What is the Nasal Polyposis Treatment Market Size?
The global nasal polyposis treatment market size accounted for USD 5.27 billion in 2025 and is predicted to increase from USD 5.61 billion in 2026 to approximately USD 9.15 billion by 2034, expanding at a CAGR of 6.32% from 2025 to 2034. This market is growing due to the rising prevalence of chronic rhinosinusitis and the increasing adoption of biologic therapies for effective term management.
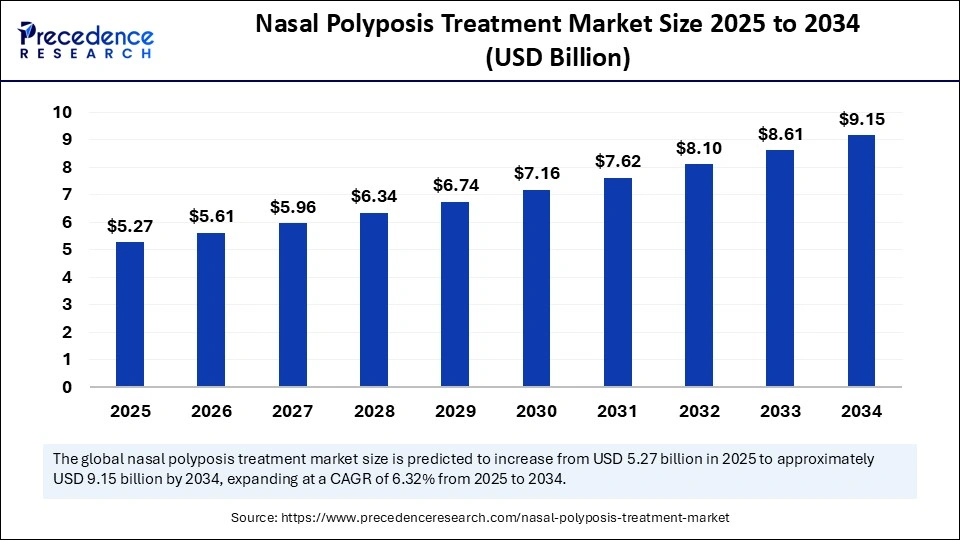
Market Highlights
- North America dominated the market, holding the largest market share of 34% in 2024.
- The Asia Pacific is expected to grow at a notable rate from 2025 to 2034.
- By treatment modality, the pharmacotherapy devices segment held the largest market share of 55% in 2024.
- By treatment modality, the biologic therapies segment is growing at a strong CAGR between 2025 and 2034.
- By drug class, the corticosteroids segment contributed more than 45% of market share in 2024.
- By drug class, the biologics segment is expected to grow at the fastest rate from 2025 to 2034
- By route of administration, the intranasal segment held the major market share of 45% in 2024.
- By route of administration, the subcutaneous injection segment is growing at a solid CAGR between 2025 and 2034.
- By patient segment/severity, the moderate CRSwNP segment accounted for the highest market share of 35% in 2024.
- By patient segment/severity, the severe/refractory CRSwNP segment is poised to grow at a healthy CAGR from 2025 to 2034.
- By prescriber/end user, the ENT/otolaryngology clinics & specialists segment led the nasal polyposis treatment market with a 45% share in 2024.
- By prescriber/ed user, the primary care/general practice segment is expected to grow at the fastest rate between 2025 and 2034.
Market Overview
Breathing Easier: How Biologics Are Transforming the Nasal Polyposis Treatment Market?
The nasal polyposis treatment market is witnessing steady growth as the number of people worldwide suffering from chronic rhinosinusitis with nasal polyps continues to rise. Adoption is being accelerated by growing knowledge of biologic treatments and their effectiveness in lowering inflammation and recurrence rates. Additionally, pharmaceutical companies are improving patient outcomes and treatment options through R&D investments.
Case Study: In Oct 2025, Amgen and AstraZeneca announced FDA approval of Tezspire (tezepelumab-ekko) as an add-on treatment for patients aged 12 and above with chronic rhinosinusitis with nasal polyps.
Key Technological Shifts in the Nasal Polyposis Treatment Market
- Rise of Biologic Therapies: By targeting the underlying inflammatory pathways (IL-4, IL-5, and IgE), targeted biologics like dupilumab, omalizumab, and Tezepelumab are transforming the treatment of nasal polyposis. The need for corticosteroids and surgery is greatly decreased by these medications.
- Precision Medicine & Biomarker-Based Treatment: Advances in molecular diagnostics are enabling the identification of specific biomarkers to tailor therapy for individual patients, improving treatment success rates and minimizing side effects.
Key Trends
- Rising Use of Biologics: Targeted biologics are replacing steroids and surgery, offering long-term relief with fewer side effects.
- Preference for Non-Surgical Options: Patients increasingly opt for medical therapies due to faster recovery and reduced recurrence.
- Expanding Clinical Pipelines: Leading pharma companies are advancing monoclonal antibody research for chronic rhinosinusitis with nasal polyps.
- Focus on Personalized Care: Precision medicine using biomarkers is enabling more tailored and effective treatments.
Nasal Polyposis Treatment Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 5.27 Billion |
| Market Size in 2026 | USD 5.61 Billion |
| Market Size by 2034 | USD 9.15 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 6.32% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Treatment Modality, Drug Class, Route of Administration, Patient Segment / Severity, Prescriber / End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Nasal Polyposis Treatment Market Segments Insights
[[segment_insights]]
Nasal Polyposis Treatment Market Regional Insights
[[regional_insights]]
Nasal Polyposis Treatment Market Value Chain
[[value_chain]]
Nasal Polyposis Treatment Market Companies
[[market_company]]
Other Players in the Market
- GlaxoSmithKline (GSK): GSK is a global biopharma company that integrates science, technology, and advanced R&D to develop specialty medicines and vaccines. Its respiratory portfolio includes innovative treatments for chronic rhinosinusitis, asthma, and allergic conditions, leveraging biologics and inhalation technologies.
- Novartis: Novartis focuses on breakthrough therapies and advanced biologics, emphasizing precision medicine in respiratory, immunology, and chronic inflammatory diseases. The companys strategy combines deep scientific expertise with digital innovation to improve patient outcomes in airway and allergy-related disorders.
- F. Hoffmann-La Roche / Genentech: Roche and its U.S. biotechnology subsidiary Genentech are global leaders in pharmaceuticals and diagnostics, developing targeted biologic therapies for respiratory and immunological conditions. Their pipeline includes monoclonal antibodies and precision therapies addressing inflammation and tissue repair.
- AstraZeneca: AstraZeneca is a major player in respiratory and immunology therapeutics, with a portfolio addressing asthma, COPD, and chronic rhinosinusitis. The companys biologic Fasenra (benralizumab) and pipeline therapies target Type 2 inflammation, reflecting its leadership in precision respiratory care.
- OptiNose (OptiNose US): OptiNose develops nasal delivery-based therapeutics for upper airway diseases, including chronic rhinosinusitis with nasal polyps (CRSwNP). Its product XHANCE (fluticasone propionate) uses exhalation delivery technology to enhance drug deposition and efficacy in sinonasal disorders.
- Intersect ENT: Intersect ENT specializes in localized drug delivery implants for sinus and nasal inflammatory conditions. Its PROPEL and SINUVA steroid-releasing implants improve postoperative outcomes and reduce recurrence in patients with chronic rhinosinusitis, bridging pharmaceuticals and minimally invasive devices.
- Teva Pharmaceutical Industries: Teva offers a broad range of respiratory and immunology medicines, including inhalation therapies and biologics. Its focus on affordable treatment access and biosimilars supports management of chronic airway and allergic disorders globally.
- Merck & Co.: Merck develops immunomodulators, anti-inflammatory drugs, and vaccines addressing respiratory and allergic diseases. The company leverages advanced monoclonal antibody and immunotherapy platforms to develop targeted biologics for chronic rhinosinusitis and asthma.
- Pfizer : Pfizer portfolio includes anti-inflammatory biologics and immunotherapies addressing upper respiratory and allergic conditions. The company is expanding into precision inflammation medicine through mRNA and biologic innovation for respiratory health.
- Johnson & Johnson (Janssen): Janssen Pharmaceuticals develops biologic therapies targeting immune-mediated diseases, including nasal polyposis, asthma, and chronic sinusitis. Its IL-4 and IL-13 pathway inhibitors represent advancements in inflammatory airway disease management.
- Amgen: Amgen focuses on immunology and inflammation therapeutics, leveraging biologics that target cytokine signaling and immune cell regulation. Its research aims to address respiratory and inflammatory disorders through antibody engineering and precision biologic therapies.
- Bayer: Bayer provides pharmaceutical and consumer health products targeting respiratory and allergy relief, including nasal sprays and anti-inflammatory treatments. The company innovation pipeline includes novel intranasal and anti-inflammatory molecules for upper airway management.
- Sun Pharmaceutical Industries: Sun Pharma manufactures generic and specialty formulations in respiratory and allergy care. The company focuses on affordable access to intranasal corticosteroids, antihistamines, and inflammation-modulating therapies in emerging markets.
- Cipla: Cipla is a global leader in inhalation and nasal delivery systems, offering steroids, bronchodilators, and nasal decongestants. Its emphasis on innovative drug-device combinations strengthens its role in respiratory and allergy treatment segments.
- Takeda Pharmaceutical: Takedas immunology and inflammation division develops biologic and targeted small-molecule therapies for chronic inflammatory airway diseases. The companys focus on gut-lung axis and immune pathway research reflects its integrated approach to systemic inflammation.
- Lyra Therapeutics: Lyra develops bioabsorbable drug-eluting implants for chronic rhinosinusitis and nasal inflammation. Its LYR-210 and LYR-220 programs deliver continuous, localized therapy directly to sinus tissue, offering a novel alternative to systemic treatments.
- Allakos: Allakos focuses on monoclonal antibody therapies targeting eosinophilic and mast-cell-driven inflammation. Its investigational biologic lirentelimab (AK002) shows promise in treating chronic rhinosinusitis and allergic inflammatory diseases.
- Sandoz (Novartis Sandoz Division): Sandoz develops biosimilar and generic anti-inflammatory biologics, expanding patient access to therapies for respiratory and immune-mediated conditions. Its work in biosimilar monoclonal antibodies supports affordability and accessibility in chronic inflammatory disease management.
Recent Developments
- In Oct 2024, GSK announced positive Phase III results for its ultra-long-acting biologic Depemokimab in the ANCHOR-1 & ANCHOR-2 trials for CRSwNP.(Source: https://www.gsk.com)
- In June 2025, Dupixent (dupilumab) presented head-to-head Phase IV data showing superiority over Xolair (omalizumab) in CRSwNP with co-existing asthma.(Source: https://www.sanofi.com)
Nasal Polyposis Treatment Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
