs provides access to advanced containment facilities and regulatory expertise. These partnerships are critical given the complexity and safety concerns in high-potency API handling, cementing contract manufacturing's dominant role.
The contract development services are the fastest-growing in the high-potency API (HPAPI) CDMO sector, driven by increasing outsourcing of early-stage formulation, process development, and scale-up activities. Rising demand for customized solutions, coupled with the need to accelerate clinical timelines, drives reliance on development-focused CDMOs. The integration of advanced analytical tools and AI in development processes is also fueling growth in this segment.
Application Insights
Why Oncology is Dominating the High-Potency API (HPAPI) CDMO Market?
The oncology services have become a dominant force in the high-potency API (HPAPI) CDMO industry, caused by the rising incidence of disease risk worldwide, combined with the emphasis on targeted and highly potent therapies, which drives the expansion of this segment. Many breakthrough oncology drugs rely on high-potency API for therapeutic efficacy, reinforcing oncology's market leadership.
The antibody-drug conjugate services are the fastest-growing in the high-potency API (HPAPI) CDMO sector, thanks to their role in precision oncology. ADCs combine the targeting ability of antibodies with the potency of high-potency HPAPI, offering high efficacy with reduced systemic toxicity. The increasing number of ADCs in clinical pipelines and recent regulatory approvals highlight their strong growth trajectory.
End User Insight
Why are pharmaceutical companies dominating the High-Potency API (HPAPI) CDMO Market?
The pharmaceutical companies have become a dominant force in the high-potency API (HPAPI) CDMO industry, due to their extensive drug pipelines and manufacturing scale. Their strong financial resources allow significant investments in both in-house capabilities and outsourcing partnerships. Continuous demand for innovative therapies and expansion of global product portfolios ensure pharmaceutical companies retain their leading role.
The biotechnology companies' services are the fastest-growing in the high-potency API (HPAPI) CDMO sector, driven by spearhead innovations in biologics, gene therapies, and ADCs. Their focus on niche, targeted therapies drives demand for specialized HPAPIs. Many biotech firms rely heavily on CDMOs for both development and manufacturing support, accelerating their market footprint. With growing investments and venture funding in biotech, this segment is poised for rapid expansion.
Regional Insights
Why Does North America Dominate the High-Potency API (HPAPI) CDMO Market?
By region, North America dominated the global market for high-potency API (HPAPI) CDMOs in 2024, owing to its strong pharmaceutical and biotechnology ecosystem. The region is home to leading pharma companies with extensive pipelines in oncology and targeted therapies, creating sustained demand for high-potency API (HPAPI). CDMOs in the U.S. and Canada benefit from advanced infrastructure, high regulatory standards, and significant R&D investments. Furthermore, North America has been at the forefront of adopting innovative technologies such as continuous manufacturing and AI integration. The concentration of skilled talent and strong collaboration between pharma companies, research institutions, and CDMOs further strengthens its leadership position.
A recent study stated that competitive pressures and rapid technological progress are driving the pharmaceutical companies to accelerate product launches. Partnering with a contract manufacturer can significantly speed up this process; however, careful due diligence is essential to ensure compliance with regulatory standards and adherence to best practices, particularly when working with highly potent active pharmaceutical ingredients.
(Source: https://www.pharmaceutical-technology.com)
How Is Asia Pacific Becoming the Fastest-Growing High-Potency API (HPAPI) CDMO Market?
Asia Pacific is the fastest-growing in the high-potency API (HPAPI) CDMO market sector, driven by expanding pharmaceutical manufacturing hubs in countries like China and India. Lower production costs, coupled with growing investments in high-containment facilities, are making the region attractive for outsourcing HPAPI production. Governments are also supporting the growth of advanced drug manufacturing through favorable policies and funding initiatives. Additionally, rising incidences of cancer and chronic diseases in the Asia-Pacific are creating local demand for potent drugs. Many global CDMOs are expanding into the region through partnerships and facility setups to tap into this growth. With its cost advantage and rapidly improving capabilities, Asia-Pacific is set to become a powerful force in the HPAPI CDMO market.
- In February 2025, Granules India Limited announced on Friday that it has signed an acquisition agreement for Senn Chemicals AG, a Switzerland-based CDMO specializing in peptides. According to the company, this acquisition strengthens its position in the high-growth peptide therapeutics sector. The move is aligned with Granules' strategy to expand into fast-growing peptide anti-diabetic and anti-obesity markets, including GLP-1 receptor agonist and other next-generation therapies, the company stated. (Source: https://www.moneycontrol.com)
High-Potency API (HPAPI) CDMO Market - Value Chain Analysis
- R&D: A high-potency API contract development and manufacturing organization, research and development, specializing in the development and production of highly potent drugs. These compounds are effective at very low doses but pose substantial risks, including occupational hazards and cross-contamination, requiring specialized facilities and stringent safety protocols.
- Clinical Trials and Regulatory Approvals: High Potency active pharmaceutical ingredients development entails a CDMO overseeing specialized processes from drug discovery to commercialization, while ensuring full regulatory compliance for clinical trials and market authorization.
- Raw Materials Procurement: Sourcing raw materials for high-potency active pharmaceutical ingredients development at a CDMO demands stringent supplier qualification, through quality control, and secure handling procedures, given the compound's high biological activity and toxicity.
- Formulation and Final Dosage Preparation: They offer highly potent small molecule development services of API and intermediates, operating from our development facility in India. The development lab can handle molecules with OEL 0.1 µg/m3
- Distribution to Hospitals, Pharmacies: High-potency active pharmaceutical ingredient CDMOs are specialized facilities designed to manage potent drugs for hospitals and pharmacies. They employ advanced containment technologies, rigorous protocols, and highly trained personnel to ensure worker safety and prevent cross-contamination. The distribution process to end-users involves a complex supply chain, where the HPAPI is either complex supply chain, where the HPAPI is either formulated into a finished drug product by the CDMO or delivery by a pharmaceutical company.
High-Potency API (HPAPI) CDMO Market Companies

- Lonza Group
- Catalent Inc.
- WuXi STA (WuXi AppTec)
- Samsung Biologics
- Siegfried Holding AG
- Piramal Pharma Solutions
- Cambrex Corporation
- CordenPharma International
- Recipharm AB
- Sterling Pharma Solutions
- Asymchem Laboratories
- Albany Molecular Research (AMRI)
- Novasep
- Ajinomoto Bio-Pharma Services
- Fareva Group
Recent Developments
- In August 2025, Veranova, a leading developer and manufacturer of specialty and complex active pharmaceutical ingredients (APIs) for the pharmaceutical and biotech industries, announced that a routine cGMP (current Good Manufacturing Practice) surveillance inspection of its Devens, Massachusetts facility, conducted by the FDA New England District on July 31, 2025, was completed without any Form 483 observations and received a No Action Indicated (NAI) classification. (Source: https://www.contractpharma.com)
- In January 2025, AGC Pharma Chemicals unveiled a major expansion of its HPAPI capabilities in Barcelona. The upgraded facility features a cutting-edge R&D laboratory and advanced manufacturing infrastructure, designed to support smooth scale-up from gram-level to ton-scale production. This strategic move is intended to address the rising demand. (Source: https://www.agcpharmachemicals.com)
Segment Covered in the Report
By Type of HPAPI
- Innovative HPAPIs
- Generic HPAPIs
- By Synthesis Method
- Synthetic HPAPIs
- Biotech HPAPIs
By Service Type
- Contract Manufacturing Services
- Contract Development Services
- Analytical & Regulatory Support Services
By Application
- Oncology
- Hormonal Disorders
- Antibody-Drug Conjugates (ADCs)
- Central Nervous System (CNS) Disorders
- Cardiovascular & Others
By End User
- Pharmaceutical Companies
- biotechnology companies
- CROs & Academic Institutes
By Region
- North America
- Asia-Pacific
- Europe
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content


 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
