What is the MRI Safe Implantable Defibrillator Devices Market Size?
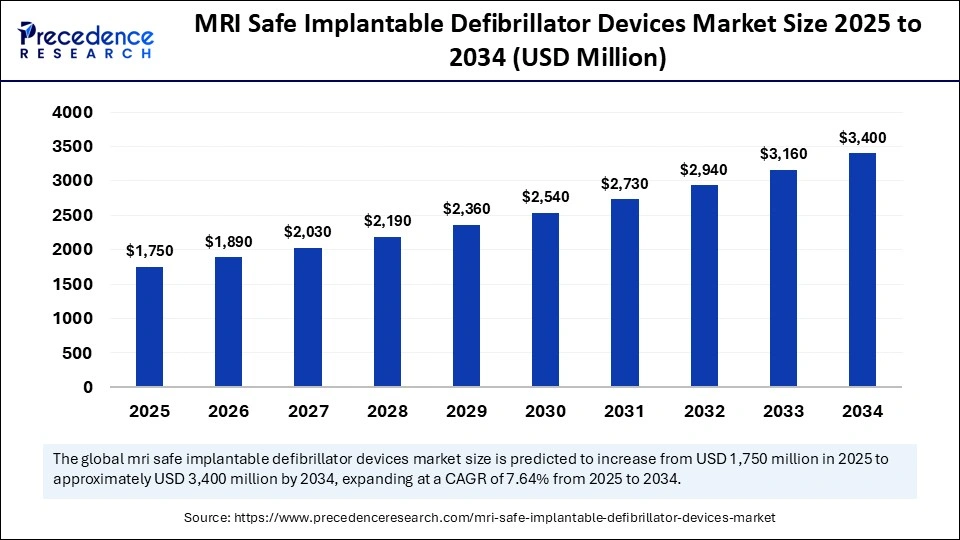
The global MRI safe implantable defibrillator devices market size accounted for USD 1,750 million in 2025 and is predicted to increase from USD 1,890 million in 2026 to approximately USD 3,400 million by 2034, expanding at a CAGR of 7.64% from 2025 to 2034. The market for MRI safe implantable defibrillator devices is driven by rising cardiac disorders, increasing MRI procedures, and technological advancements in safer, more efficient implantable devices.
Market Highlights
- North America led the MRI safe implantable defibrillator devices market with around 40% of share in 2024.
- The Asia Pacific is expected to grow the fastest CAGR between 2025 and 2034.
- By product type, the single-chamber ICDs segment accounted for the highest market share of 35% in 2024.
- By product type, the biventricular / CRT-D ICDs segment is growing at a fastest CAGR between 2025 and 2034.
- By battery technology, the lithium-ion segment captured approximately 70% of the market share in 2024.
- By battery technology, the lithium silver vanadium oxide segment is growing at the fastest CAGR from 2025 to 2034.
- By end-user/treatment setting, the hospitals / cardiac centers segment captured approximately 41.6% of market share in 2024.
- By end-user/treatment setting, the specialty clinics segment is expanding at a notable CAGR between 2025 and 2034.
Drive for MRI-Compatible Defibrillators by Safer Heart Solutions
The rising cases of patients with cardiovascular diseases, as well as the increasing demand for imaging procedures with compatibility considerations, continue to drive demand for MRI safe implantable defibrillator devices that detect and correct abnormal heart rhythms and can be safely used in patients who may undergo magnetic resonance imaging. MRI-conditional defibrillators are similar to standard defibrillator devices but utilize unique circuit and material designs that limit electromagnetic interference with imaging.
Demand for MRI safe defibrillator devices is gaining traction among cardiac care centers adopting these devices, as well as among developers of miniaturized devices, and among post-approval health systems favoring devices with supportive uses. Given patient safety concerns and diagnostic accuracy, manufacturers are working to develop exciting innovations that deliver therapeutic efficacy and MRI compatibility, thereby creating an important reference point for the development of cardiac rhythm management technology.
AI and Digital Innovation Transforming Medical Device Development
The MRI safe implantable defibrillator market is flourishing as manufacturers integrate advanced biocompatible materials, miniaturized circuitry, and improved shielding techniques for better stability in powerful magnetic fields. The development toward leadless and subcutaneously positioned devices presents less risk for infection and fewer complications during procedures. In improving device battery chemistry and power management, improved safety and reliability, and longevity leave the indwelling defibrillator caring for the patient longer with more dependability.
Systems based on AI-generated diagnostics enable personalized therapy adaptation, improving both patient safety with issues related to delivery and therapy accuracy. These advancements in device technology lead to increased use of MRI compatible devices for either preventative therapy or post-cardiac event treatment - either close to the implant site or remote from the implant site allowing for better imaging diagnostics without sacrificing device performance. On-going Research and Development efforts in material science to mitigate electromagnetic interference will continue to develop new designs in cardiac implant technology.
- In March 2025, Medtronic Japan Co., Ltd. announced the official approval and launch of the Aurora EV ICD MRI system, with the Epsila EV MRI Lead, in Japan, an extravascular, implantable cardioverter-defibrillator with MRI compatibility at both 1.5 T and 3 T.
Five Healthcare Trends Shaping 2025 and Beyond
- Increasing Trend of MRI-Compatible Cardiac Devices: Manufacturers are prioritizing MRI safe defibrillators in response to the increasing demand for safer imaging protocols in patients with heart disease. The healthcare system is conducting more MRI procedures in patients with heart disease, and the technology for shielding devices has also advanced.
- Convergence of Remote Monitoring with AI-Driven Diagnostics: Next-generation MRI safe defibrillators utilize AI-driven algorithms and remote patient monitoring capabilities. These capabilities support clinical decision-making, detect arrhythmias, and enhance progress and outcomes with patient-reported real-time cardiac data.
- Storage Minimization and Longer Battery Life: Manufacturers are focusing on minimizing the design of devices to conserve battery life. Reducing storage minimizes replacements, enhances comfort for patients, and improves MRI device compatibility with minimal impact on functionality.
- Increased Clinical Adoption in Aging Populations: Increased incidence and prevalence of cardiovascular diseases among older patients are contributing to a rise in demand for MRI safe implantable defibrillators. Hospitals are embracing MRI-capable systems as part of comprehensive cardiac care protocols in geriatric populations.
MRI Safe Implantable Defibrillator Devices Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1,750 Million |
| Market Size in 2026 | USD 1,750 Million |
| Market Size by 2034 | USD 3,400 Million |
| Market Growth Rate from 2025 to 2034 | CAGR of 7.64% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Product Type, Battery Technology, End-User/Treatment Setting, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
MRI Safe Implantable Defibrillator Devices Market Segment Insights
[[segment_insights]]
MRI Safe Implantable Defibrillator Devices Market Regional Insights
[[regional_insights]]
MRI Safe Implantable Defibrillator Devices Market Value Chain

MRI Safe Implantable Defibrillator Devices Market Companies
[[market_company]]
Recent Developments
- In December 2024, Philips announced its initiative to advance cardiac MRI technology through AI-driven research, focusing on enhancing image clarity, diagnostic accuracy, and workflow efficiency to improve cardiovascular care and accessibility for patients worldwide.(Source: https://www.usa.philips.com)
- January 2025, Researchers from Brown University conducted a study on Transvenous implantable cardioverter-defibrillators (ICDs), highlighting their effectiveness in preventing sudden cardiac death and comparing their safety and performance with emerging extravascular ICD systems.(Source: https://www.acc.org)
Exclusive Insights
From an analysts perspective, the market for MRI safe implantable defibrillators has a strong growth opportunity, supported by three impactful trends: an increased incidence of cardiac arrhythmias and SCA in aging populations, increasing MRI diagnostic use and acceptance in cardiac care, and the technical need to ensure that implantable supports their use in imaging. From an opportunity perspective with MRI safe devices, business participants can access higher priced product tiers and new geographic markets where MRI imaging growth is occurring. However, there are some potential pitfalls, regulatory and reimbursement challenges, costs for devices that may limit adoption in price sensitive conditions, and clinician continued concerns over lead-heating/artefact during MRI scanning.
Importantly, the market should continue to chase three trends: integration of remote monitoring into MRI safe ICDs, partnerships between device companies and radiology/imaging centers, and movement away from basic implantable model towards more differentiated devices within MRI safe segment. Overall, from a business /market perspective, companies that receive regulatory approval, build irons an efficient cost model for manufacturing MRI compatible products, and align efforts with imaging workflow stakeholders, should be well positioned to access this premium niche of the broader implantable defibrillator market.
MRI Safe Implantable Defibrillator Devices Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
