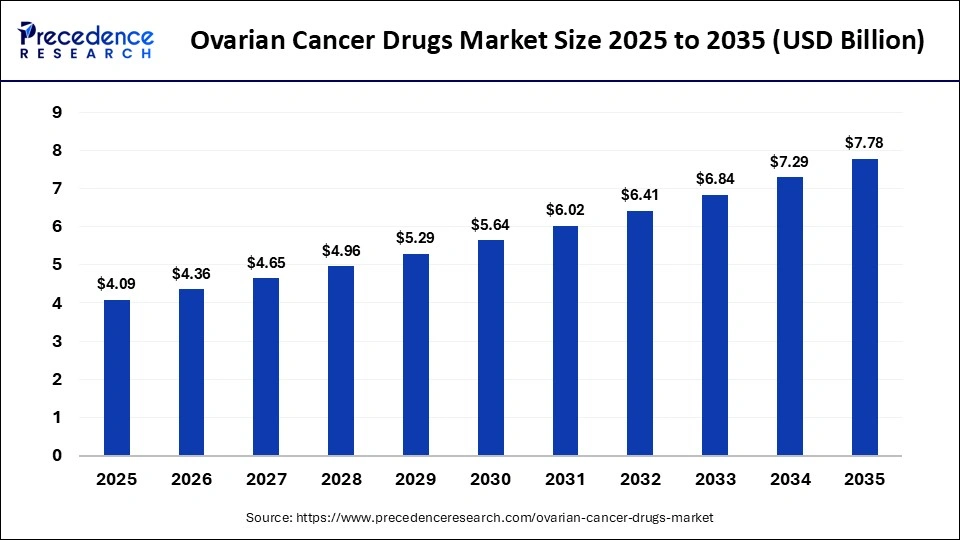
What is the Ovarian Cancer Drugs Market Size in 2026?
The global ovarian cancer drugs market size accounted for USD 4.09 billion in 2025 and is predicted to increase from USD 4.36 billion in 2026 to approximately USD 7.78 billion by 2035, expanding at a CAGR of 6.64% from 2026 to 2035. The market is driven by the rising prevalence of ovarian cancer and the increasing adoption of targeted therapies.
Key Takeaways
- North America dominated the ovarian cancer drugs market with a share of 44.0% in 2025.
- Asia Pacific is expected to grow at the fastest rate in the market during the forecast period.
- By therapeutic class, the PARP inhibitors segment registered its dominance in the global market in 2025.
- By therapeutic class, the PD-L1/PD-1 Inhibitors segment is expected to grow at the fastest rate during the forecast period.
- By treatment type, the targeted therapy segment held a dominant position in the market with a share of 44.3% in 2025.
- By treatment type, the chemotherapy segment is the second-largest shareholder and is expected to grow at a significant rate between 2026 and 2035.
- By tumor type, the epithelial ovarian cancer segment dominated the global market in 2025.
- By tumor type, the germ cell ovarian tumors segment is expected to expand at the fastest CAGR during the forecast period.
- By distribution channel, the hospital pharmacies segment accounted for a 57.1% share in the market in 2025.
- By distribution channel, the online pharmacies segment is expected to grow at the fastest rate during the forecast period.
Market Overview
The ovarian cancer drugs market involves researching, producing, and marketing medications that treat ovarian cancer, which causes death among the highest number of women with gynecological cancers. Patient treatment has changed over time with the introduction of specialized therapies, including anti-angiogenic drugs like bevacizumab and poly (ADP-ribose) polymerase (PARP) inhibitors, which are linked to better outcomes and longer progression-free survival. The market's growth is driven by increased use of targeted therapies, supportive regulatory approvals, and more investment in research and development. The market is expanding in emerging regions due to favorable government policies, regulatory support, and improved healthcare infrastructure.
How is AI Influencing the Ovarian Cancer Drugs Market?
Artificial intelligence is revolutionizing the ovarian cancer drugs market by advancing drug discovery and development. AI also helps in the development of personalized treatment regimens. The genomics, proteomics, and clinical trial data sets are analyzed with AI algorithms in order to find new drug targets and determine the effects expected on a patient to speed up the development of targeted therapies like PARP inhibitors. Machine learning models can be applied to optimize the clinical trial design as well as to define the appropriate patient population and predict the treatment outcomes in order to reduce the costs and schedules. Integrating AI algorithms into diagnostic solutions improves the early detection process through an improved analysis of the images and biomarker evaluation.
Ovarian Cancer Drugs Market Trends
- The integration of artificial intelligence is streamlining drug discovery, clinical trial design, and personalized therapies, enabling faster innovation, lower costs, and improved outcomes for ovarian cancer patients.
- There is a growing shift toward multi-modal treatment approaches, with clinicians increasingly adopting combination regimens, such as PARP inhibitors alongside chemotherapy or immunotherapy, to overcome resistance and enhance survival rates.
- The recurrence and resistance associated with conventional chemotherapy are driving the development of novel therapies, particularly for hard-to-treat ovarian cancer subtypes, including selective glucocorticoid receptor antagonists.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 4.09 Billion |
| Market Size in 2026 | USD 4.36 Billion |
| Market Size by 2035 | USD 7.78 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 6.64% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Therapeutic Class, Treatment Type, Tumor Type, Distribution Channel, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Rising Prevalence of Ovarian Cancer and Increasing R&D
The rising global prevalence of the disease, particularly among the aging female population, is driving the ovarian cancer drugs market. Advances in targeted therapies, such as PARP inhibitors and anti-angiogenic drugs like bevacizumab, have significantly improved progression-free survival and overall treatment outcomes, boosting their adoption. Additionally, increasing research and development efforts by pharmaceutical companies are accelerating new drug discovery and the shift toward personalized treatment approaches.
Restraint
High Treatment Costs and Accessibility Challenges
High treatment costs, particularly for targeted and biologic therapies such as PARP inhibitors, limit accessibility for patients in low- and middle-income regions. Inadequate reimbursement coverage and disparities in healthcare infrastructure across emerging markets further restrict widespread adoption. Additionally, side effects associated with both traditional chemotherapy and targeted treatments can reduce patient compliance and hinder long-term use.
Opportunity
Demand for Personalized Medicine
The rising demand for personalized medicine creates immense opportunities in the ovarian cancer drugs market. Advances in molecular study and genomic profiling are leading to the development of personalized treatments based on the profile of an individual patient, which enhances patient outcomes. Artificial intelligence is also streamlining drug discovery, optimizing clinical trials, and helping in the early detection of cancer, which significantly boosts the market.
Segment Insights
Therapeutic Class Insights
The PARP Inhibitors Segment Led the Ovarian Cancer Drugs Market?
The PARP inhibitors segment registered its dominance in the market while holding a major share in 2025 due to their strong efficacy in patients with BRCA-mutated and homologous recombination deficiency (HRD)-positive ovarian cancer. Their ability to significantly improve progression-free survival has led to widespread use as maintenance therapy following platinum-based chemotherapy. Additionally, growing regulatory approvals and inclusion in clinical treatment guidelines are further supporting market expansion.
The PD-L1/PD-1 inhibitors segment is expected to grow at the fastest rate during the forecast period, owing to the increasing demand for immunotherapy. These inhibitors are particularly effective in treating resistant and recurrent cases by activating the body's immune system to target tumor cells. Additionally, increasing awareness among oncologists and patients about the benefits of immunotherapy is likely to further boost their adoption.
The angiogenesis inhibitors segment is expected to grow at a notable rate during the projection period, as they inhibit tumor blood vessel formation and slow cancer progression. Agents such as bevacizumab are widely used in both first- and second-line treatment of ovarian cancer, contributing to an expanding patient base. Additionally, the increasing preference for combination regimens involving targeted therapies is further driving adoption.
The antibody drug conjugates (ADCs) segment is expected to grow at a considerable rate throughout the forecast period, as they offer a targeted approach by delivering cytotoxic agents directly to cancer cells through specific antibodies. This mechanism makes them particularly effective in managing recurrent cases that are resistant to conventional chemotherapy. Furthermore, increasing research and development activities are expanding the ADC pipeline, supporting market growth.
Treatment Type Insights
Ovarian Cancer Drugs Market Share, By Treatment Type, 2025-2035 (%)
| Treatment Type | 2025 | 2035 |
| Targeted Therapy | 44.30% | 51.00% |
| Chemotherapy | 41.20% | 34.50% |
| Immunotherapy | 8.50% | 11.00% |
| Others | 6.00% | 3.50% |
The Targeted Therapy Segment Held a 44.3% Market Share in 2025
The targeted therapy segment dominated the ovarian cancer drugs market by holding a major share of 44.3% in 2025. This is mainly due to the increased usage of Folate Receptor alpha (FRa) targeted ADCs and PARP inhibitors. Targeted therapies are developed to specifically recognize and destroy cancer cells without harming normal cells, which significantly enhances treatment outcomes with fewer side effects. Targeted therapy with drugs like bevacizumab, which is an angiogenesis inhibitor, is effective in reducing and delaying the growth of progressive epithelial ovarian cancers.
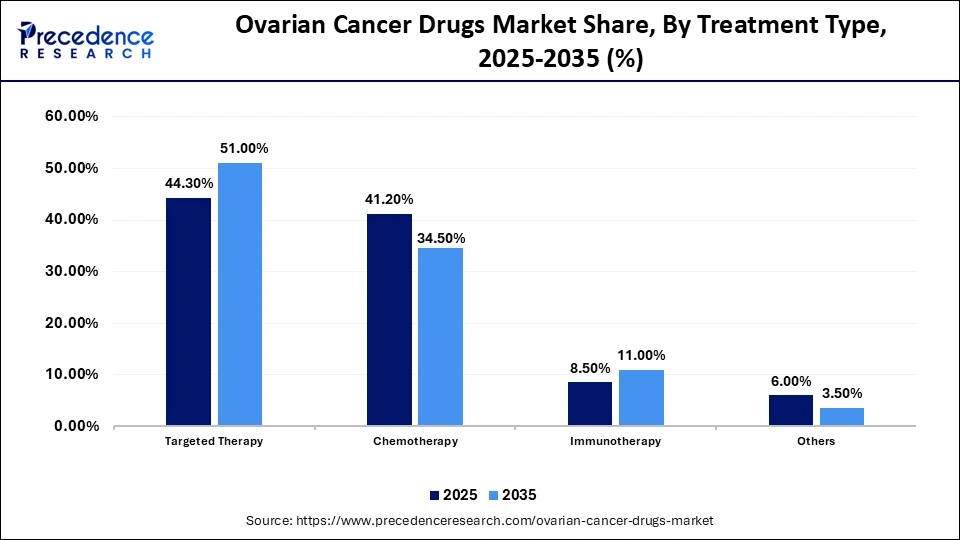
The chemotherapy segment held a 41.2% share of the market in 2025 due to its widespread usage as the first-line backbone treatment for ovarian cancer. Its proven effectiveness, combined with strong clinical acceptance, continues to boost its demand, especially among newly diagnosed patients. Additionally, chemotherapy remains essential in combination regimens, often used alongside targeted therapies to enhance treatment outcomes.
The immunotherapy segment held a 8.5% share of the market in 2025 and is poised for notable growth due to its promising results in improving response rate. Immunotherapy provides a new mechanism whereby the patient's immune system is used to attack cancerous cells. The growth of the segment is also driven by rising investments by pharmaceutical firms in the production of novel immuno-oncology therapies.
Tumor Type Insights
Why Did the Epithelial Ovarian Cancer Segment Dominate the Market in 2025?
The epithelial ovarian cancer segment dominated the ovarian cancer drugs market with a major share in 2025. This is mainly due to its high incidence rate, as it is the most common type of ovarian cancer in the world. Most of the treatments that have been approved, including PARP, angiogenesis inhibitors, and targeted ADCs, are particularly effective for epithelial ovarian cancer. The high prevalence of this kind of tumor is contributing to the driving need for advanced and combination therapeutic approaches.
The germ cell ovarian tumors segment is expected to grow at the fastest rate during the forecast period, driven by increasing research into rare ovarian cancer subtypes. These tumors primarily affect younger women, creating a demand for therapies that preserve fertility while improving long-term outcomes. Growth in this segment is further supported by the development of innovative and targeted treatments specific to germ cell malignancies.
The stromal cell ovarian tumors segment is expected to grow at a notable rate during the forecast period, driven by increased clinical focus on rare ovarian cancers with distinct molecular profiles. Development of targeted and personalized therapies is advancing to address these slow-growing tumors. Enhanced diagnostic capabilities further support growth by enabling earlier detection and more effective treatment.
Distribution Channel Insights
Ovarian Cancer Drugs Market Share, By Distribution Channel, 2025-2035 (%)
| Distribution Channel | 2025 | 2035 |
| Hospital Pharmacy | 57.10% | 52.00% |
| Retail Pharmacy | 34.80% | 38.00% |
| Online Pharmacy | 8.10% | 10.00% |
The Hospital Pharmacies Segment Held a 57.1% Share of the Market in 2025
The hospital pharmacies segment dominated the ovarian cancer drugs market with the largest share of 57.1% in 2025, as hospitals play a critical role in the first-line oncology treatments and IV drugs administration. Hospital pharmacies ensure patients have timely access to complex ovarian cancer therapies and are closely integrated with oncology departments to support treatment monitoring and follow-up care.
The retail pharmacies segment held a 34.8% share of the market in 2025, driven by the growing use of oral PARP inhibitors and other maintenance therapies. Retail pharmacies provide patients with convenient access to long-term medications without hospital visits. Additionally, partnerships with healthcare providers and insurance companies help expand the availability of ovarian cancer treatments through retail channels.
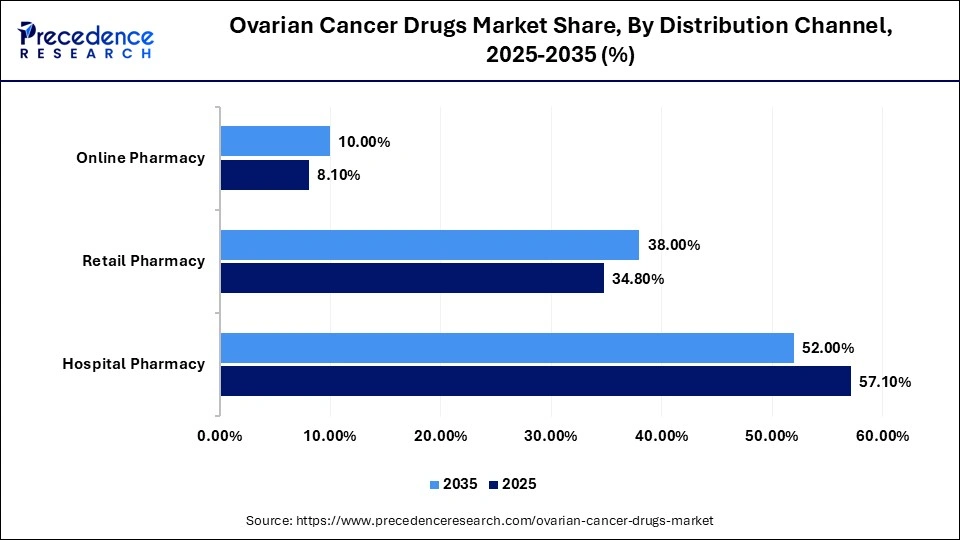
The online pharmacies segment held a 8.1% share of the market in 2025 due to the increased preference of patients for convenience and digital healthcare. Online platforms allow patients to order prescribed medications from home without visiting a hospital or pharmacy. Expanded services such as home delivery and teleconsultations further support the growth of this segment.
Regional Insights
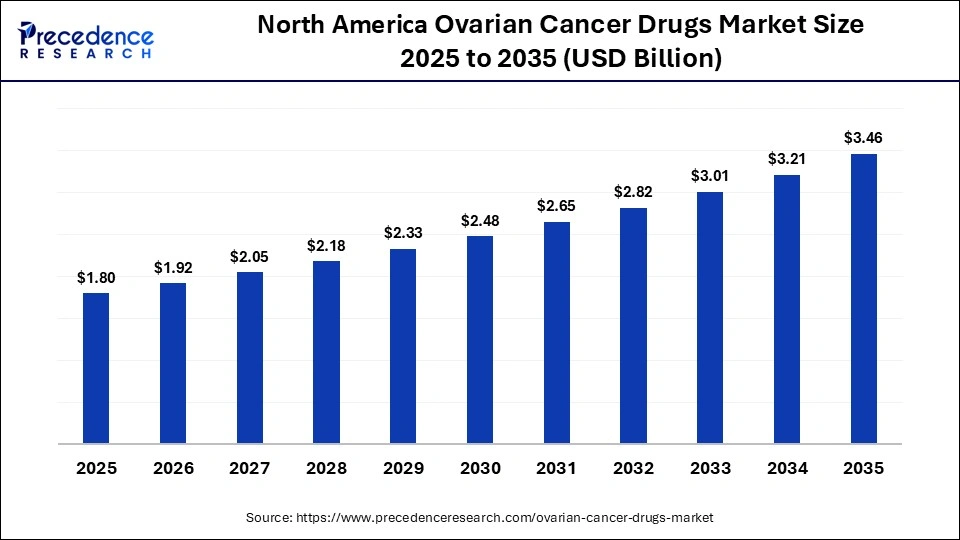
North America Ovarian Cancer Drugs Market Size and Growth 2026 to 2035
The North America ovarian cancer drugs market size is estimated at USD 1.80 billion in 2025 and is projected to reach approximately USD 3.46 billion by 2035, with a 6.75% CAGR from 2026 to 2035.
Why Did North America Lead the Global Ovarian Cancer Drugs Market?
North America dominated the ovarian cancer drugs market with a major share of 44.0% in 2025 because of the high healthcare spending and the widespread availability of approved treatments by the FDA, including PARP inhibitors and antibody-drug conjugates (ADCs). The region also benefits from its well-established healthcare system and advanced medical services, which ensure timely access to treatments, early diagnosis, and improved patient outcomes. There is a strong emphasis on the development of novel cancer treatments. The presence of leading pharmaceutical companies, research institutions, and universities is also supporting the region's dominance by facilitating the development of novel treatments.
U.S. Ovarian Cancer Drugs Market Size and Growth 2026 to 2035
The U.S. ovarian cancer drugs market size is calculated at USD 1.35 billion in 2025 and is expected to reach nearly USD 2.61 billion in 2035, accelerating at a strong CAGR of 6.81% between 2026 and 2035.
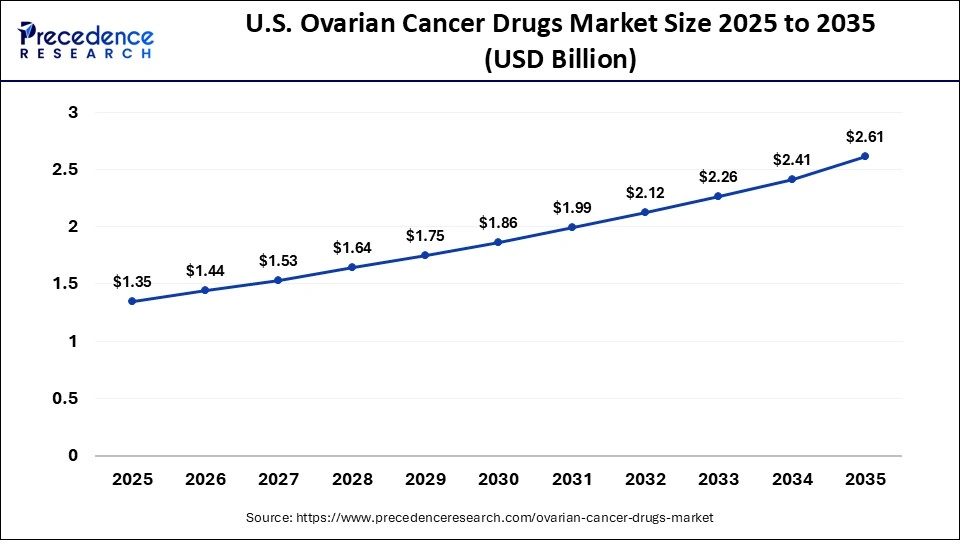
U.S. Market Analysis
The U.S. ovarian cancer drugs market is growing due to the high prevalence of ovarian cancer and increasing awareness of early diagnosis and treatment options. Advanced healthcare infrastructure, strong adoption of innovative therapies like PARP inhibitors, immunotherapies, and targeted treatments, along with significant investment in research and development by pharmaceutical companies, are driving market expansion. Additionally, favorable reimbursement policies and robust clinical trial activity further support growth in the U.S. market.
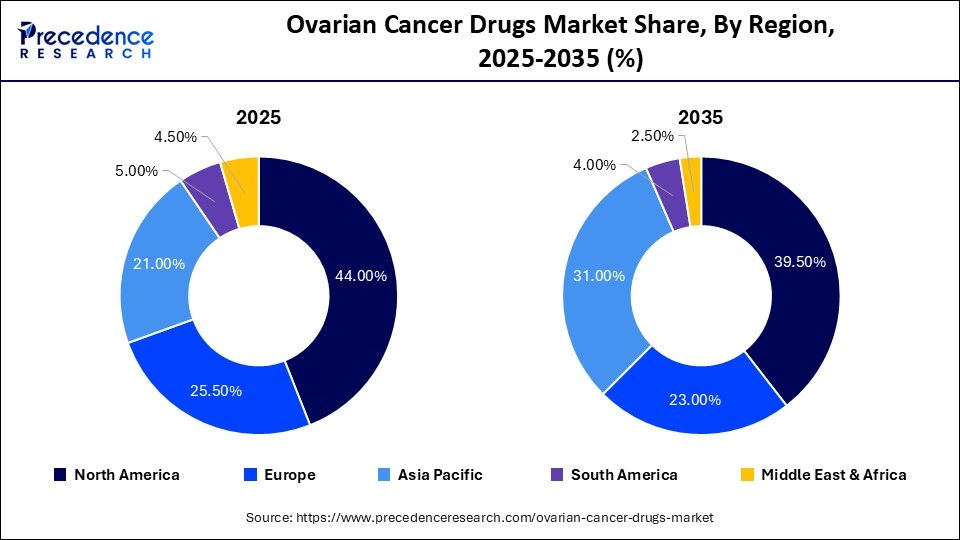
Why is Asia Pacific Undergoing the Fastest Growth in the Ovarian Cancer Drugs Market?
Asia Pacific is expected to grow at the fastest CAGR of 8.0% in the coming years, driven by rising ovarian cancer incidence and improved access to advanced treatments in countries like China and India. A large patient population and increasing disease prevalence create strong demand for effective therapies. Market growth is further supported by government initiatives promoting cancer awareness and screening, increased investment in pharmaceutical research and clinical trials, and the entry of international pharmaceutical companies, along with greater availability of affordable generics and targeted treatments.
Ovarian Cancer Drugs Market Value Chain Analysis
- R&D
This stage focuses on developing novel drugs, including PARP inhibitors, angiogenesis inhibitors, and antibody-drug conjugates, which improve the efficacy and favorable outcome of the treatment.
Key Players: AstraZeneca, GlaxoSmithKline, and Clovis Oncology
- Clinical Trials and Regulatory Approvals
Ovarian cancer medications need to undergo clinical trials and regulatory approvals to ensure the safety, effectiveness, and availability of the drugs are achieved.
Key Players: Pfizer, Merck & Co., and Corcept Therapeutics Incorporated
- Distribution to Hospitals & Pharmacies
Manufactured drugs are then distributed to hospitals, retail pharmacies, and online pharmacies. This network helps in patient compliance and continuity, especially in IV interventions and oral maintenance treatments.
Key Players: McKesson Corporation, AmerisourceBergen, and Cardinal Health
Ovarian Cancer Drugs Market Companies
- AstraZeneca plc (UK)
- F. Hoffmann-La Roche Ltd.
- GlaxoSmithKline plc (GSK) (UK)
- AbbVie Inc. (USA)
- Merck & Co., Inc. (USA)
- Pfizer Inc. (USA)
- Bristol Myers Squibb (BMS) (USA)
- Clovis Oncology (USA)
- Johnson & Johnson (Janssen) (USA)
- Eli Lilly and Company (USA)
- Boehringer Ingelheim International GmbH (Germany)
- Eisai Co., Ltd. (Japan)
- Amgen Inc. (USA)
- Genmab A/S (Denmark)
- Daiichi Sankyo Company, Limited (Japan)
Recent Developments
- In March 2026, the U.S. FDA approved Corcept Therapeutics Incorporated's Lifyorli (relacorilant) in combination with nab-paclitaxel for the treatment of adults with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer. (Source: https://ir.corcept.com)
- In August 2025, Allarity Therapeutics received Fast Track designation from the FDA for stenoparib, an experimental dual PARP and WNT inhibitor of advanced ovarian cancer. This label facilitates the rapid advancement and testing of the treatment in patients who have few treatment choices.(Source: https://allarity.com)
- In May 2025, the FDA granted accelerated approval to Verastem's Avmapki Fakzynja Co-pack (avutometinib and defactinib) for adults with KRAS-mutated recurrent low-grade serous ovarian cancer (LGSOC) who have previously received systemic therapy.(Source: https://www.fda.gov)
Segments Covered in the Report
By Therapeutic Class
- PARP Inhibitors
- Angiogenesis Inhibitors
- PD-L1/PD-1 Inhibitors (Immunotherapy)
- Antibody-Drug Conjugates (ADCs)
By Treatment Type
- Targeted Therapy
- Chemotherapy (Platinum-based, Taxanes, etc.)
- Hormonal Therapy
- Radiation Therapy
By Tumor Type
- Epithelial Ovarian Cancer (Serous, Endometrioid, Clear Cell, Mucinous)
- Germ Cell Ovarian Tumors
- Stromal Cell Ovarian Tumors
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
