What is the Mucopolysaccharidosis Treatment Market Size?
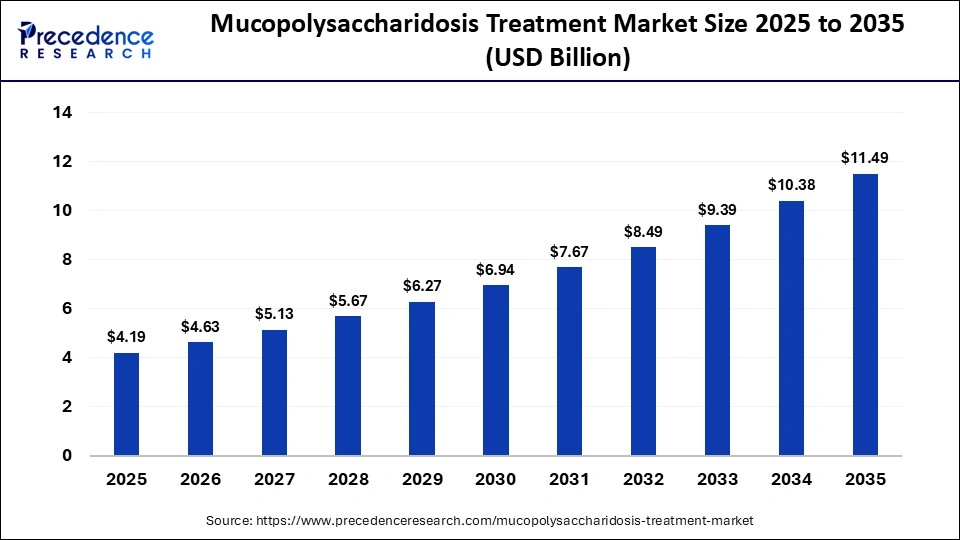
The global mucopolysaccharidosis treatment market size was calculated at USD 4.19 billion in 2025 and is predicted to increase from USD 4.63 billion in 2026 to approximately USD 11.49 billion by 2035, expanding at a CAGR of 10.61% from 2026 to 2035. The market is primarily driven by intensified research efforts, greater awareness of the disease, and supportive regulatory and government healthcare initiatives.
Key Takeaways
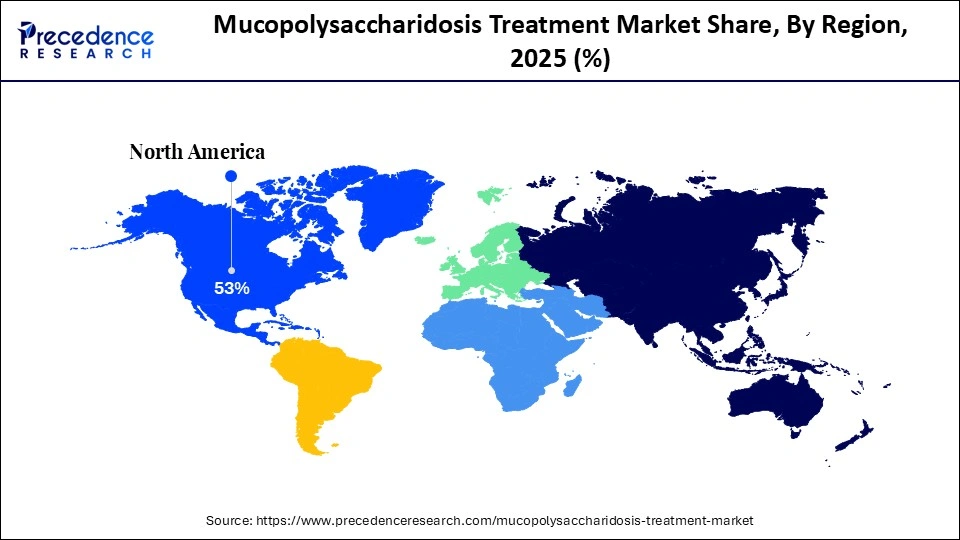
- By region, North America dominated the mucopolysaccharidosis treatment market in 2025.
- By region, Asia Pacific is expected to grow at the fastest rate over the forecast period.
- By therapy type, the enzyme replacement therapy segment accounted for the largest market share in 2025.
- By therapy type, the stem cell therapy segment is anticipated to experience the fastest growth over the forecast period.
- By type of MPS, the MPS I segment held the largest share of the global market in 2025.
- By type of MPS, the MPS II segment is expected to grow at a significant CAGR over the forecast period.
- By end user, the hospitals segment accounted for the largest share of the global market in 2025.
- By end user, the home-infusion segment is projected to grow at a significant rate over the forecast period.
Mucopolysaccharidosis Treatment Market Overview
Mucopolysaccharidoses (MPS) are a group of rare, inherited metabolic disorders that include several subtypes, each presenting a range of physical and neurological symptoms. Treatment approaches for mucopolysaccharidoses differ based on the specific orthopedic conditions present in each patient. For instance, bone marrow transplantation is used for Hurler syndrome, while surgical interventions such as osteotomies are performed to correct bone alignment. Guided growth procedures are used to address lower-extremity misalignment, including knock knees.
In addition, in cases of cervical instability and underdevelopment, posterior spinal fusion is performed to correct kyphosis. Clinicians and specialists are progressively moving toward disease-modifying interventions, which are reshaping prescription practices across major healthcare systems and driving the market.
Impact of Artificial Intelligence on the Mucopolysaccharidosis Treatment Market
Artificial intelligence is playing an important role in data‑driven healthcare research, and its use in diagnosing mucopolysaccharidosis (MPS) is steadily growing. In a recent study, researchers applied a supervised machine learning algorithm, Classification and Regression Trees (CART), to classify MPS subtypes using glycosaminoglycan (GAG) biomarker profiles. To improve interpretability, explainable AI methods such as SHAP (SHapley Additive exPlanations) were used to assess feature-level importance, revealing how specific biomarkers influenced model predictions.
In parallel, graph-based modeling approaches have also been explored in MPS research to analyze patient similarity networks. In such frameworks, graph neural networks (GNNs) can be used, and explanation tools like PG Explainer help provide insights into how patient-to-patient relationships influence classification outcomes. These explainable AI components provide complementary interpretability by clarifying both diagnostic feature contributions and relational patterns among patients.
Early diagnosis of mucopolysaccharidosis is crucial, as it significantly improves patient response to treatment and reduces the risk of complications or mortality. Researchers supported by APMAD have developed a machine-learning-based screening approach using UAE health records to accelerate early identification. The study proposes a cost-effective, non-invasive diagnostic support tool that leverages real-world electronic health record (EHR) data and advanced AI techniques to assist clinicians in identifying suspected cases earlier and supporting treatment planning.
Mucopolysaccharidosis Treatment Market Trends
- Rising focus on blood-brain barrier penetrating therapies for treating neurological manifestations of Mucopolysaccharidosis, improving drug delivery to the central nervous system, and expanding therapeutic potential for rare lysosomal storage disorders.
- Growing research focus on glycosaminoglycan pathways is improving therapeutic strategies and enhancing understanding of disease progression in mucopolysaccharidosis treatment.
- Research into small molecule drugs and substrate reduction therapies (SRTs) is growing to complement or provide alternatives to ERT, especially for hard‑to‑treat symptom areas.
- Growing emphasis on quality of life, caregiver support, and long‑term follow‑up assessments is shaping treatment decisions and care frameworks.
- Biotech and pharma companies are increasingly forming collaborations with research institutions and patient advocacy groups to accelerate R&D and broaden treatment access.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 4.19 Billion |
| Market Size in 2026 | USD 4.63 Billion |
| Market Size by 2035 | USD 11.49 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 10.61% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Therapy type, Type of MPS, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Analysis
Therapy Type Insights
How Did the Enzyme Replacement Therapy Segment Lead the Market?
The enzyme replacement therapy segment led the mucopolysaccharidosis treatment market in 2025, driven by its established efficacy in reducing glycosaminoglycan accumulation in somatic tissues. ERT involves periodic intravenous administration of disease-specific enzymes produced using recombinant DNA technology. The therapy primarily targets non–central nervous system symptoms and has shown improvements in liver size, respiratory function, mobility, and joint flexibility, particularly when initiated early.
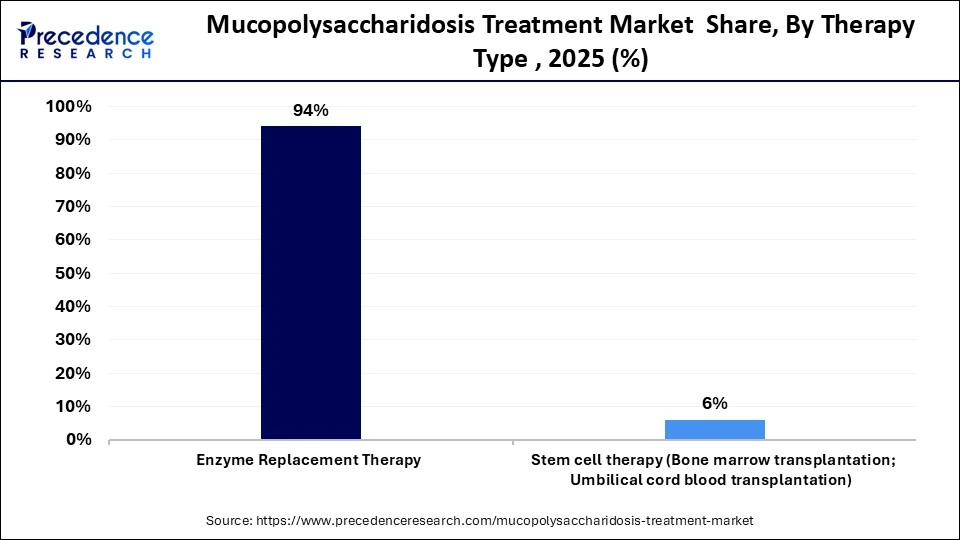
This therapy is approved in the U.S., the European Union, and several other countries for patients with mucopolysaccharidosis types I, II, IIIA, VI, and VII. Researchers are also exploring combinations of enzyme replacement therapy with blood-brain barrier-crossing technologies to address neurological manifestations. Its established therapeutic effectiveness, together with ongoing advances in clinical research, continues to reinforce its position as a central treatment option in the market.
The stem cell therapy segment is projected to grow rapidly in the upcoming period, driven by emerging clinical evidence and its potential to address both somatic and neurological manifestations of the disease. Within this approach, hematopoietic stem cell transplantation, including bone marrow transplantation and umbilical cord blood transplantation, is gaining attention, particularly for mucopolysaccharidosis types I and VII, especially in pediatric patients diagnosed early.
This approach is widely used for Hurler syndrome, where donor stem cells produce the deficient enzyme α-L-iduronidase. The activity of this enzyme decreases the buildup of substrates in body tissues and leads to better clinical outcomes. Umbilical cord blood has emerged as a promising stem cell source for transplantation in several genetic disorders. Since MPS type I is a progressive multisystem disorder, patients receiving transplantation early in the disease course often demonstrate improved outcomes, supporting the adoption of these stem cell therapies.
Type of MPS Insights
Why Does the MPS I Segment Dominate the Market?
The MPS I segment dominated the mucopolysaccharidosis treatment market in 2025 because it is the most prevalent and well-characterized form of the disease. The most severe manifestation of MPS I is Hurler syndrome, which affects approximately 1 in 100,000 newborns and occurs equally in males and females. Comparatively milder forms of MPS I occur in nearly 1 out of every 500,000 newborns, contributing to the segment's sustain dominance.
MPS I is a rare, inherited metabolic disorder caused by a deficiency of the enzyme α-L-iduronidase, resulting in the accumulation of complex sugars in body tissues. This progressive buildup can affect multiple organs and is associated with skeletal deformities, vision and hearing impairment, and cognitive decline. Aldurazyme remains the most widely used therapy for MPS I as an enzyme replacement treatment. It supplies the missing enzyme to facilitate the breakdown of accumulated glycosaminoglycans, helping manage disease manifestations and supporting treatment adoption in the segment.
The MPS II segment is projected to grow at the fastest rate during the forecast period due to increased diagnostic capabilities that enable earlier detection. MPS II, also known as Hunter syndrome, is a rare genetic disorder with an incidence rate ranging from 0.38 to 1.09 per 100,000 live male births. It is the only X-linked inherited form of mucopolysaccharidosis and therefore primarily occurs in boys. Hunter syndrome is classified as a lysosomal storage disorder and is caused by variations in the IDS gene, which provides instructions for producing the IDS enzyme.
Multiple therapeutic approaches are available for the management of MPS II, including enzyme replacement therapy, hematopoietic stem cell transplantation, substrate reduction therapy, and gene therapy. Early treatment has demonstrated positive clinical outcomes, as patients who receive therapy early in life are often able to achieve motor milestones such as sitting and walking. These favorable outcomes are expected to support the growth of this treatment segment.
End User Insights
How Does the Hospital Segment Lead the Market?
The hospitals segment led the mucopolysaccharidosis treatment market with the largest share in 2025, primarily due to their advanced infrastructure for administering long-term infusion therapies and conducting specialized genetic diagnostics. The dominance of this segment is closely linked to the need for multidisciplinary care teams, including metabolic specialists, geneticists, orthopedic surgeons, and pediatricians, who are essential for managing the multisystem complications of the disease. Hospitals also serve as key centers for early diagnosis, particularly among newborn and pediatric patients. In addition, their clinical infrastructure enables effective monitoring and management of infusion-related reactions, reinforcing their critical role in treatment delivery and supporting segment growth.
The home infusion segment is projected to grow at a significant rate over the forecast period. In this approach, medications are delivered directly to the patient's home, and a trained nurse administers the infusion while remaining in close coordination with healthcare specialists. Many patients require regular infusion therapies to manage lifelong conditions, and receiving treatment at home is often considered more convenient and comfortable than visiting an infusion center. Reduced hospital visits and continuous support from home care nurses further enhance patient convenience. In addition, studies indicate that home infusion therapy can be as safe and effective as treatments administered in clinical infusion centers, supporting the expansion of this segment.
Regional Insights
How Big is the North America Mucopolysaccharidosis Treatment Market Size?
The North America mucopolysaccharidosis treatment market size is estimated at USD 2.22 billion in 2025 and is projected to reach approximately USD 6.15 billion by 2035, with a 10.73% CAGR from 2026 to 2035.
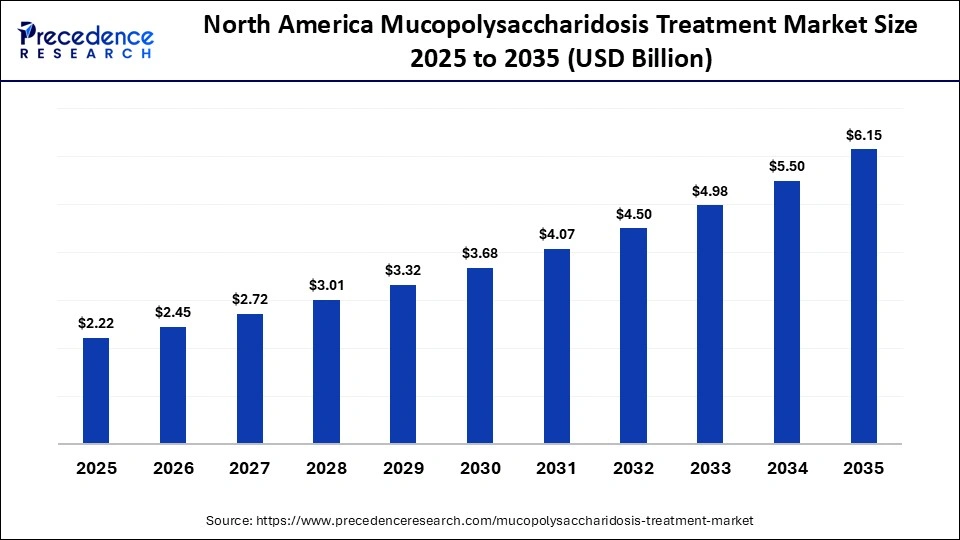
What Made North America the Dominant Region in the Mucopolysaccharidosis Treatment Market?
North America dominated the mucopolysaccharidosis treatment market in 2025, accounting for the largest share. This leadership is driven by a mature healthcare ecosystem, early adoption of advanced medical technologies, and ongoing transformation initiatives. The region's position in the market is further supported by well-established healthcare infrastructure, significant R&D investments, and favorable regulatory pathways.
What is the Size of the U.S. Mucopolysaccharidosis Treatment Market?
The U.S. mucopolysaccharidosis treatment market size is calculated at USD 1.67 billion in 2025 and is expected to reach nearly USD 4.64 billion in 2035, accelerating at a strong CAGR of 12.02% between 2026 to 2035.
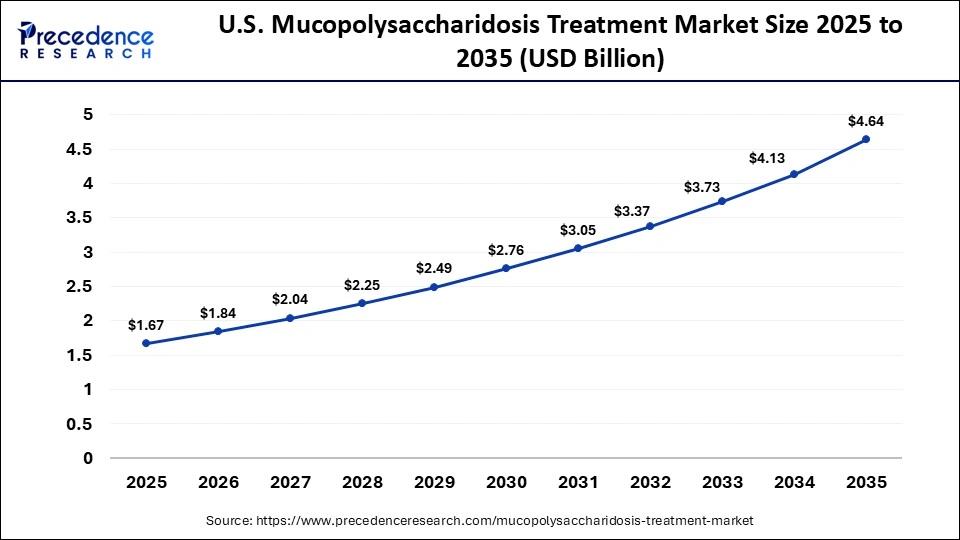
The U.S. remains at the forefront due to its proactive rare disease research ecosystem and the availability of multiple FDA-approved therapies. A majority of transplanted patients are reported from North America, reflecting its advanced clinical capabilities and access to specialized treatment options. In addition, sustainability trends are reshaping the healthcare landscape, driving demand for energy-efficient, environmentally compliant, and optimized solutions.
Within North America, the U.S. represents the largest share of the market, while Canada also contributes meaningfully to regional growth through increasing awareness, improved diagnostic capabilities, and expanding access to treatment options. Moreover, Latin America offers significant growth potential through infrastructure development and mounting industrial activity. These factors collectively support ongoing growth in healthcare outcomes and market expansion across the region.
How is the Opportunistic Rise of Asia Pacific in the Mucopolysaccharidosis Treatment Market?
Asia Pacific is expected to grow at the fastest CAGR in the market, driven by improving healthcare infrastructure, expanding rare disease awareness, and increasing access to advanced therapies across China, India, and Southeast Asian economies. At the same time, governments in several developing nations are introducing initiatives to support rare disease funding, improving access to essential treatments such as enzyme replacement therapy. Rising awareness of rare diseases and the availability of cost-effective treatment options are further drawing attention to rare disease management across the region.
Within the region, Japan remains a leader in drug development for rare diseases, underpinned by strong research and development capabilities. China is experiencing rapid growth, supported by increasing healthcare expenditure and government initiatives focused on rare disease treatment. India is also showing promising potential due to its expanding generic pharmaceutical sector and collaborations for innovative therapies. Meanwhile, emerging Southeast Asian markets such as Indonesia and Malaysia are gradually increasing their focus on rare disease therapies, contributing to the overall expansion of the regional market.
What are the Advancements in the Mucopolysaccharidosis Treatment Market in Europe?
Europe is expected to have significant growth during the forecast period, driven by a well-established healthcare system and strong regulatory support for rare diseases. Countries such as Germany, France, and the UK are leading players as they have been at the forefront of rare disease research and treatment. The European Union's Orphan Drug Regulation also provides incentives for the development of therapies for rare diseases, which thus leads to market exclusivity and financial support.
Germany Market Trends
The region benefits from a supportive regulatory framework, which helps in fostering innovation in the MPS treatment market. The market is expected to witness steady growth, supported by ongoing advancements in treatment options and increasing awareness about MPS.
What are the Key Trends in the Mucopolysaccharidosis Treatment Market in Latin America?
Latin America is expected to have significant growth in the market. This growth is due to the rising prevalence of MPS, coupled with improving healthcare infrastructure and growing awareness about rare diseases, which is driving market growth in this region. Countries such as Brazil and Argentina are leading players as they are witnessing increased investment in healthcare and biotechnology.
Brazil Market Trends
The region benefits from favorable government initiatives and funding for rare disease research, which helps in propelling market growth. This reflects the region's potential for market expansion.
How is the Middle East and Africa Region Growing in the Mucopolysaccharidosis Treatment Market?
The Middle East and Africa region is anticipated to witness significant growth in the market. This growth and development are driven by various factors such as developing healthcare infrastructure, increased government initiatives, and growing awareness of rare disorders. Countries such as Saudi Arabia, the UAE, and South Africa are leading players in the region.
Saudi Arabia Market Trends
Factors such as a higher awareness among the patient population towards advanced treatment options, increasing diagnostic sophistication, the presence of significant clinical trials, and the presence of major clinical-stage biopharmaceutical companies are driving the country's market landscape.
Mucopolysaccharidosis Treatment Market Companies
- Ultragenyx Pharmaceutical Inc.: Ultragenyx Pharmaceutical Inc. develops treatments for rare and ultra-rare genetic diseases, aiming its Mucopolysaccharidosis (MPS) portfolio on enzyme replacement therapy and also adeno-associated virus (AAV) gene therapies, aiming at highly specific patient populations.
- JCR Pharmaceuticals Co., Ltd.: JCR Pharmaceuticals Co., Ltd. is a global specialty pharmaceutical firm known for its groundbreaking therapies for rare lysosomal storage disorders, which include MPS. Their main offering in this space involves an approved drug for MPS II and an expanding pipeline of investigational drugs for a few types of MPS.
- REGENXBIO Inc.: REGENXBIO provides investigational, one-time gene therapies designed to treat the neurological and even systemic manifestations of MPS Type I and Type II. Their approach utilizes a proprietary adeno-associated virus (AAV) vector platform to deliver functional genes directly to the central nervous system (CNS).
- Sangamo Therapeutics, Inc.: Sangamo Therapeutics provides a potential one-time in vivo genome editing therapy. Sangamo's approach aims to permanently correct the underlying genetic cause and also allow a patient's liver to produce its own corrective enzymes.
- Denali Therapeutics Inc.: Denali Therapeutics Inc. provides AVLAYAH, an FDA-approved Enzyme Replacement Therapy for MPS Type II, also called Hunter syndrome. It is the first therapy specifically designed to tackle the neurological manifestations of the disease by crossing the blood-brain barrier.
Other Major Key Players
- BioMarin Pharmaceutical Inc.
- Takeda Pharmaceutical Company Limited
- Sanofi (Sanofi Genzyme)
- Abeona Therapeutics Inc.
- GC Pharma (Green Cross Corporation)
- Sarepta Therapeutics, Inc.
- Orchard Therapeutics
- Immusoft Corporation
- ArmaGen, Inc.
- Amicus Therapeutics, Inc.
Recent Developments
- In 2025, at ICIEM 2025, JCR Pharmaceuticals Co., Ltd. presented new data from its pipeline of brain-penetrant enzyme replacement therapies at the 15th International Congress of Inborn Errors of Metabolism (ICIEM) in Kyoto, Japan, held from September 2–6, 2025. Highlights included five-year clinical data demonstrating sustained neurocognitive and somatic benefits of Pabinafusp alfa (JR-141) in patients with mucopolysaccharidosis type II (MPS II, or Hunter syndrome), suggesting improvements in patients treated before significant neuronopathy and stabilization in those with advanced disease.
(Source: https://www.businesswire.com) - In 2025, MEDIPAL HOLDINGS CORPORATION (TSE 7459, “MEDIPAL”) and JCR Pharmaceuticals Co., Ltd. (TSE 4552, “JCR”) announced that the European Commission (EC) has granted orphan drug designation (ODD) to JR-446, an investigational therapy for mucopolysaccharidosis type IIIB (MPS IIIB, or Sanfilippo syndrome type B), following its recent designation by the U.S. Food and Drug Administration (FDA).
(Source: https://www.businesswire.com) - In 2025, Denali Therapeutics announced that the U.S. Food and Drug Administration (FDA) extended its review of the Biologics License Application (BLA) for Tividenofusp alfa, a treatment for mucopolysaccharidosis type II (MPS II), also known as Hunter syndrome.
(Source: https://www.globenewswire.com) - In 2025, REGENXBIO Inc. sannounced that the U.S. Food and Drug Administration (FDA) had accepted for review its Biologics License Application (BLA) seeking accelerated approval of clemidsogene lanparvovec (RGX-121) for the treatment of mucopolysaccharidosis type II (MPS II), also known as Hunter syndrome.
(Source: https://www.prnewswire.com)
Segments Covered in the Report
By Therapy Type
- Enzyme Replacement Therapy
- Stem cell therapy (Bone marrow transplantation; Umbilical cord blood transplantation)
By Type of MPS
- MPS I
- MPS II
- MPS IV A
- MPS VI
- MPS VII
By End User
- Hospital
- Specialty Clinics
- Medical Research Centers
- Home-infusion
By Region
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
