What is U.S. Point of Care Infectious Disease Testing Market Size?
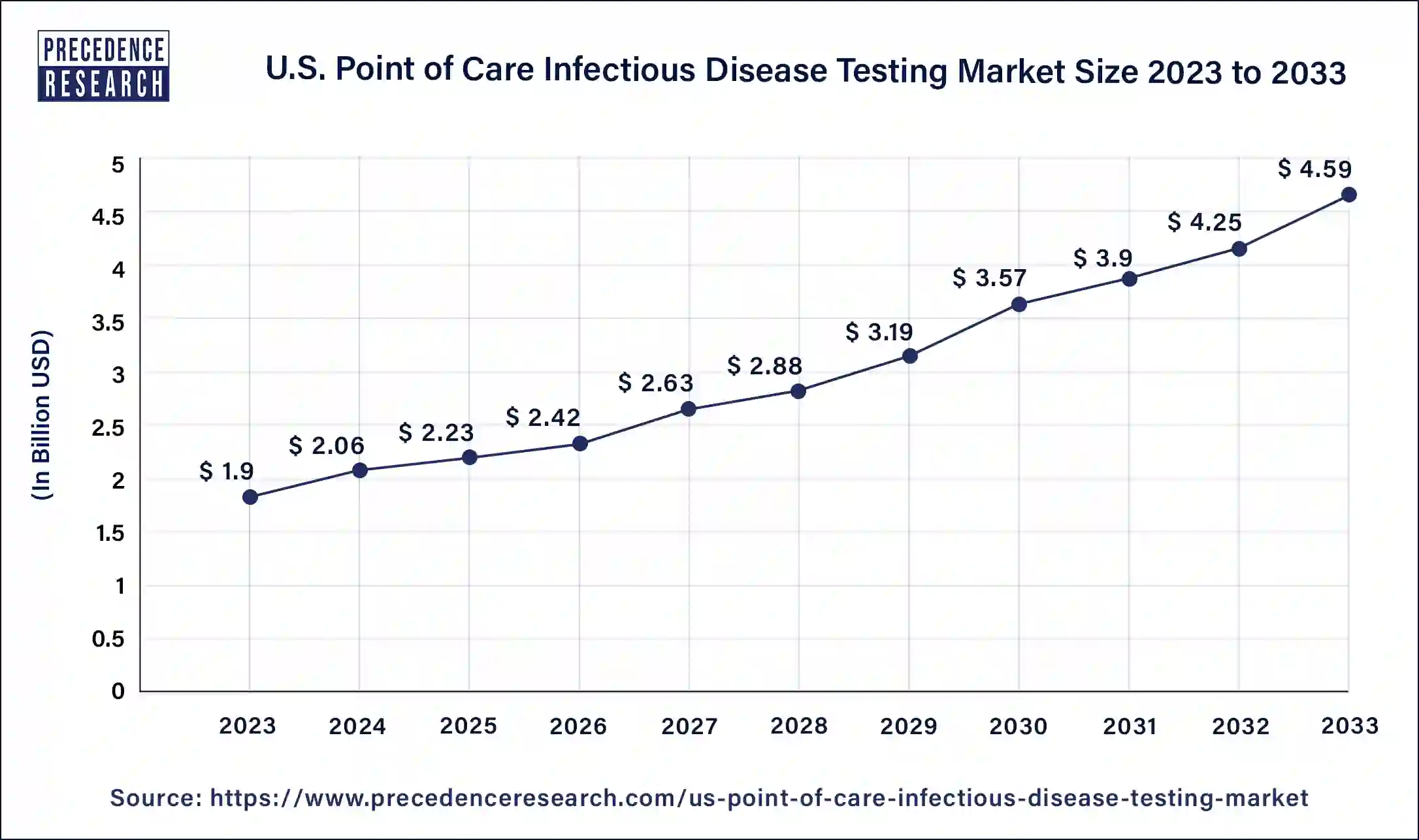
The U.S. point of care infectious disease testing market size is calculated at USD 2.23 billion in 2025 and is expected to reach around USD 5.27 billion by 2035. The market is expanding at a solid CAGR of 8.98% over the forecast period 2026 to 2035. The U.S. point of care infectious disease testing market is driven by the rising need for quick diagnosis to start therapy early.
Market Highlights
- By disease, the influenza/flu segment has held a major revenue share of 24.42% in 2025.
- By disease, the respiratory syncytial virus (RSV) segment is the second largest in the market in 2025.
- By end-user, the hospitals segment dominated market with the largest revenue share of 38.56% in 2025.
What is the Role of AI in the US Point of Care Infectious Disease Testing Market?
Artificial intelligence (AI) based POCT offers significant benefit over traditional methods including improved workflow efficiency, higher accuracy, and faster results. In diagnostics, AI algorithms can analyze medical images like CT scans, X-rays, and MRIs, identifying subtle anomalies that may be missed by the human eye. This aids in the early detection of diseases like cancer and allows timely interventions to improve patient outcomes.
AI based models has ability to analyze vast clinical and experimental datasets may allow precise disease diagnosis, treatment prognosis, and the prediction or development of new anti-microbial compounds. AI in healthcare benefits include support mental health, assists in improving surgery, leads to better data driven decision within the healthcare system, helps in prevention and control of many diseases, and helps in prediction of many risks and diseases.
Market Overview
Since infections can spread from person to person and throughout populations, pathogenic microbes pose a threat to public health and the economy and cause infectious diseases. Effective diagnostic tools are required to deliver precise and timely assistance for case identification, transmission disruption, and proper treatment administration. Point of care (POC) tests function as a personal " radar " and deliver actionable data close to the patient.
Point of care tests (POCT) detect a range of biomarkers associated with infectious disorders, such as viral particles, nucleic acids, proteins , and antibodies. These include compact molecular diagnostic systems, lateral flow assays, microfluidics , plasmonic technologies, and paper-based assays, among others. POCTs provide the cornerstone of infectious illness diagnosis and therapy that is "patient centralized."
Point-of-care testing (POCT) has become increasingly popular due to changes in health management models, advances in interdisciplinary technologies, and growing public awareness of disease prevention. Numerous affordable, lightweight, and quick POCT devices have been developed to support patient prognostic improvement, disease control, and health management.
U.S. Point of Care Infectious Disease Testing Market Data and Statistics
- In May 2023, Sensible Diagnostics created a tiny, inexpensive sample-to-answer device to complete PCR in ten minutes. The company plans to introduce its system with affordable test cartridges in early 2025, first concentrating on point-of-care testing for infectious diseases.
- According to the World Health Organization, by the end of 2022, an estimated 39.0 million people were HIV positive.
U.S. Point of Care Infectious Disease Testing Market Growth Factors
- Growing awareness among people for point of care testing.
- Technological innovations in point-of-care testing devices.
- Increasing prevalence of infectious diseases.
- Focus on rapid diagnostics in hospitals and clinics.
U.S. Point of Care Infectious Disease Testing Market Trends
- Advances in Point-of-Care Disease Diagnostics: The advancements include immunoassays, molecular nucleic acid amplification tests, and CRISPR-based platforms. Moreover, the integration of AI and other advanced technologies is delivering the point of care.
- Technological Advances: These include biosensors, microfluidics, CRISPR-based assays, isothermal amplification, and AI-enhanced platforms, which are expanding the capabilities of point-of-care diagnostics. Moreover, cloud-based reporting, digital connectivity, and the integration with electronic health systems and real-time surveillance are also expanding the U.S. point of care infectious disease testing market.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 2.23 Billion |
| Market Size in 2026 | USD 2.42 Billion |
| Market Size by 2035 | USD 5.27 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 8.98% |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Disease and End-user |
Market Dynamics
Drivers
Latest trends of consumer-centric services coupled with high demand for portable and rapid testing devices
Consumer-centric services prioritize patient's wants and preferences and place greater emphasis on user experience, convenience, and accessibility. This translates into an increasing need for testing solutions in infectious disease testing that are simple to use, yield fast findings, and can be carried out outside of conventional healthcare facilities like clinics and hospitals.
Numerous point-of-care testing devices that address these needs have been developed due to the growth of consumer-centric services. Because these devices are frequently small, lightweight, and easy to use, people can conduct tests independently or with little help from medical experts. This accessibility facilitates early diagnosis and treatment of infectious diseases and gives people more control over their healthcare. This drives the growth of the U.S. point of care infectious disease testing market.
Restraint
Limitations associated with point of care testing
The former frequently show lower sensitivity and specificity when comparing point-of-care testing to laboratory-based assays. This indicates that false-positive and false-negative test findings are more frequent, which might result in incorrect diagnosis and treatment. The reduced accuracy of POCT can be a significant problem in the case of infectious diseases, where precise diagnosis is essential for patient treatment and public health. The operator's proficiency and expertise can impact the accuracy of point-of-care tests.
Errors and incorrect diagnoses can be caused by improper procedures or unskilled people interpreting the results. Although it might be resource-intensive, ensuring healthcare providers using POCT devices have the necessary training and proficiency is crucial.
Opportunity
Need for better and regular monitoring systems in near-patient settings
Infectious disease prevalence remains a concern for world health. Epidemics of HIV/AIDS, hepatitis, influenza, and sexually transmitted infections (STIs) in the United States underscore the need for prompt and efficient diagnostic services. New point-of-care test instruments that provide quick and precise findings are the product of technological breakthroughs.
These gadgets are becoming more affordable, portable, and user-friendly, making them appropriate for usage in various near-patient environments, including community health centers, clinics, and emergency rooms. This opens an opportunity for the growth of the U.S. point of care infectious disease testing market.
Segment Insights
Disease Insights
The influenza/flu segment underwent notable growth in the market during 2025.
- In April 2025, the launch of the Metrix COVID/Flu Test the second assay on the Metrix Molecular Platform for use at the point of care and in the home was announced by SEKISUI Diagnostics, a global medical diagnostics manufacturer. The Metrix COVID/Flu Test is a molecular test intended for the qualitative detection and differentiation of RNA from SARS-CoV-2, Influenza-A and Influenza-B from anterior nares swab samples.
The respiratory syncytial virus (RSV) segment is expected to grow at the fastest rate in the market during the forecast period of 2026 to 2035.
- In May 2025, the launch of a new rapid diagnostic testing tool to detect respiratory syncytial virus (RSV) in professional healthcare settings was announced by SEKISUI Diagnostics, a global medical diagnostics manufacturer. The OSOM RSV Test is a rapid immunochromatographic assay for the qualitative detection of RSV nucleoprotein antigen in anterior nasal swab specimens from patients with signs and symptoms of respiratory infections in just 15 minutes.
U.S Point of Care Infectious Disease Testing Market Revenue (USD Mn), By Disease, 2024 to 2025
| By Disease | 2023 | 2024 | 2025 |
| Pneumonia or Streptococcus-associated infections | 314.52 | 341.46 | 370.85 |
| Respiratory syncytial virus (RSV) POC | 410.21 | 442.37 | 477.25 |
| TB and drug-resistant TB POC | 234.64 | 253.29 | 273.54 |
| Influenza/Flu POC | 465.85 | 509.89 | 558.25 |
| HIV POC | 155.6 | 168.59 | 182.73 |
| Others (Clostridium difficile POC, HBV POC, etc.) | 327.81 | 349.15 | 371.94 |
End-user Insights
The hospitals segment enjoyed a prominent position in the market during 2024 and is set to experience the fastest rate of the market growth from 2026 to 2035.
- In June 2025, next-generation CMAT Advantage to Accelerate Chronic Disease Detection and Transform Point of Care Diagnostics was launched by Brina Medical (US) LLC.
U.S Point of Care Infectious Disease Testing Market Revenue (USD Mn), By End User 2023-2025
| By End User | 2023 | 2024 | 2025 |
| Hospitals | 735.96 | 800.29 | 870.58 |
| Clinics | 344.82 | 372.12 | 401.75 |
| Home | 166.98 | 179.37 | 192.76 |
| Assisted Living Healthcare Facilities | 192.77 | 209.36 | 227.48 |
| Laboratories | 310.44 | 336.35 | 364.57 |
| Others (Ambulatory Surgical Centers, etc.) | 157.65 | 167.24 | 177.42 |
Country-level Insights
U.S. Point of Care Infectious Disease Testing Market Analysis
The U.S. healthcare industry is massively expanding due to the growing preference for decentralized care, and technological advancements like miniaturized biosensors, multiplex testing panels, and CRISPR-based molecular testing. In May 2025, the Association for Diagnostics and Laboratory Medicine introduced changes to point-of-care regulatory requirements in the year 2025. The US Centers for Medicare and Medicaid Services (CMS) and the College of American Pathologists (CAP) are renowned as the leading testing providers, which hold a sharper focus on accuracy and proficiency testing.
Value Chain Analysis
- R&D
This stage revolves around molecular diagnostics, clinical performance studies, advanced biosensors, CRISPR, and regulatory submission.
Key Players: Abbott Laboratories, Danaher Corporation, F. Hoffmann-La Roche Ltd, Becton, Dickinson and Company, Thermo Fisher Scientific Inc., bioMérieux SA.
- Distribution to Hospitals, Pharmacies
This stage prioritizes reimbursement models for outpatient and remote monitoring, and direct supply by retail clinics and pharmacies.
Key Players: Cardinal Health, McKesson Corporation, Cencora, Henry Schein, Abbott Laboratories, F. Hoffmann-La Roche Ltd, Becton, Dickinson and Company, Danaher Corporation, QuidelOrtho Corporation.
- Patient Support and Services
This stage focuses on unified enrollment and access, AI-enhanced interpretation, guidance, integrated care, surveillance, and regulatory and quality standards.
Key Players: Abbott, F. Hoffmann-La Roche Ltd, Danaher Corporation, Becton, Dickinson and Company, QuidelOrtho Corporation, Siemens Healthineers.
- Clinical Trials and Regulatory Approvals
This stage includes platform and decentralized clinical trials, usability studies, and the U.S. FDA regulatory processes.
Key Players: Abbott Laboratories, F. Hoffmann-La Roche Ltd, Danaher Corporation, Becton, Dickinson and Company, Siemens Healthineers, bioMérieux, Inflammatix, Cue Health, and NowDiagnostics.
- Formulation and Final Dosage Preparation
This stage is transitioning due to enzyme and primer optimization, nanoparticle integration, microfluidic integration, and multiplexing.
Key Players: QuidelOrtho, Mylab Discovery Solutions, Becton, Dickinson and Company, Abbott Laboratories, F. Hoffmann-La Roche Ltd, Danaher Corporation, bioMérieux.
U.S. Point of Care Infectious Disease Testing Market Companies

- Beckton Dickinson: BD Veritor Plus System, InfoWiFi modules, etc.
- Roche: cobas liat SARS-CoV-2, influenza A/B & RSV assay, cobas liat Bordetella test, cobas respiratory flex, cobas liat CT/NG & MG, cobas pulse system.
- Biomerieux: BIOFIRE SPOTFIRE system, BIOFIRE SPOTFIRE R/ST panel, BIOFIRE SPOTFIRE R/ST panel mini, VETFIRE, SpinChip diagnostics, next-generation POC.
- Siemens: CLINITEST rapid COVID-19 antigen test, urinalysis systems, inflammation & sepsis monitoring, blood gas analysis.
- Bio-Rad: BioPlex 2200 system, exact diagnostics molecular QC, Geenius confirmatory solution, StarBright dyes, chromogenic media.
Recent Developments
- In July 2025, to expand INDICAID portfolio, Phase Scientific, a fastest growing biotech company known for its science driven innovation and commitment to improve healthcare outcomes announced that it has entered into an exclusive U.S. distribution agreement with Lumos Diagnostics for FebriDx, a rapid point of care (POC) test that aids in the diagnosis of bacterial acute respiratory infection and differentiation from non-bacterial etiology in approximately 10 minutes using a single drop of blood.
(Source: PHASE Scientific Launches FebriDx in U.S. to Expand INDICAID Portfolio ) - In December 2024, a molecular diagnostic test for the H5N1 virus was launched by Amid reports of the first severe human case of bird flu in the U.S. According to the clinical testing giant, its H5 test can be used to help check potential human exposure, as the virus has begun to spread through outbreaks at poultry farms and dairy herds across the country as well as among wild bird populations.
(Source: Labcorp launches bird flu test amid first severe case in the US )
Segments Covered in the Report
By Disease
- Pneumonia Or Streptococcus Associated Infections
- Respiratory Syncytial Virus (RSV)
- TB and Drug Resistant TB POC
- Influenza/Flu POC
- HIV POC
- Others
By End-user
- Hospitals
- Clinics
- Home
- Assisted Living Healthcare Facilities
- Laboratories
- Others
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
