What is the Interleukin Inhibitors Market Size?
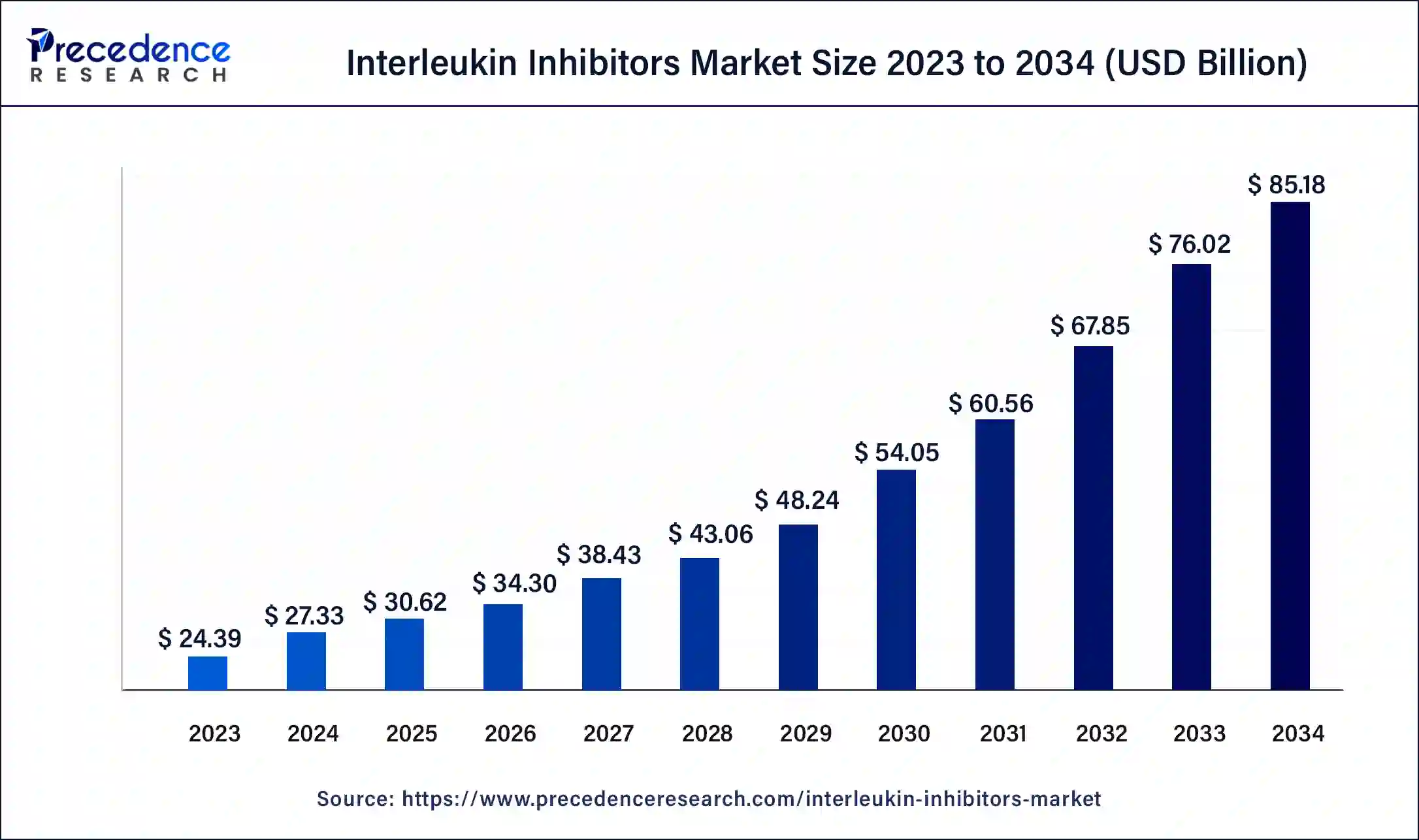
The global interleukin inhibitors market size is calculated at USD 30.62 billion in 2025 and is predicted to increase from USD 34.30 billion in 2026 to approximately USD 93.68 billion by 2035, expanding at a CAGR of 11.83% from 2026 to 2035. The growing cases of hypereosinophilic syndrome across the world are driving the growth of the interleukin inhibitors market.
Interleukin Inhibitors Market Key Takeaways
- The global interleukin inhibitors market was valued at USD 30.62 billion in 2025.
- It is projected to reach USD 93.68 billion by 2035.
- The interleukin inhibitors market is expected to grow at a CAGR of 11.83% from 2026 to 2035.
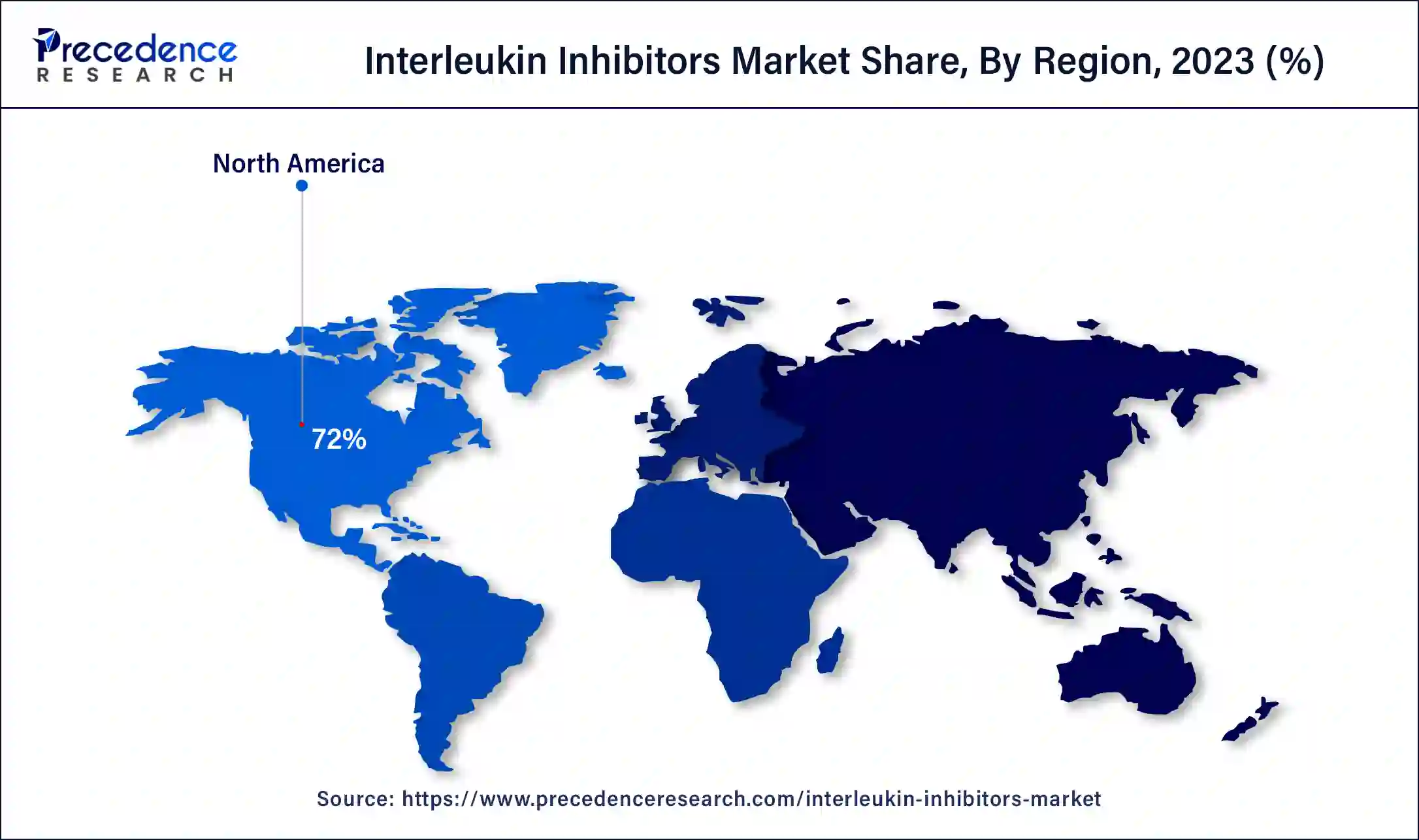
- North America led the interleukin inhibitors market with the highest market share of 72% in 2025.
- By region, Asia-Pacific is expected to attain the fastest rate of growth during the forecast period.
- By application, the psoriasis segment held the largest share of the market in 2025.
- By application, the psoriatic arthritis segment is estimated to exhibit the fastest growth rate during the forecast period.
- By type, the IL-23 segment dominated the market in 2025.
- By type, the IL-17 segment is expected to grow at the fastest growth rate during the forecast period.
What is the Interleukin Inhibitor?
The interleukin inhibitors market is one of the most important industries in the biopharmaceutical sector. This industry deals in manufacturing and distributing superior-grade interleukin inhibitors across the world. The interleukin inhibitors market is mainly driven by the rise in the number of government approvals for using various therapeutics, along with increasing research activities for developing interleukin inhibitors.
This industry comprises various types of interleukin inhibitors such as IL-17, IL-23, IL-1, IL-5, IL-6, and others. Interleukin inhibitors mainly find applications in the treatment of psoriasis, psoriatic arthritis, rheumatoid arthritis, asthma, inflammatory bowel disease (IBD), and some others. This industry is expected to grow significantly with the developments in the pharmaceutical and healthcare industries.
- In May 2024, Almirall launched lebrikizumab (EBGLYSS). Lebrikizumab (EBGLYSS) is ranked as the No.1 interleukin inhibitor by TNF BIO 2406b in the Nordic region for the treatment of severe atopic dermatitis in adults.
How Can AI Improve the Interleukin Inhibitors Industry?
AI plays an important role in the development of the interleukin inhibitors industry. Nowadays, most of the pharmaceutical companies have started integrating AI in drug discovery and development. Also, the applications of AI in determining the efficacy of medicines, along with identifying the side effects, have helped the pharmaceutical industry to a large extent.
Recently, researchers discovered two new IL inhibitors named ‘Celecoxib' and ‘Dexamethasone' that use AI to help in the treatment of various autoimmune diseases. Thus, AI can help in developing new varieties of interleukin inhibitors, thereby improving the landscape of the interleukin inhibitors industry.
How is AI contributing to the Interleukin Inhibitors Industry?
AI is transforming the way interleukin inhibitors are developed with accelerated discovery and intelligent targeting. High-level models can filter molecular candidates in response to cytokine pathways. Selective small molecules are designed through generative tools to be used orally. Predictive analytics predict protein binding.
Clinical data is also used to determine the trend of patient response through AI. AI-based repurposing points out pre-existing drugs with interleukin-blocking characteristics in the changing therapeutic environments.
Interleukin Inhibitors Market Growth Factors
- The growing developments related to IL-17 inhibitors have led to market growth for interleukin inhibitors.
- There is a rise in the number of government initiatives related to biopharmaceutical sectors across the globe.
- Rising preference of doctors for using interleukin inhibitors to treat various diseases.
- There are growing investments from public and private sector entities to develop the interleukin inhibitors industry.
- The advancements in medical therapies impact industrial growth positively.
- The increasing cases of various types of skin diseases have positively accelerated the market.
- There is a rising demand for effective medicine for the treatment of plaque-type psoriasis.
- The upsurge in the number of research activities associated with IL-23.
- The increasing adoption of biosimilars related to Tumor Necrosis Factor (TNF) inhibitors boosts the market growth.
- The growing demand for medicines associated with the treatment of inflammatory bowel diseases has driven the growth of the interleukin inhibitors market.
Market Outlook
- Industry Growth Overview: There is an increasing number of cases of chronic inflammation. Targeted biologics are becoming increasingly popular in the world.
- Global Expansion: Europe is experiencing established reimbursement and biosimilar inclusion of immunology care.
- Major investors: Novartis, AbbVie, Johnson and Johnson, Eli Lilly, Regeneron, Amgen, and Sanofi are the forces behind innovation.
Market Scope
| Report Coverage | Details |
| Market Size by 2035 | USD 93.68 Billion |
| Market Size in 2025 | USD 30.62 Billion |
| Market Size in 2026 | USD 34.30 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 11.83% |
| Largest Market | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Type, Application, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Driver
Increasing cases of autoimmune disorders across the world
The cases of autoimmune diseases such as systemic lupus erythematosus, inflammatory bowel disease, multiple sclerosis, Guillain-barre syndrome, asthma, graves' disease, and some others have increased rapidly across the world. With the rise in number of patients suffering from autoimmune diseases, the demand for effective therapies has also increased for treatment. These diseases can be treated using IL inhibitor-based therapies that, in turn, drive the growth of the interleukin inhibitors market during the forecast period.
According to a report published by the global landscape of autoimmune disease in February 2024, autoimmune diseases cumulatively affect around 5 to 10% of the population in industrially developed countries, and the prevalence of autoimmune disease in developing countries is much lower than in developed countries around the world.
- In May 2024, GSK announced that the company had received positive results for using depemokimab from the phase III clinical trials SWIFT-1 and SWIFT-2. Depemokimab is an interleukin inhibitor that is effective in treating asthma patients.
Restraint
Side effects and high treatment cost
The interleukin inhibitor industry has gained prominent attention due to its capability to treat severe diseases. Although there are several applications of interleukin inhibitors in healthcare sectors, certain problems exist in this industry. Firstly, the overuse of interleukin inhibitors can cause several health issues such as headache, rash, joint pain, nausea, fatigue, gastritis, and some others that retards the use of this therapy in some patients. Secondly, the cost of using interleukin inhibitors is increasing day by day, which prevents poor consumers from using them. Thus, numerous side effects associated with the overuse of interleukin inhibitors, along with the increasing cost of interleukin inhibitors, are expected to restrain the growth of the interleukin inhibitors market during the forecast period.
Opportunity
Advancements in IL-6 and IL-1 inhibitors
Various developments have been taking place in the interleukin inhibitors industry in recent times. The researchers have started developing several varieties of IL inhibitors to treat numerous autoimmune diseases. Recently, IL inhibitor companies have increased their emphasis on developing IL-6 and IL-1 inhibitors. IL-1 inhibitors can be used in the treatment of rheumatoid arthritis, and IL-6 inhibitors are found to be effective against cancer and some inflammatory diseases. Thus, the ongoing advancements associated with IL-6 and IL-1 inhibitors are expected to create ample growth opportunities for the market players in the upcoming days.
- In June 2022, Ferring Pharmaceuticals collaborated with I-Mab. This collaboration is done to develop olamkicept (IL-6 inhibitor) for treating inflammatory bowel disease (IBD) and some other inflammatory conditions.
Segment Insights
Application Insights
The psoriasis segment dominated the market in 2025. The rising cases of plaque psoriasis and inverse psoriasis among the people across the world has driven the market growth. Also, the growing emphasis on developing biologics for managing psoriasis is likely to boost market growth. Moreover, the increasing application of interleukin inhibitors for the treatment of guttate psoriasis and erythrodermic psoriasis is expected to drive the growth of the interleukin inhibitors market during the forecast period.
- According to the National Psoriasis Foundation, the total number of psoriasis patients across the world was 125 million in 2022.
- In October 2023, UCB announced that BIMZELX was approved by the U.S. Food and Drug Administration (FDA) for treating patients suffering from moderate to severe plaque psoriasis.
The psoriatic arthritis segment is expected to grow with the highest CAGR during the forecast period. The increasing cases of psoriatic arthritis among the people of the world have boosted the market growth. Also, the growing adoption of interleukin inhibitors for the treatment of psoriatic arthritis among patients is likely to drive the market growth to some extent. Moreover, the ongoing research and developmental activities associated with IL-23 for the treatment of psoriatic arthritis are expected to propel the growth of the interleukin inhibitors market during the forecast period.
- According to the National Psoriasis Foundation, around 30 % of the patients suffering from psoriasis are expected to develop psoriatic arthritis in their lifetime.
- In August 2022, Johnson & Johnson announced that the U.S. Food and Drug Administration (FDA) approved Stelara. Stelara is an interleukin inhibitor that helps in the treatment of active psoriatic arthritis (PsA) in patients aged more than six years.
Type Insights
The IL-23 segment dominated the interleukin inhibitors market in 2025. The growing demand for IL-23 inhibitors for treating moderate to severe psoriasis among patients has driven the market growth. Also, the rising application of IL-23 inhibitors for the treatment of Crohn's disease and ulcerative colitis is likely to propel the market growth to some extent. Moreover, the increasing use of IL-23 inhibitors for blocking downstream signaling pathways and inhibiting inflammatory responses is expected to boost the growth of the interleukin inhibitor market during the forecast period.
- In June 2024, Johnson & Johnson announced that TREMFYA gained positive results in the pivotal Phase 3 GRAVITI investigational study. TREMFYA is an IL-23 inhibitor that is found effective for treating adult patients suffering from moderate to severe active Crohn's disease.
- In June 2022, AbbVie made an announcement stating that the U.S. Food and Drug Administration (FDA) had approved SKYRIZI. SKYRIZI is an interleukin-23 (IL-23) inhibitor that is used for the treatment of moderately to severely active Crohn's disease (CD).
The IL-17 segment is expected to grow with the highest CAGR during the forecast period. The increasing application of IL-17 inhibitors for blocking the inflammatory pathway of cytokine has driven the market growth. Also, the rising demand for IL-17 inhibitors to improve psoriasis symptoms and help reduce inflammation by autoimmune diseases is likely to boost market growth. Moreover, the growing demand for IL-17 inhibitors for the treatment of plaque psoriasis and psoriatic arthritis is expected to propel the growth of the interleukin inhibitors market during the forecast period.
- In April 2024, ARCA Biopharma announced its collaboration with Oruka Therapeutics. This collaboration is aimed at developing an IL-17A/F inhibitor named ‘ORKA-002' for the treatment of chronic skin diseases.
- In September 2022, ACELYRIN announced an investment of US$ 300 million in developing an IL-17 inhibitor for the treatment of psoriatic arthritis (PsA) and axial spondyloarthritis (AxSpA).
Regional Insights
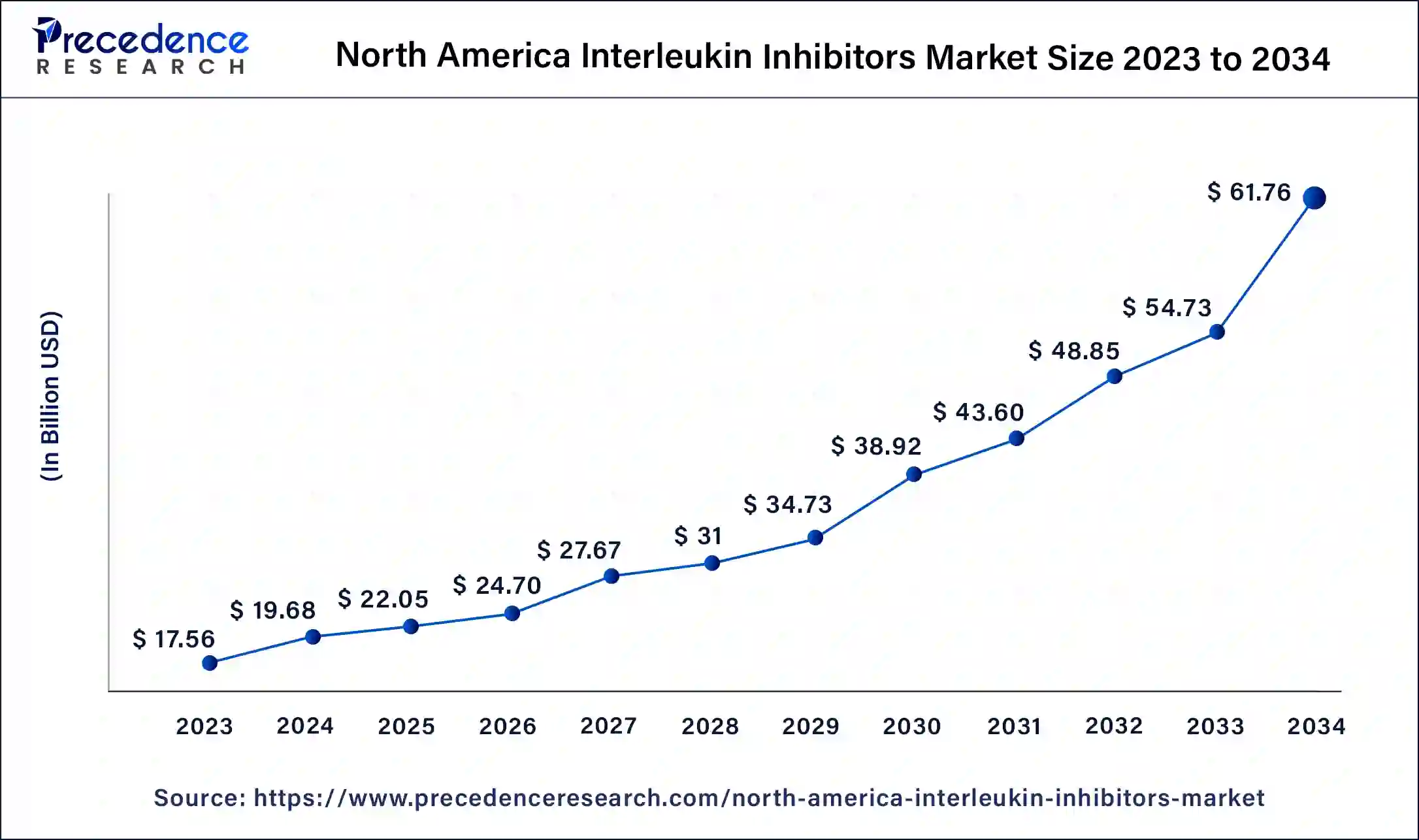
What is the North America Interleukin Inhibitors Market Size?
The North America interleukin inhibitors market size was exhibited at USD 22.05 billion in 2025 and is projected to be worth around USD 68.02 billion by 2035, poised to grow at a CAGR of 11.92% from 2026 to 2035.
North America held the largest share of the interleukin inhibitors market in 2025. The growth of the market in this region is mainly driven by the rising advancements in the healthcare sector along with growing investment by the government in countries such as the U.S. and Canada for developing the pharmaceutical industry. The cases of various types of cancers, such as breast cancer, bladder cancer, kidney cancer, prostate cancer, lymphoma, and some others, have increased across the North American region, increasing the demand for IL-6 inhibitors that in turn increases the demand for interleukin inhibitors, thereby driving the market growth.
The rising prevalence of neurological disorders such as epilepsy, autism, ADD, learning disabilities, neuromuscular disorders, brain tumors, cerebral palsy, and some others has increased in the North American region, which increases the demand for IL-6 inhibitors for treatment, which in turn drives the market growth to some extent. Moreover, several local market players of interleukin inhibitors, such as Johnson and Johnson, Abbvie, Eli Lily, and some others, are constantly engaged in developing interleukin inhibitors and adopting strategies such as partnerships, acquisitions, launches, and business expansions, which in turn drives the growth of the interleukin inhibitors market in this region.
- In March 2023, the government of Canada announced an investment of around US$ 59 million under the Canadian Critical Drug Initiative to boost drug research, commercialization, and manufacturing of pharma products in the Alberta province.
- In July 2023, Eli Lily announced the acquisition of Dice Therapeutics. This acquisition is done at a price tag of US$ 2.4 billion, and it aims to develop advanced interleukin inhibitors-based oral drugs to treat chronic autoimmune and inflammatory diseases.
Asia Pacific is expected to be the fastest-growing region during the forecast period. Rising investments in the healthcare system and growing advancements in the biopharmaceutical sector in countries such as Japan, China, India, South Korea, Israel, and others are expected to drive market growth to some extent. Moreover, the increasing prevalence of cardiovascular diseases increases the demand for interleukin inhibitor-based therapies for effective treatment, thereby driving market growth. For instance, in September 2023, the WHO (World Health Organisation) announced that around 3.9 million people in the Southeast Asia region die due to cardiovascular disease every year.
Furthermore, various local companies producing interleukin inhibitors, such as Teva, Sun Pharma, Sunshine Guojian, Chugai Pharmaceutical Co., Ltd., and some others, are developing advanced interleukin inhibitors across the Asia-Pacific region, which is expected to drive the growth of the interleukin inhibitors market in this region.
The government of India's Department of Health and Family Welfare announced that in FY 2024-25, the government is likely to invest around Rs 87,656.90 crores in developing the healthcare sector in India, an increase of around 12.93% from the revised investment of Rs 77,624.79 crores in FY 2023-2024.
- In August 2022, Chugai Pharmaceutical Co., Ltd. launched Mitchga. Mitchga is an anti-IL-31 receptor that helps in the treatment of itching associated with atopic dermatitis.
Value Chain Analysis of the Interleukin Inhibitors Market
- R&D: Discovery of high-cost discovery of select cytokines such as IL-17 and IL-23, which are inhibitors of pathways of autoimmune disease in inflammatory processes.
Key players: Novartis, AbbVie, Eli Lilly - Clinical Trials and Regulatory Approvals: Intensive testing covers both safety and effectiveness, and then the approvals are vetted by the health authority.
Key players: IQVIA, ICON plc, PPD (Thermo Fisher Scientific) - Preparation of Formulation and Final Dosage: Precision engineering involves assembling the biological components into injectable or oral pharmaceuticals, which are stable.
Key players: Lonza, Catalent, Samsung Bioepis
Interleukin Inhibitors Market Companies
- Johnson and Johnson Services, Inc.: It provides Stelara and Tremfya as IL pathway treatment against psoriasis, psoriatic arthritis, and inflammatory bowel diseases.
- Novartis AG: It offers Cosentyx, an IL-17A inhibitor that treats plaque psoriasis, psoriatic arthritis, and ankylosing spondylitis by targeting immune inflammation.
- AbbVie Inc.: Provides Skyrizi, which is an IL-23 p19 subunit based on selective inhibition of inflammatory pathways in the treatment of psoriasis and Crohn's conditions.
Other Major Key Players
- Eli Lilly and Company
- Regeneron Pharmaceuticals, Inc.
- AstraZeneca
- Bausch Health
- F. Hoffmann-La Roche Ltd
- GlaxoSmithKline plc.
- Teva Pharmaceuticals Industries, Ltd.
Recent Developments
- In March 2025, four ustekinumab alternatives exist, and two more are entering the market, increasing access to cost-effective treatments compared to Janssen's Stelara. Celltrion is working with pharmacy benefit managers to improve formulary coverage for its new product, Steqeyma. (https://www.centerforbiosimilars.com )
- In April 2024, ImmunityBio, Inc. announced that the U.S. Food and Drug Administration (FDA) had approved ANKTIVA. ANKTIVA is an interleukin inhibitor therapy that is found effective for the treatment of BCG-unresponsive non-muscle invasive bladder cancer (NMIBC).
- In March 2024, Boehringer Ingelheim announced that the US Food and Drug Administration had approved SPEVIGO. SPEVIGO is an interleukin inhibitor used to treat generalized pustular psoriasis (GPP).
- In February 2024, Sanofi announced that the U.S. Food and Drug Administration (FDA) had approved Dupixent (dupilumab). Dupixent is an interleukin inhibitor used to treat patients with uncontrolled chronic obstructive pulmonary disease (COPD).
- In December 2023, LEO Pharma Inc. made an announcement stating that the U.S. Food and Drug Administration (FDA) had approved Adbry. Adbry is recognized as the first treatment for patients suffering from moderate-to-severe atopic dermatitis.
- In October 2023, Eli Lilly and Company announced that the U.S. Food and Drug Administration (FDA) had approved Omvoh. Omvoh is an interleukin-23p19 injection that helps in the treatment of active ulcerative colitis (UC) in adults.
- In October 2023, Novartis announced that the U.S. Food and Drug Administration (FDA) had approved Cosentyx. Cosentyx is an interleukin-17A inhibitor-based therapy that is found effective for the treatment of moderate to severe hidradenitis suppurativa (HS).
Segments Covered in the Report
By Type
- IL-17
- IL-23
- IL-1
- IL-5
- IL-6
- Others
By Application
- Psoriasis
- Psoriatic Arthritis
- Rheumatoid Arthritis
- Asthma
- Inflammatory Bowel Disease (IBD)
- Others
By Geography
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
