Respiratory Syncytial Virus (RSV) Diagnostic Market Revenue to Attain USD 4.34 Bn by 2035
Respiratory Syncytial Virus (RSV) Diagnostic Market Revenue and Trends 2026 to 2035
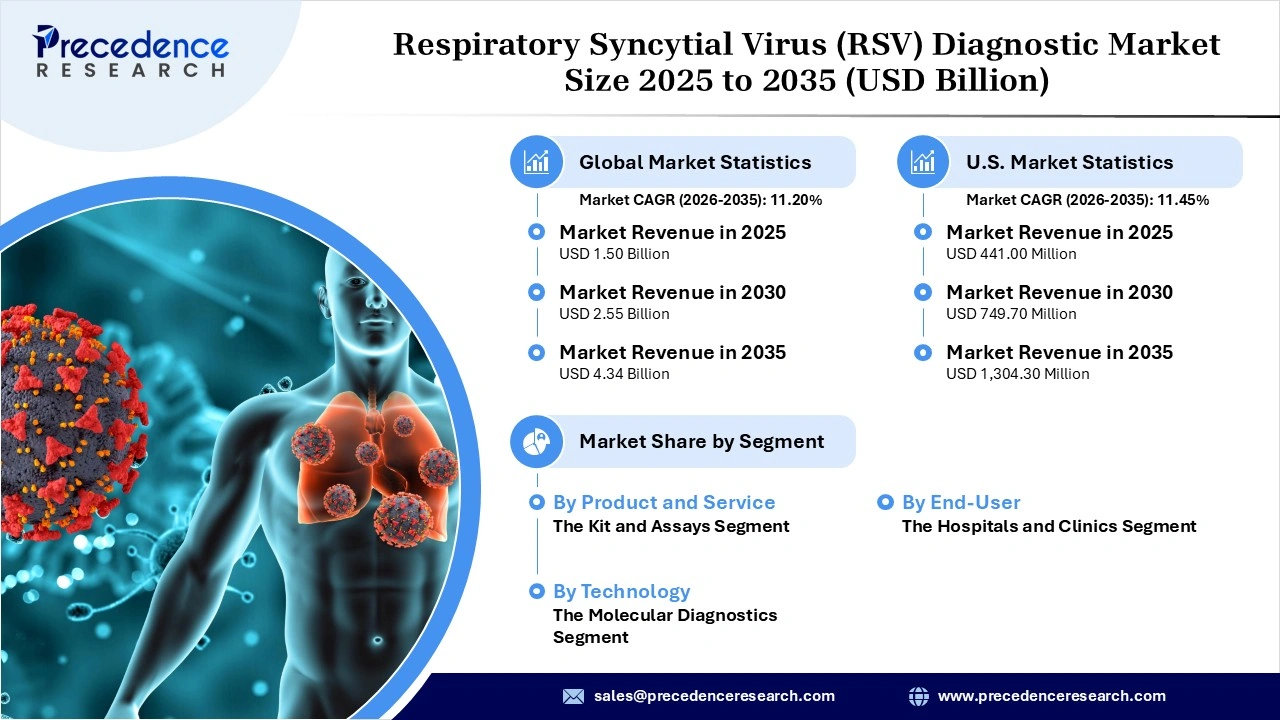
The global respiratory syncytial virus (RSV) diagnostic market revenue was valued at USD 1.50 billion in 2025 and is expected to attain around USD 4.34 billion by 2035, growing at a CAGR of 11.20% during forecast period. The market for diagnostics related to respiratory syncytial virus (RSV) is progressively establishing its importance within the clinical landscape, driven by increasing efforts in infection monitoring, an expanded application of multiplex molecular assays, and a gradual transition towards expedited point-of-care testing. This approach transforms detection into a primary response instead of relying on delayed confirmations.
What are the Growth Factors of the Respiratory Syncytial Virus (RSV) Diagnostic Market?
Rapid and ongoing technological innovation is driving advances in the RSV diagnostic market. Growing hospitalization in infants and elderly patients necessitated early initiation of rapid and precise detection systems compared to symptom assessment alone. At the same time, there is an expansion in the application of multiplex molecular panels for RSV detection in combination with other respiratory pathogens throughout a testing period, and the reduction of time spent and uncertainty related to detection. Moreover, decentralized testing methods are slowly becoming a part of standard care practices, as point-of-care tools start making their presence felt in clinics that until now depended on central laboratory facilities. Combined with the continuing growth of surveillance initiatives, this sector sees increases driven less by a trend and more by need.
Segment Insights
- By product and service, the kit and assays segment accounted for a considerable revenue share in the market in 2025, largely as a result of its reliance on ready-to-use formats. Since such tools were still quite reliable and simple to use, they were still commonly used in laboratories to be part of the normal workflow for RSV detection as opposed to an alternative.
- By technology, the molecular diagnostics segment accounted for a considerable revenue share of 55.0% in the market in 2025, largely due to demand for high-sensitivity PCR methods and multiplex panels. The ability to check for RSV while detecting influenza and COVID-19 simultaneously not just shortened time but also increased efficiency, making a full respiratory test almost automatic.
- By end-user, the hospitals and clinics segment held a major revenue share of 43.1% in the market in 2025, and this was mainly driven by the fact that such facilities are major responders to serious RSV cases in 2025. Given the growth of testing schedules due to children and the elderly being hospitalized, this context has led to these settings serving as crucial diagnostic sites in which timely returns are essential for clinical needs, instead of convenience.
Regional Insights
North America dominated the global respiratory syncytial virus diagnostic market with a market share of 39.2% in 2025, due to a rapid rise in cases coupled with an established diagnostic culture. Robust surveillance systems and the early integration of automated molecular technologies ensured that testing was regular, whether routine or reactive. The US was one of the central actors in this trend, resulting in the establishment of comprehensive screening and advanced laboratory networks, making RSV detection not only routine in respiratory seasons but also virtually standard practice, continuing to underscore the region’s continued leadership.
Asia-Pacific holds the market share of 22.5% market share and is expected to grow at the fastest CAGR in the market during the forecast period, driven by the progress made in the health care system. Hospital expansion and increased focus on diagnostics are helping to shift RSV testing to broader use in hospitals. China emerges as one of the key growth drivers, with strong investments in lab capacity, and India sees a gradual development in its status as hospitals digitalize their diagnostics, giving a gradual approach and change from fragmented testing environments to more integrated and responsive systems.
Respiratory Syncytial Virus (RSV) Diagnostic Market Coverage
| Report Attribute | Key Statistics |
| Market Revenue in 2025 | USD 1.50 Billion |
| Market Revenue by 2035 | USD 4.34 Billion |
| CAGR from 2026 to 2035 | 11.20% |
| Quantitative Units | Revenue in USD million/billion, Volume in units |
| Largest Market | North America |
| Base Year | 2025 |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and the Middle East & Africa |
Recent Development
- In August 2025, Roche expanded its presence in respiratory diagnostics, adding its TAGS-based multiplex PCR assay that allows for simultaneous diagnosis of RSV, influenza A/B, and SARS-CoV-2, using cobas systems. This test brought not just one further panel, but even optimized laboratory workflows with a single versatile assay that used multiple targets as part of an increasing trend toward cohesive high-throughput diagnostic solutions.
Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape overview @ https://www.precedenceresearch.com/sample/8321
You can place an order or ask any questions, please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344